Medicine:X-linked congenital stationary night blindness
| X-linked congenital stationary night blindness | |
|---|---|
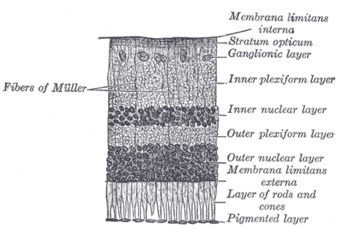 | |
| Malfunction in transmission from the photoreceptors in the outer nuclear layer to bipolar cells in the inner nuclear layer underlies CSNB. |
X-linked congenital stationary night blindness (CSNB) is a rare X-linked non-progressive retinal disorder. It has two forms, complete, also known as type-1 (CSNB1), and incomplete, also known as type-2 (CSNB2), depending on severity. In the complete form (CSNB1), there is no measurable rod cell response to light, whereas this response is measurable in the incomplete form. Patients with this disorder have difficulty adapting to low light situations due to impaired photoreceptor transmission. These patients also often have reduced visual acuity, myopia, nystagmus and strabismus. CSNB1 is caused by mutations in the gene NYX, which encodes a protein involved in retinal synapse formation or synaptic transmission. CSNB2 is caused by mutations in the gene CACNA1F, which encodes a voltage-gated calcium channel CaV1.4.
Not all congenital stationary night blindness (CSNB) are inherited in X-linked pattern. There are also autosomal dominant and recessive inheritance patterns for CSNB.
Symptoms
The X-linked varieties of congenital stationary night blindness (CSNB) can be differentiated from the autosomal forms by the presence of myopia, which is typically absent in the autosomal forms. Patients with CSNB often have impaired night vision, myopia, reduced visual acuity, strabismus and nystagmus. Individuals with the complete form of CSNB (CSNB1) have highly impaired rod sensitivity (reduced ~300x) as well as cone dysfunction. Patients with the incomplete form can present with either myopia or hyperopia.[1]
Cause
CSNB was originally believed to be caused by malfunction in neurotransmission from rods to bipolar cells in the retina. This is due to electroretinogram (ERG) measurements on CSNB patients which show a drastic decrease in the size of the scotopic b-wave in comparison to the a-wave, in CSNB2, or a complete loss of both in CSNB1. The a-wave is believed to represent the response of rods to visual input and remains largely unchanged in CSNB2 patients. The b-wave, however, is believed to result from electrical activity of bipolar cells and is decreased or non-existent in both CSNB1 and 2. CSNB1 patients also show mildly altered cone activity. Further study has demonstrated that the defects found in CSNB patients are better explained by more general defects in both the rod and cone ON-signaling pathways.
Pathophysiology
CSNB1
The complete form of X-linked congenital stationary night blindness, also known as nyctalopia, is caused by mutations in the NYX gene (Nyctalopin on X-chromosome), which encodes a small leucine-rich repeat (LRR) family protein of unknown function.[2][3] This protein consists of an N-terminal signal peptide and 11 LRRs (LRR1-11) flanked by cysteine-rich LRRs (LRRNT and LRRCT). At the C-terminus of the protein there is a putative GPI anchor site. Although the function of NYX is yet to be fully understood, it is believed to be located extracellularly. A naturally occurring deletion of 85 bases in NYX in some mice leads to the "nob" (no b-wave) phenotype, which is highly similar to that seen in CSNB1 patients.[4] NYX is expressed primarily in the rod and cone cells of the retina. There are currently almost 40 known mutations in NYX associated with CSNB1, Table 1., located throughout the protein. As the function of the nyctalopin protein is unknown, these mutations have not been further characterized. However, many of them are predicted to lead to truncated proteins that, presumably, are non-functional.
