Chemistry:Americium
Americium is a synthetic chemical element; it has symbol Am and atomic number 95. It is radioactive and a transuranic member of the actinide series in the periodic table, located under the lanthanide element europium and was thus named after the Americas by analogy.[1][2][3]
Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California, at the Metallurgical Laboratory of the University of Chicago, as part of the Manhattan Project. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and released to the public only in November 1945. Most americium is produced by uranium or plutonium being bombarded with neutrons in nuclear reactors – one tonne of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detectors, as well as in neutron sources and industrial gauges. Several unusual applications, such as nuclear batteries or fuel for space ships with nuclear propulsion, have been proposed for the isotope 242mAm, but they are as yet hindered by the scarcity and high price of this nuclear isomer.
Americium is a relatively soft radioactive metal with a silvery appearance. Its most common isotopes are 241Am and 243Am. In chemical compounds, americium usually assumes the oxidation state +3, especially in solutions. Several other oxidation states are known, ranging from +2 to +7, and can be identified by their characteristic optical absorption spectra. The crystal lattices of solid americium and its compounds contain small intrinsic radiogenic defects, due to metamictization induced by self-irradiation with alpha particles, which accumulates with time; this can cause a drift of some material properties over time, more noticeable in older samples.
History

Although americium was likely produced in previous nuclear experiments, it was first intentionally synthesized, isolated and identified in late autumn 1944, at the University of California, Berkeley, by Glenn T. Seaborg, Leon O. Morgan, Ralph A. James, and Albert Ghiorso. They used a 60-inch cyclotron at the University of California, Berkeley.[4] The element was chemically identified at the Metallurgical Laboratory (now Argonne National Laboratory) of the University of Chicago. Following the lighter neptunium, plutonium, and heavier curium, americium was the fourth transuranium element to be discovered. At the time, the periodic table had been restructured by Seaborg to its present layout, containing the actinide row below the lanthanide one. This led to americium being located right below its twin lanthanide element europium; it was thus by analogy named after the Americas: "The name americium (after the Americas) and the symbol Am are suggested for the element on the basis of its position as the sixth member of the actinide rare-earth series, analogous to europium, Eu, of the lanthanide series."[5][6][7]
The new element was isolated from its oxides in a complex, multi-step process. First plutonium-239 nitrate (239PuNO3) solution was coated on a platinum foil of about 0.5 cm2 area, the solution was evaporated and the residue was converted into plutonium dioxide (PuO2) by calcining. After cyclotron irradiation, the coating was dissolved with nitric acid, and then precipitated as the hydroxide using concentrated aqueous ammonia solution. The residue was dissolved in perchloric acid. Further separation was carried out by ion exchange, yielding a certain isotope of curium. The separation of curium and americium was so painstaking that those elements were initially called by the Berkeley group as pandemonium[8] (from Greek for all demons or hell) and delirium (from Latin for madness).[9][10]
Initial experiments yielded four americium isotopes: 241Am, 242Am, 239Am and 238Am. Americium-241 was directly obtained from plutonium upon absorption of two neutrons. It decays by emission of a α-particle to 237Np; the half-life of this decay was first determined as 510±20 years but then corrected to 432.2 years.[11]
- The times are half-lives
The second isotope 242Am was produced upon neutron bombardment of the already-created 241Am. Upon rapid β-decay, 242Am converts into the isotope of curium 242Cm (which had been discovered previously). The half-life of this decay was initially determined at 17 hours, which was close to the presently accepted value of 16.02 h.[11]
