Physics:Isotopes of americium
| |||||||||||||||||||||||||||||||||||
Americium (95Am) is an artificial element, and thus a standard atomic weight cannot be given. Like all artificial elements, it has no known stable isotopes. The first isotope to be synthesized was 241Am in 1944. The artificial element decays by ejecting alpha particles. Americium has an atomic number of 95 (the number of protons in the nucleus of the americium atom). Despite 243Am being an order of magnitude longer lived than 241Am, the former is harder to obtain than the latter as more of it is present in spent nuclear fuel.
Nineteen radioisotopes of americium, ranging from 229Am to 247Am have been characterized; another isotope, 223Am, has also been reported but is unconfirmed. The most stable isotopes are 243Am with a half-life of 7,350 years and 241Am with a half-life of 432.6 years. All of the remaining radioactive isotopes have half-lives that are less than seven days, the majority of which are shorter than two hours. This element also has fourteen meta states, with the most stable being 242m1Am (half-life 141 years). This isomer is unusual in that its half-life is far longer than that of the ground state of the same isotope.
List of isotopes
| Nuclide [n 1] |
Z | N | Isotopic mass (u) [n 2][n 3] |
Half-life |
Decay mode [n 4] |
Daughter isotope |
Spin and parity [n 5][n 6] |
|---|---|---|---|---|---|---|---|
| Excitation energy[n 6] | |||||||
| 223Am[n 7] | 95 | 128 | 223.04584(32)# | 10(9) ms | α | 219Np | 9/2–# |
| 229Am | 95 | 134 | 229.04528(11) | 1.8(15) s | α | 225Np | 5/2–# |
| 230Am | 95 | 135 | 230.04603(15)# | 40(9) s | β+ (<70%) | 230Pu | 1–# |
| β+, SF (>30%) | (various) | ||||||
| 231Am[2] | 95 | 136 | 231.04553(32)# | 75+137 −30 s |
β+ (~83%) | 231Pu | 5/2–# |
| α (~17%) | 227Np | ||||||
| 232Am | 95 | 137 | 232.04661(32)# | 1.31(4) min | β+ (97%) | 232Pu | 1–# |
| α? (3%) | 228Np | ||||||
| β+, SF (0.069%) | (various) | ||||||
| 233Am | 95 | 138 | 233.04647(12)# | 3.2(8) min | β+? (95.5%) | 233Pu | 5/2–# |
| α (4.5%) | 229Np | ||||||
| 234Am | 95 | 139 | 234.04773(17)# | 2.32(8) min | β+ (99.95%) | 234Pu | 0–# |
| α (0.039%) | 230Np | ||||||
| β+, SF (0.0066%) | (various) | ||||||
| 235Am | 95 | 140 | 235.04791(6) | 10.3(6) min | β+ (99.60%) | 235Pu | 5/2−# |
| α (0.40%) | 231Np | ||||||
| 236Am | 95 | 141 | 236.04943(13)# | 3.6(1) min | β+ | 236Pu | 5− |
| α (4×10−3%) | 232Np | ||||||
| 236mAm | 50(50)# keV | 2.9(2) min | β+ | 236Pu | (1−) | ||
| 237Am | 95 | 142 | 237.05000(6)# | 73.6(8) min | β+ (99.975%) | 237Pu | 5/2− |
| α (0.025%) | 233Np | ||||||
| 238Am | 95 | 143 | 238.05198(6) | 98(3) min | β+ | 238Pu | 1+ |
| α (1.0×10−4%) | 234Np | ||||||
| 238mAm | 2500(200)# keV | 35(18) μs | SF | (various) | |||
| 239Am | 95 | 144 | 239.0530227(21) | 11.9(1) h | EC (99.990%) | 239Pu | 5/2− |
| α (0.010%) | 235Np | ||||||
| 239mAm | 2500(200) keV | 163(12) ns | SF | (various) | (7/2+) | ||
| 240Am | 95 | 145 | 240.055298(15) | 50.8(3) h | β+ | 240Pu | (3−) |
| α (1.9×10−4%) | 236Np | ||||||
| 240mAm | 3000(200) keV | 940(40) μs | SF | (various) | |||
| 241Am | 95 | 146 | 241.0568273(12) | 432.6(6) y | α | 237Np | 5/2− |
| SF (3.6×10−10%) | (various) | ||||||
| 241mAm | 2200(200) keV | 1.2(3) μs | SF | (various) | |||