| Mutation | Position | References | |
|---|---|---|---|
| Nucleotide | Amino acid | ||
| c.?-1_?-61del | 1_20del | Signal sequence | [3] |
| Splicing | Intron 1 | [5] | |
| c.?-63_1443-?del | 21_481del | [3] | |
| c.48_64del | L18RfsX108 | Signal sequence | [5] |
| c.85_108del | R29_A36del | N-terminal LRR | [2] |
| c.G91C | C31S | LRRNT | [3] |
| c.C105A | C35X | LRRNT | [3] |
| c.C169A | P57T | LRRNT | |
| c.C191A | A64E | LRR1 | [6] |
| c.G281C | R94P | LRR2 | [7] |
| c.301_303del | I101del | LRR2 | [3] |
| c.T302C | I101T | LRR2 | [7] |
| c.340_351del | E114_A118del | LRR3 | [3][5] |
| c.G427C | A143P | LRR4 | [3] |
| c.C452T | P151L | LRR4 | [2] |
| c.464_465insAGCGTGCCCGAGCGCCTCCTG | S149_V150dup+P151_L155dup | LRR4 | [2] |
| c.C524G | P175R | LRR5 | [3] |
| c.T551C | L184P | LRR6 | [2] |
| c.556_618delins | H186?fsX260 | LRR6 | [2] |
| c.559_560delinsAA | A187K | LRR6 | [3] |
| c.613_621dup | 205_207dup | LRR7 | [2][3] |
| c.628_629ins | R209_S210insCLR | LRR7 | [2] |
| c.T638A | L213Q | LRR7 | [2] |
| c.A647G | N216S | LRR7 | [2][5] |
| c.T695C | L232P | LRR8 | [2] |
| c.727_738del | 243_246del | LRR8 | [3] |
| c.C792G | N264K | LRR9 | [2] |
| c.T854C | L285P | LRR10 | [2] |
| c.T893C | F298S | LRR10 | [2] |
| c.C895T | Q299X | LRR10 | [5] |
| c.T920C | L307P | LRR11 | [3] |
| c.A935G | N312S | LRR11 | [3] |
| c.T1040C | L347P | LRRCT | [3] |
| c.G1049A | W350X | LRRCT | [2] |
| c.G1109T | G370V | LRRCT | [3] |
| c.1122_1457del | S374RfsX383 | LRRCT | [3][5] |
| c.1306del | L437WfsX559 | C-terminus | [5] |
| LRR: leucine-rich repeat, LRRNT and LRRCT: N- and C-terminal cysteine-rich LRRs. | |||
CSNB2
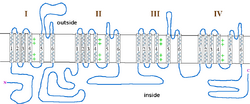
The incomplete form of X-linked congenital stationary night blindness (CSNB2) is caused by mutations in the CACNA1F gene, which encodes the voltage-gated calcium channel CaV1.4 expressed heavily in retina.[8][9] One of the important properties of this channel is that it inactivates at an extremely low rate. This allows it to produce sustained Ca2+ entry upon depolarization. As photoreceptors depolarize in the absence of light, CaV1.4 channels operate to provide sustained neurotransmitter release upon depolarization.[10] This has been demonstrated in CACNA1F mutant mice that have markedly reduced photoreceptor calcium signals.[11] There are currently 55 mutations in CACNA1F located throughout the channel, Table 2 and Figure 1. While most of these mutations result in truncated and, likely, non-functional channels, it is expected that they prevent the ability of light to hyperpolarize photoreceptors. Of the mutations with known functional consequences, 4 produce channels that are either completely non-functional, and two that result in channels which open at far more hyperpolarized potentials than wild-type. This will result in photoreceptors that continue to release neurotransmitter even after light-induced hyperpolarization.
| Mutation | Position | Effect | References | |
|---|---|---|---|---|
| Nucleotide | Amino Acid | |||
| c.C148T | R50X | N-terminus | ||
| c.151_155delAGAAA | R51PfsX115 | N-terminus | [12] | |
| c.T220C | C74R | N-terminus | [12] | |
| c.C244T | R82X | N-terminus | [13][12] | |
| c.466_469delinsGTAGGGGTGCT CCACCCCGTAGGGGTGCTCCACC |
S156VdelPinsGVKHOVGVLH | D1S2-3 | [13][14][15] | |
| Splicing | Intron 4 | [13] | ||
| c.T685C | S229P | D1S4-5 | [12] | |
| c.G781A | G261R | D1-pore | [12] | |
| c.G832T | E278X | D1-pore | ||
| c.904insG | R302AfsX314 | D1-pore | [14] | |
| c.951_953delCTT | F318del | D1-pore | [13] | |