The discovery of americium and curium in 1944 was closely related to the Manhattan Project; the results were confidential and declassified only in 1945. Seaborg leaked the synthesis of the elements 95 and 96 on the U.S. radio show for children Quiz Kids five days before the official presentation at an American Chemical Society meeting on 11 November 1945, when one of the listeners asked whether any new transuranium element besides plutonium and neptunium had been discovered during the war.[9] After the discovery of americium isotopes 241Am and 242Am, their production and compounds were patented listing only Seaborg as the inventor.[12] The initial americium samples weighed a few micrograms; they were barely visible and were identified by their radioactivity. The first substantial amounts of metallic americium weighing 40–200 micrograms were not prepared until 1951 by reduction of americium(III) fluoride with barium metal in high vacuum at 1100 °C.[13]
Occurrence

The longest-lived and most common isotopes of americium, 241Am and 243Am, have half-lives of 432.6 and 7,350 years, respectively. Therefore, any primordial americium (americium that was present on Earth during its formation) should have decayed by now. Trace amounts of americium probably occur naturally in uranium minerals as a result of neutron capture and beta decay (238U → 239Pu → 240Pu → 241Am), though the quantities would be tiny and this has not been confirmed.[14][15][16] Extraterrestrial long-lived 247Cm is probably also deposited on Earth and has 243Am as one of its intermediate decay products, but again this has not been confirmed.[16]
Existing americium is concentrated in the areas used for the atmospheric nuclear weapons tests conducted between 1945 and 1980, as well as at the sites of nuclear incidents, such as the Chernobyl disaster. For example, the analysis of the debris at the testing site of the first U.S. hydrogen bomb, Ivy Mike, (1 November 1952, Enewetak Atoll), revealed high concentrations of various actinides including americium; but due to military secrecy, this result was not published until later, in 1956.[17] Trinitite, the glassy residue left on the desert floor near Alamogordo, New Mexico, after the plutonium-based Trinity nuclear bomb test on 16 July 1945, contains traces of americium-241. Elevated levels of americium were also detected at the crash site of a US Boeing B-52 bomber aircraft, which carried four hydrogen bombs, in 1968 in Greenland.[18]
In other regions, the average radioactivity of surface soil due to residual americium is only about 0.01 picocuries per gram (0.37 mBq/g). Atmospheric americium compounds are poorly soluble in common solvents and mostly adhere to soil particles. Soil analysis revealed about 1,900 times higher concentration of americium inside sandy soil particles than in the water present in the soil pores; an even higher ratio was measured in loam soils.[19]
Americium is produced mostly artificially in small quantities, for research purposes. A tonne of spent nuclear fuel contains about 100 grams of various americium isotopes, mostly 241Am and 243Am.[20] Their prolonged radioactivity is undesirable for the disposal, and therefore americium, together with other long-lived actinides, must be neutralized. The associated procedure may involve several steps, where americium is first separated and then converted by neutron bombardment in special reactors to short-lived nuclides. This procedure is well known as nuclear transmutation, but it is still being developed for americium.[21][22]
The transuranic elements up to fermium, including americium, should have been present in the natural nuclear fission reactor at Oklo, but any quantities produced then would have long since decayed away.[23]
Synthesis and extraction
Isotope nucleosynthesis
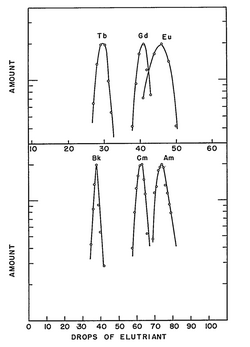
Americium has been produced in small quantities in nuclear reactors for decades, and kilograms of its 241Am and 243Am isotopes have been accumulated by now.[24] Nevertheless, since it was first offered for sale in 1962, its price, about US$1,500 per gram (US$43,000/oz) of 241Am, remains almost unchanged owing to the very complex separation procedure.[25] The heavier isotope 243Am is produced in much smaller amounts; it is thus more difficult to separate, resulting in a higher cost of the order US$100,000–US$160,000 per gram (US$2,800,000–US$4,500,000/oz).[26][27]