| 242Am | 95 | 147 | 242.0595474(12) | 16.02(2) h | β− (82.7%) | 242Cm | 1− |
| EC (17.3%) | 242Pu | ||||||
| 242m1Am | 48.60(5) keV | 141(2) y | IT (99.55%) | 242Am | 5− | ||
| α (0.45%) | 238Np | ||||||
| SF (<4.7×10−9%) | (various) | ||||||
| 242m2Am | 2200(80) keV | 14.0(10) ms | SF | (various) | (2+, 3−) | ||
| IT | 242Am | ||||||
| 243Am | 95 | 148 | 243.0613799(15) | 7350(9) y | α | 239Np | 5/2− |
| SF (3.7×10−9%) | (various) | ||||||
| 243mAm | 2300(200) keV | 5.5(5) μs | SF | (various) | |||
| 244Am | 95 | 149 | 244.0642829(16) | 10.01(3) h | β− | 244Cm | (6−) |
| 244m1Am | 89.3(16) keV | 26.13(43) min | β− (99.96%) | 244Cm | 1+ | ||
| EC (0.0364%) | 244Pu | ||||||
| 244m2Am | 2000(200)# keV | 0.90(15) ms | SF | (various) | |||
| 244m3Am | 2200(200)# keV | ~6.5 μs | SF | (various) | |||
| 245Am | 95 | 150 | 245.0664528(20) | 2.05(1) h | β− | 245Cm | 5/2+ |
| 245mAm | 2400(400)# keV | 0.64(6) μs | SF | (various) | |||
| 246Am | 95 | 151 | 246.069774(19)# | 39(3) min | β− | 246Cm | 7− |
| 246m1Am | 30(10)# keV | 25.0(2) min | β− | 246Cm | 2(−) | ||
| 246m2Am | 2000(800)# keV | 73(10) μs | SF | (various) | |||
| 247Am | 95 | 152 | 247.07209(11)# | 23.0(13) min | β− | 247Cm | 5/2# |
- ↑ mAm – Excited nuclear isomer.
- ↑ ( ) – Uncertainty (1σ) is given in concise form in parentheses after the corresponding last digits.
- ↑ # – Atomic mass marked #: value and uncertainty derived not from purely experimental data, but at least partly from trends from the Mass Surface (TMS).
- ↑
Modes of decay:
CD: Cluster decay EC: Electron capture IT: Isomeric transition SF: Spontaneous fission - ↑ ( ) spin value – Indicates spin with weak assignment arguments.
- ↑ 6.0 6.1 # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).
- ↑ The discovery of this isotope is uncertain due to disagreements between theoretical predictions and reported experimental data.[1]
Actinides vs fission products
Actinides and fission products by half-life
| ||||||||
|---|---|---|---|---|---|---|---|---|
| Actinides[3] by decay chain | Half-life range (y) |
Fission products of 235U by yield<ref>Specifically from thermal neutron fission of U-235, e.g. in a typical nuclear reactor.</ref> | ||||||
| 4n | 4n+1 | 4n+2 | 4n+3 | |||||
| 4.5–7% | 0.04–1.25% | <0.001% | ||||||
| 228Ra№ | 4–6 | † | 155Euþ | |||||
| 244Cmƒ | 241Puƒ | 250Cf | 227Ac№ | 10–29 | 90Sr | 85Kr | 113mCdþ | |
| 232Uƒ | 238Puƒ | 243Cmƒ | 29–97 | 137Cs | 151Smþ | 121mSn | ||
| 248Bk[4] | 249Cfƒ | 242mAmƒ | 141–351 |
No fission products | ||||
| 241Amƒ | 251Cfƒ[5] | 430–900 | ||||||
| 226Ra№ | 247Bk | 1.3 k – 1.6 k | ||||||
| 240Pu | 229Th | 246Cmƒ | 243Amƒ | 4.7 k – 7.4 k | ||||
| 245Cmƒ | 250Cm | 8.3 k – 8.5 k | ||||||
| 239Puƒ | 24.1 k | |||||||
| 230Th№ | 231Pa№ | 32 k – 76 k | ||||||
| 236Npƒ | 233Uƒ | 234U№ | 150 k – 250 k | ‡ | 99Tc₡ | 126Sn | ||
| 248Cm | 242Pu | 327 k – 375 k | 79Se₡ | |||||
| 1.53 M | 93Zr | |||||||
| 237Npƒ | 2.1 M – 6.5 M | 135Cs₡ | 107Pd | |||||
| 236U | 247Cmƒ | 15 M – 24 M | 129I₡ | |||||
| 244Pu | 80 M |
... nor beyond 15.7 M years[6] | ||||||
| 232Th№ | 238U№ | 235Uƒ№ | 0.7 G – 14.1 G | |||||
|
Legend for superscript symbols | ||||||||
Americium-241