| c.G1106A | G369D | D1S6 | Activates ~20mV more negative than wild-type, increases time to peak current and decreases inactivation, increased Ca2+ permeability. | [8][10][13][12][16] |
| c.1218delC | W407GfsX443 | D1-2 | [9][13][17] | |
| c.C1315T | Q439X | D1-2 | [12] | |
| c.G1556A | R519Q | D1-2 | Decreased expression | [8][18] |
| c.C1873T | R625X | D2S4 | [13][12] | |
| c.G2021A | G674D | D2S5 | [10][13][14] | |
| c.C2071T | R691X | D2-pore | [6] | |
| c.T2258G | F753C | D2S6 | [12] | |
| c.T2267C | I756T | D2S6 | Activates ~35mV more negative than wild-type, inactivates more slowly | |
| Splicing | Intron 19 | [12] | ||
| c.T2579C | L860P | D2-3 | [12] | |
| c.C2683T | R895X | D3S1-2 | [5][6][9][13] | |
| Splicing | Intron 22 | [12][14] | ||
| Splicing | Intron 22 | [12] | ||
| c.C2783A | A928D | D3S2-3 | [10][13] | |
| c.C2905T | R969X | D3S4 | [8][12] | |
| c.C2914T | R972X | D3S4 | [17] | |
| Splicing | Intron24 | [13] | ||
| c.C2932T | R978X | D3S4 | [14] | |
| c.3006_3008delCAT | I1003del | D3S4-5 | [13] | |
| c.G3052A | G1018R | D3S5 | [12] | |
| c.3125delG | G1042AfsX1076 | D3-pore | [13] | |
| c.3166insC | L1056PfsX1066 | D3-pore | [8][9][13][12] | |
| c.C3178T | R1060W | D3-pore | [8][12] | |
| c.T3236C | L1079P | D3-pore | Does not open without BayK, activates ~5mV more negative than wild-type | [12][16] |
| c.3672delC | L1225SfsX1266 | D4S2 | [9][13] | |
| c.3691_3702del | G1231_T1234del | D4S2 | [8][12] | |
| c.G3794T | S1265I | D4S3 | [6] | |
| c.C3886A | R1296S | D4S4 | [6] | |
| c.C3895T | R1299X | D4S4 | [9][13][12] | |
| Splicing | Intron 32 | [12] | ||
| c.C4075T | Q1359X | D4-pore | [8][12] | |
| c.T4124A | L1375H | D4-pore | Decreased expression | [8][12][18] |
| Splicing | Intron 35 | [12] | ||
| c.G4353A | W1451X | C-terminus | Non-functional | [9][10][13][16] |
| c.T4495C | C1499R | C-terminus | [12] | |
| c.C4499G | P1500R | C-terminus | [12] | |
| c.T4523C | L1508P | C-terminus | [12] | |
| Splicing | intron 40 | [13] | ||
| c.4581delC | F1528LfsX1535 | C-terminus | ||
| c.A4804T | K1602X | C-terminus | [8][12] | |
| c.C5479T | R1827X | C-terminus | [12] | |
| c.5663delG | S1888TfsX1931 | C-terminus | [13] | |
| c.G5789A | R1930H | C-terminus | [6] | |
Genetics
Only three rhodopsin mutations have been found associated with congenital stationary night blindness (CSNB).[19] Two of these mutations are found in the second transmembrane helix of rhodopsin at Gly-90 and Thr-94. Specifically, these mutations are the Gly90Asp [20] and the Thr94Ile, which has been the most recent one reported.[21] The third mutation is Ala292Glu, and it is located in the seventh transmembrane helix, in proximity to the site of retinal attachment at Lys-296.[22] Mutations associated with CSNB affect amino acid residues near the protonated Schiff base (PSB) linkage. They are associated with changes in conformational stability and the protonated status of the PSB nitrogen.[23]
Footnotes
- ↑ "Evidence for genetic heterogeneity in X-linked congenital stationary night blindness". Am J Hum Genet 62 (4): 865–875. 1998. doi:10.1086/301781. PMID 9529339.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 "Mutations in NYX, encoding the leucine-rich proteoglycan nyctalopin, cause X-linked complete congenital stationary night blindness". Nat Genet 26 (3): 319–323. 2000. doi:10.1038/81619. PMID 11062471.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 3.15 3.16 "The complete form of X-linked congenital stationary night blindness is caused by mutations in a gene encoding a leucine-rich repeat protein". Nat Genet 26 (3): 324–327. 2000. doi:10.1038/81627. PMID 11062472.