Americium is not synthesized directly from uranium – the most common reactor material – but from the plutonium isotope 239Pu. The latter needs to be produced first, according to the following nuclear process:
The capture of two neutrons by 239Pu (a so-called (n,γ) reaction), followed by a β-decay, results in 241Am:
The plutonium present in spent nuclear fuel contains about 12% of 241Pu. Because it beta-decays to 241Am, 241Pu can be extracted and may be used to generate further 241Am.[25] However, this process is rather slow: half of the original amount of 241Pu decays to 241Am after about 15 years, and the 241Am amount reaches a maximum after 70 years.[28]
The obtained 241Am can be used for generating heavier americium isotopes by further neutron capture inside a nuclear reactor. In a light water reactor (LWR), 79% of 241Am converts to 242Am and 10% to its nuclear isomer 242mAm:[note 1][29]
Americium-242 has a half-life of only 16 hours, which makes its further conversion to 243Am extremely inefficient. The latter isotope is produced instead in a process where 239Pu captures four neutrons under high neutron flux:
Metal generation
Most synthesis routines yield a mixture of different actinide isotopes in oxide forms, from which isotopes of americium can be separated. In a typical procedure, the spent reactor fuel (e.g. MOX fuel) is dissolved in nitric acid, and the bulk of uranium and plutonium is removed using a PUREX-type extraction (Plutonium–URanium EXtraction) with tributyl phosphate in a hydrocarbon. The lanthanides and remaining actinides are then separated from the aqueous residue (raffinate) by a diamide-based extraction, to give, after stripping, a mixture of trivalent actinides and lanthanides. Americium compounds are then selectively extracted using multi-step chromatographic and centrifugation techniques[30] with an appropriate reagent. A large amount of work has been done on the solvent extraction of americium. For example, a 2003 EU-funded project codenamed "EUROPART" studied triazines and other compounds as potential extraction agents.[31][32][33][34][35] A bis-triazinyl bipyridine complex was proposed in 2009 as such a reagent is highly selective to americium (and curium).[36] Separation of americium from the highly similar curium can be achieved by treating a slurry of their hydroxides in aqueous sodium bicarbonate with ozone, at elevated temperatures. Both Am and Cm are mostly present in solutions in the +3 valence state; whereas curium remains unchanged, americium oxidizes to soluble Am(IV) complexes which can be washed away.[37]
Metallic americium is obtained by reduction from its compounds. Americium(III) fluoride was first used for this purpose. The reaction was conducted using elemental barium as reducing agent in a water- and oxygen-free environment inside an apparatus made of tantalum and tungsten.[13][38][39]
An alternative is the reduction of americium dioxide by metallic lanthanum or thorium:[39][40]
Physical properties
In the periodic table, americium is located to the right of plutonium, to the left of curium, and below the lanthanide europium, with which it shares many physical and chemical properties. Americium is a highly radioactive element. When freshly prepared, it has a silvery-white metallic lustre, but then slowly tarnishes in air. With a density of 12 g/cm3, americium is less dense than both curium (13.52 g/cm3) and plutonium (19.8 g/cm3); but has a higher density than europium (5.264 g/cm3)—mostly because of its higher atomic mass. Americium is relatively soft and easily deformable and has a significantly lower bulk modulus than the actinides before it: Th, Pa, U, Np and Pu.[41] Its melting point of 1173 °C is significantly higher than that of plutonium (639 °C) and europium (826 °C), but lower than for curium (1340 °C).[40][42]
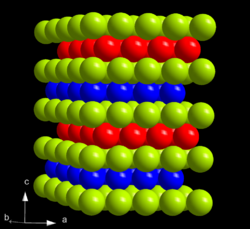