Americium-241 (alpha emitter, half-life 432.6 years) is the most common isotope of americium in nuclear waste.[7] It is the isotope used in normal ionization smoke detectors, which work as an ionization chamber. It is a potential fuel for long-lifetime radioisotope thermoelectric generators, with a half-life longer than that of the standard plutonium-238 (87.7 years) or the alternative strontium-90 (28.91 years). Its decay heat is 0.114 W/g; its rate of spontaneous fission 1.2/g/s.
The alpha decay of 241Am is accompanied by a significant emission of gamma rays. Its presence in plutonium is determined by the original concentration of 241Pu (which decays to it) and the sample age. Older samples of plutonium containing plutonium-241 build up 241Am, and chemical separation of americium from such plutonium (e.g. during reworking of plutonium pits) may be required.
Americium-242m
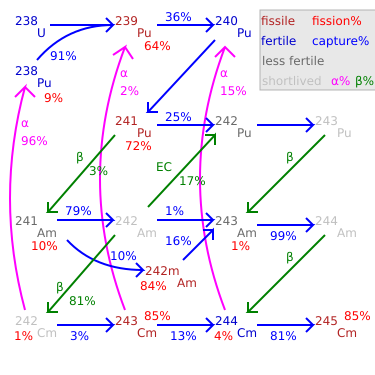
Fission percentage is 100 minus shown percentages.
Total rate of transmutation varies greatly by nuclide.
245Cm–248Cm are long-lived with negligible decay.
Americium-242m (half-life 141 years) is one of the rare cases, like 108mAg, 166mHo, 180mTa, 186mRe, 192mIr, 210mBi, 212mPo and others, where a higher-energy nuclear isomer is more stable than its ground state. While that ground state, 242Am, decays with half-life 16.02 hours by beta emission or electron capture, in a typical example of spin-forbiddenness the isomer does not decay by those modes, but falls to the ground state very slowly (99.55% of decays) or emits an alpha particle (0.45%, partial half-life 31 ky).
242mAm is fissile with a low critical mass, comparable to that of 239Pu.[9] It has a very high fission cross section, and is quickly destroyed if it is produced in a nuclear reactor. It has been investigated whether this isotope could be used for a novel type of nuclear rocket.[10][11]
Americium-243

Americium-243, an alpha emitter, has a half-life of 7350 years[12], the longest of all americium isotopes. It is formed in the nuclear fuel cycle mainly by neutron capture on plutonium-242 followed by beta decay.[13] Production increases exponentially with increasing burnup as a total of 5 neutron captures on 238U are required. If MOX-fuel is used, particularly MOX-fuel high in 241Pu and 242Pu, more americium overall and more 243Am will be produced.
It decays by either emitting an alpha particle (decay energy 5.439 MeV)[14] to become 239Np, which then quickly goes to 239Pu, or, very rarely, spontaneous fission. The fission rate is about 60% that of americium-241 or about 0.7/g/s.[15]
As for the other americium isotopes, and more generally for all alpha emitters, 243Am is carcinogenic in case of internal contamination after being inhaled or ingested. 243Am also presents a risk of external irradiation associated with the gamma ray emitted by its short-lived decay product 239Np. The external irradiation risk for the other two americium isotopes (241Am and 242mAm) is less than 10% of that for americium-243.[7]
References
- ↑ Sun, M. D. (2017). "New short-lived isotope 223Np and the absence of the Z = 92 subshell closure near N = 126". Physics Letters B 771: 303–308. doi:10.1016/j.physletb.2017.03.074. Bibcode: 2017PhLB..771..303S. https://www.researchgate.net/publication/317142406.