- ↑ "Identification of the gene and the mutation responsible for the mouse nob phenotype". Invest Ophthalmol Vis Sci 44 (1): 378–384. 2003. doi:10.1167/iovs.02-0501. PMID 12506099.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 "Mutations in the CACNA1F and NYX genes in British CSNBX families". Hum Mutat 21 (2): 169–169. 2003. doi:10.1002/humu.9106. PMID 12552565.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 Cite error: Invalid
<ref>tag; no text was provided for refs namedZeitz_2005 - ↑ 7.0 7.1 "CSNB1 in Chinese families associated with novel mutations in NYX". J Hum Genet 51 (7): 634–640. 2006. doi:10.1007/s10038-006-0406-5. PMID 16670814.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 "An L-type calcium-channel gene mutated in incomplete X-linked congenital stationary night blindness". Nat Genet 19 (3): 260–263. 1998. doi:10.1038/940. PMID 9662399.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 "Loss-of-function mutations in a calcium-channel alpha1-subunit gene in Xp11.23 cause incomplete X-linked congenital stationary night blindness". Nat Genet 19 (3): 264–267. 1998. doi:10.1038/947. PMID 9662400.
- ↑ 10.0 10.1 10.2 10.3 10.4 "The CACNA1F gene encodes an L-type calcium channel with unique biophysical properties and tissue distribution". J Neurosci 24 (7): 1707–1718. 2004. doi:10.1523/JNEUROSCI.4846-03.2004. PMID 14973233.
- ↑ "Mutation of the calcium channel gene Cacna1f disrupts calcium signaling, synaptic transmission and cellular organization in mouse retina". Hum Mol Genet 14 (20): 3035–3046. 2005. doi:10.1093/hmg/ddi336. PMID 16155113.
- ↑ 12.00 12.01 12.02 12.03 12.04 12.05 12.06 12.07 12.08 12.09 12.10 12.11 12.12 12.13 12.14 12.15 12.16 12.17 12.18 12.19 12.20 12.21 12.22 12.23 12.24 12.25 12.26 12.27 12.28 "Thirty distinct CACNA1F mutations in 33 families with incomplete type of XLCSNB and Cacna1f expression profiling in mouse retina". Eur J Hum Genet 10 (8): 449–456. 2002. doi:10.1038/sj.ejhg.5200828. PMID 12111638.
- ↑ 13.00 13.01 13.02 13.03 13.04 13.05 13.06 13.07 13.08 13.09 13.10 13.11 13.12 13.13 13.14 13.15 13.16 13.17 13.18 Cite error: Invalid
<ref>tag; no text was provided for refs namedBoycott_2001 - ↑ 14.0 14.1 14.2 14.3 14.4 "Novel CACNA1F mutations in Japanese patients with incomplete congenital stationary night blindness". Invest Ophthalmol Vis Sci 42 (7): 1610–6. 2001. PMID 11381068.
- ↑ "Retinal and optic disc atrophy associated with a CACNA1F mutation in a Japanese family". Arch Ophthalmol 121 (7): 1028–1033. 2003. doi:10.1001/archopht.121.7.1028. PMID 12860808.
- ↑ 16.0 16.1 16.2 "Congenital stationary night blindness type 2 mutations S229P, G369D, L1068P, and W1440X alter channel gating or functional expression of Ca(v)1.4 L-type Ca2+ channels". J Neurosci 25 (1): 252–259. 2005. doi:10.1523/JNEUROSCI.3054-04.2005. PMID 15634789.
- ↑ 17.0 17.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedAllen_2003 - ↑ 18.0 18.1 "Effects of congenital stationary night blindness type 2 mutations R508Q and L1364H on Cav1.4 L-type Ca2+ channel function and expression". J Neurochem 96 (6): 1648–1658. 2006. doi:10.1111/j.1471-4159.2006.03678.x. PMID 16476079.
- ↑ Pere Garriga, and Joan Manyosa. The eye photoreceptor protein rhodopsin. Structural implications for retinal disease. Volume 528, Issues 1–3, 25 September 2002, Pages 17–22.
- ↑ V.R. Rao, G.B. Cohen and D.D. Oprian Nature 367 (1994), pp. 639–642.
- ↑ N. al-Jandal, G.J. Farrar, A.S. Kiang, M.M. Humphries, N. Bannon, J.B. Findlay, P. Humphries and P.F. Kenna Hum. Mutat. 13 (1999), pp. 75–81.
- ↑ T.P. Dryja, E.L. Berson, V.R. Rao and D.D. Oprian Nat. Genet. 4 (1993), pp. 280–283.
- ↑ P.A. Sieving, J.E. Richards, F. Naarendorp, E.L. Bingham, K. Scott and M. Alpern Proc. Natl. Acad. Sci. USA 92 (1995), pp. 880–884.
External links
| Classification | |
|---|---|
| External resources |