At ambient conditions, americium is present in its most stable α form which has a hexagonal crystal symmetry, and a space group P63/mmc with cell parameters a = 346.8 pm and c = 1124 pm, and four atoms per unit cell. The crystal consists of a double-hexagonal close packing with the layer sequence ABAC and so is isotypic with α-lanthanum and several actinides such as α-curium.[38][42] The crystal structure of americium changes with pressure and temperature. When compressed at room temperature to 5 GPa, α-Am transforms to the β modification, which has a face-centered cubic (fcc) symmetry, space group Fm3m and lattice constant a = 489 pm. This fcc structure is equivalent to the closest packing with the sequence ABC.[38][42] Upon further compression to 23 GPa, americium transforms to an orthorhombic γ-Am structure similar to that of α-uranium. There are no further transitions observed up to 52 GPa, except for an appearance of a monoclinic phase at pressures between 10 and 15 GPa.[41] There is no consistency on the status of this phase in the literature, which also sometimes lists the α, β and γ phases as I, II and III. The β-γ transition is accompanied by a 6% decrease in the crystal volume; although theory also predicts a significant volume change for the α-β transition, it is not observed experimentally. The pressure of the α-β transition decreases with increasing temperature, and when α-americium is heated at ambient pressure, at 770 °C it changes into an fcc phase which is different from β-Am, and at 1075 °C it converts to a body-centered cubic structure. The pressure-temperature phase diagram of americium is thus rather similar to those of lanthanum, praseodymium and neodymium.[43]
As with many other actinides, self-damage of the crystal structure due to alpha-particle irradiation is intrinsic to americium. It is especially noticeable at low temperatures, where the mobility of the produced structure defects is relatively low, by broadening of X-ray diffraction peaks. This effect makes somewhat uncertain the temperature of americium and some of its properties, such as electrical resistivity.[44] So for americium-241, the resistivity at 4.2 K increases with time from about 2 μOhm·cm to 10 μOhm·cm after 40 hours, and saturates at about 16 μOhm·cm after 140 hours. This effect is less pronounced at room temperature, due to annihilation of radiation defects; also heating to room temperature the sample which was kept for hours at low temperatures restores its resistivity. In fresh samples, the resistivity gradually increases with temperature from about 2 μOhm·cm at liquid helium to 69 μOhm·cm at room temperature; this behavior is similar to that of neptunium, uranium, thorium and protactinium, but is different from plutonium and curium which show a rapid rise up to 60 K followed by saturation. The room temperature value for americium is lower than that of neptunium, plutonium and curium, but higher than for uranium, thorium and protactinium.[45]
Americium is paramagnetic in a wide temperature range, from that of liquid helium, to room temperature and above. This behavior is markedly different from that of its neighbor curium which exhibits antiferromagnetic transition at 52 K.[46] The thermal expansion coefficient of americium is slightly anisotropic and amounts to (7.5±0.2)×10−6 /°C along the shorter a axis and (6.2±0.4)×10−6 /°C for the longer c hexagonal axis.[42] The enthalpy of dissolution of americium metal in hydrochloric acid at standard conditions is −620.6±1.3 kJ/mol, from which the standard enthalpy change of formation (ΔfH°) of aqueous Am3+ ion is −621.2±2.0 kJ/mol. The standard potential Am3+/Am0 is −2.08±0.01 V.[47]
Chemical properties
Americium metal readily reacts with oxygen and dissolves in aqueous acids. The most stable oxidation state for americium is +3.[48] The chemistry of americium(III) has many similarities to the chemistry of lanthanide(III) compounds. For example, trivalent americium forms insoluble fluoride, oxalate, iodate, hydroxide, phosphate and other salts.[48] Compounds of americium in oxidation states +2, +4, +5, +6 and +7 have also been studied. This is the widest range that has been observed with actinide elements. The color of americium compounds in aqueous solution is as follows: Am3+ (yellow-reddish), Am4+ (yellow-reddish), AmV
O+
2; (yellow), AmVI
O2+
2 (brown) and AmVII
O5−
6 (dark green).[49][50] The absorption spectra have sharp peaks, due to f-f transitions' in the visible and near-infrared regions. Typically, Am(III) has absorption maxima at ca. 504 and 811 nm, Am(V) at ca. 514 and 715 nm, and Am(VI) at ca. 666 and 992 nm.[51][52][53][54]
Americium compounds with oxidation state +4 and higher are strong oxidizing agents, comparable in strength to the permanganate ion (MnO−
4) in acidic solutions.[55] Whereas the Am4+ ions are unstable in solutions and readily convert to Am3+, compounds such as americium dioxide (AmO2) and americium(IV) fluoride (AmF4) are stable in the solid state.