- ↑ Wang, J. G.Expression error: Unrecognized word "et". (2026). "Decay properties of new isotopes 235Bk and 231Am". Physics Letters B 875 (140365). doi:10.1016/j.physletb.2026.140365.
- ↑ Plus radium (element 88). While actually a sub-actinide, it immediately precedes actinium (89) and follows a three-element gap of instability after polonium (84) where no nuclides have half-lives of at least four years (the longest-lived nuclide in the gap is radon-222 with a half life of less than four days). Radium's longest lived isotope, at 1,600 years, thus merits the element's inclusion here.
- ↑ Milsted, J.; Friedman, A. M.; Stevens, C. M. (1965). "The alpha half-life of berkelium-247; a new long-lived isomer of berkelium-248". Nuclear Physics 71 (2): 299. doi:10.1016/0029-5582(65)90719-4. Bibcode: 1965NucPh..71..299M.
"The isotopic analyses disclosed a species of mass 248 in constant abundance in three samples analysed over a period of about 10 months. This was ascribed to an isomer of Bk248 with a half-life greater than 9 y. No growth of Cf248 was detected, and a lower limit for the β− half-life can be set at about 104 y. No alpha activity attributable to the new isomer has been detected; the alpha half-life is probably greater than 300 y." - ↑ This is the heaviest nuclide with a half-life of at least four years before the "Sea of Instability".
- ↑ Excluding those "classically stable" nuclides with half-lives significantly in excess of 232Th; e.g., while 113mCd has a half-life of only fourteen years, that of 113Cd is nearly eight quadrillion years.
- ↑ 7.0 7.1 "Americium" . Argonne National Laboratory, EVS. Retrieved 25 December 2009.
- ↑ Sasahara, Akihiro; Matsumura, Tetsuo; Nicolaou, Giorgos; Papaioannou, Dimitri (April 2004). "Neutron and Gamma Ray Source Evaluation of LWR High Burn-up UO2 and MOX Spent Fuels". Journal of Nuclear Science and Technology 41 (4): 448–456. doi:10.3327/jnst.41.448. https://www.tandfonline.com/doi/pdf/10.1080/18811248.2004.9715507?needAccess=true.
- ↑ "Critical Mass Calculations for 241Am, 242mAm and 243Am". http://typhoon.jaea.go.jp/icnc2003/Proceeding/paper/6.5_022.pdf.
- ↑ "Extremely Efficient Nuclear Fuel Could Take Man To Mars In Just Two Weeks" (Press release). Ben-Gurion University Of The Negev. December 28, 2000.
- ↑ Ronen, Yigal; Shwageraus, E. (2000). "Ultra-thin 242mAm fuel elements in nuclear reactors". Nuclear Instruments and Methods in Physics Research A 455 (2): 442–451. doi:10.1016/s0168-9002(00)00506-4. Bibcode: 2000NIMPA.455..442R.
- ↑ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf.
- ↑ "Americium-243" . Oak Ridge National Laboratory. Retrieved 25 December 2009.
- ↑ National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory. http://www.nndc.bnl.gov/nudat2/.
- ↑ Calculated from Nubase data.
Sources
- Isotope masses from:
- Half-life, spin, and isomer data selected from the following sources.
- National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory. http://www.nndc.bnl.gov/nudat2/.
- IAEA - Nuclear Data Section. Live Chart of Nuclides. Vienna International Centre.
- Lide, David R., ed (2004). "11. Table of the Isotopes". CRC Handbook of Chemistry and Physics (85th ed.). Boca Raton, Florida: CRC Press. ISBN 978-0-8493-0485-9.
Lua error: Internal error: The interpreter has terminated with signal "24".
Lua error: Internal error: The interpreter has terminated with signal "24".