The pentavalent oxidation state of americium was first observed in 1951.[56] In acidic aqueous solution the AmO+
2 ion is unstable with respect to disproportionation.[57][58][59] The reaction
- 3[AmO
2]+
+ 4H+
→ 2[AmO
2]2+ + Am3+ + 2H
2O
is typical. The chemistry of Am(V) and Am(VI) is comparable to the chemistry of uranium in those oxidation states. In particular, compounds like Li
3AmO
4 and Li
6AmO
6 are comparable to uranates and the ion AmO2+
2 is comparable to the uranyl ion, UO2+
2. Such compounds can be prepared by oxidation of Am(III) in dilute nitric acid with ammonium persulfate.[60] Other oxidising agents that have been used include silver(I,III) oxide,[54] ozone and sodium persulfate.[53]
Chemical compounds
Oxygen compounds
Three americium oxides are known, with the oxidation states +2 (AmO), +3 (Am2O3) and +4 (AmO2). Americium(II) oxide was prepared in minute amounts and has not been characterized in detail.[61] Americium(III) oxide is a red-brown solid with a melting point of 2205 °C.[62] Americium(IV) oxide is the main form of solid americium which is used in nearly all its applications. As most other actinide dioxides, it is a black solid with a cubic (fluorite) crystal structure.[63]
The oxalate of americium(III), vacuum dried at room temperature, has the chemical formula Am2(C2O4)3·7H2O. Upon heating in vacuum, it loses water at 240 °C and starts decomposing into AmO2 at 300 °C, the decomposition completes at about 470 °C.[48] The initial oxalate dissolves in nitric acid with the maximum solubility of 0.25 g/L.[64]
Halides
Halides of americium are known for the oxidation states +2, +3 and +4,[65] where the +3 is most stable, especially in solutions.[66]
| Oxidation state | F | Cl | Br | I |
|---|---|---|---|---|
| +4 | Americium(IV) fluoride AmF4 pale pink |
|||
| +3 | Americium(III) fluoride AmF3 pink |
Americium(III) chloride AmCl3 pink |
Americium(III) bromide AmBr3 light yellow |
Americium(III) iodide AmI3 light yellow |
| +2 | Americium(II) chloride AmCl2 black |
Americium(II) bromide AmBr2 black |
Americium(II) iodide AmI2 black |
Reduction of Am(III) compounds with sodium amalgam yields Am(II) salts – the black halides AmCl2, AmBr2 and AmI2. They are very sensitive to oxygen and oxidize in water, releasing hydrogen and converting back to the Am(III) state. Specific lattice constants are:
- Orthorhombic AmCl2: a = 896.3±0.8 pm, b = 757.3±0.8 pm and c = 453.2±0.6 pm
- Tetragonal AmBr2: a = 1159.2±0.4 pm and c = 712.1±0.3 pm.[67] They can also be prepared by reacting metallic americium with an appropriate mercury halide HgX2, where X = Cl, Br or I:[68]
Americium(III) fluoride (AmF3) is poorly soluble and precipitates upon reaction of Am3+ and fluoride ions in weak acidic solutions:
The tetravalent americium(IV) fluoride (AmF4) is obtained by reacting solid americium(III) fluoride with molecular fluorine:[69][70]
Another known form of solid tetravalent americium fluoride is KAmF5.[69][71] Tetravalent americium has also been observed in the aqueous phase. For this purpose, black Am(OH)4 was dissolved in 15-M NH4F with the americium concentration of 0.01 M. The resulting reddish solution had a characteristic optical absorption spectrum which is similar to that of AmF4 but differed from other oxidation states of americium. Heating the Am(IV) solution to 90 °C did not result in its disproportionation or reduction, however a slow reduction was observed to Am(III) and assigned to self-irradiation of americium by alpha particles.[52]
Most americium(III) halides form hexagonal crystals with slight variation of the color and exact structure between the halogens. So, chloride (AmCl3) is reddish and has a structure isotypic to uranium(III) chloride (space group P63/m) and the melting point of 715 °C.[65] The fluoride is isotypic to LaF3 (space group P63/mmc) and the iodide to BiI3 (space group R3). The bromide is an exception with the orthorhombic PuBr3-type structure and space group Cmcm.[66] Crystals of americium(III) chloride hexahydrate (AmCl3·6H2O) can be prepared by dissolving americium dioxide in hydrochloric acid and evaporating the liquid. Those crystals are hygroscopic and have yellow-reddish color and a monoclinic crystal structure.[72]
Oxyhalides of americium in the form AmVIO2X2, AmVO2X, AmIVOX2 and AmIIIOX can be obtained by reacting the corresponding americium halide with oxygen or Sb2O3, and AmOCl can also be produced by vapor phase hydrolysis:[68]
- AmCl3 + H2O -> AmOCl + 2HCl
Chalcogenides and pnictides
The known chalcogenides of americium include the sulfide AmS2,[73] selenides AmSe2 and Am3Se4,[73][74] and tellurides Am2Te3 and AmTe2.[75] The pnictides of americium (243Am) of the AmX type are known for the elements phosphorus, arsenic,[76] antimony and bismuth. They crystallize in the rock-salt lattice.[74]
Silicides and borides
Americium monosilicide (AmSi) and "disilicide" (nominally AmSix with: 1.87 < x < 2.0) were obtained by reduction of americium(III) fluoride with elementary silicon in vacuum at 1050 °C (AmSi) and 1150−1200 °C (AmSix). AmSi is a black solid isomorphic with LaSi, it has an orthorhombic crystal symmetry. AmSix has a bright silvery lustre and a tetragonal crystal lattice (space group I41/amd), it is isomorphic with PuSi2 and ThSi2.[77] Borides of americium include AmB4 and AmB6. The tetraboride can be obtained by heating an oxide or halide of americium with magnesium diboride in vacuum or inert atmosphere.[78][79]
Organoamericium compounds

Analogous to uranocene, americium is predicted to form the organometallic compound amerocene with two cyclooctatetraene ligands, with the chemical formula (η8-C8H8)2Am.[80] A cyclopentadienyl complex is also known that is likely to be stoichiometrically AmCp3.[81][82]
Formation of the complexes of the type Am(n-C3H7-BTP)3, where BTP stands for 2,6-di(1,2,4-triazin-3-yl)pyridine, in solutions containing n-C3H7-BTP and Am3+ ions has been confirmed by EXAFS. Some of these BTP-type complexes selectively interact with americium and therefore are useful in its selective separation from lanthanides and another actinides.[83]
Biological aspects
Americium is an artificial element of recent origin, and thus does not have a biological requirement.[84][85] It is harmful to life. It has been proposed to use bacteria for removal of americium and other heavy metals from rivers and streams. Thus, Enterobacteriaceae of the genus Citrobacter precipitate americium ions from aqueous solutions, binding them into a metal-phosphate complex at their cell walls.[86] Several studies have been reported on the biosorption and bioaccumulation of americium by bacteria[87][88] and fungi.[89] In the laboratory, both americium and curium were found to support the growth of methylotrophs.[90]
Fission
The isotope 242mAm (half-life 141 years) has the largest cross sections for absorption of thermal neutrons (5,700 barns),[91] that results in a small critical mass for a sustained nuclear chain reaction. The critical mass for a bare 242mAm sphere is about 9–14 kg (the uncertainty results from insufficient knowledge of its material properties). It can be lowered to 3–5 kg with a metal reflector and should become even smaller with a water reflector.[92] Such small critical mass is favorable for portable nuclear weapons, but those based on 242mAm are not known yet, probably because of its scarcity and high price. The critical masses of the two readily available isotopes, 241Am and 243Am, are relatively high – 57.6 to 75.6 kg for 241Am and 209 kg for 243Am.[93] Scarcity and high price yet hinder application of americium as a nuclear fuel in nuclear reactors.[94]
There has been a proposal for very compact 10-kW high-flux reactors using as little as 20 grams of 242mAm. Such low-power reactors would be relatively safe to use as neutron sources for radiation therapy in hospitals.[95]
Isotopes
About 18 isotopes and 11 nuclear isomers are known for americium, having mass numbers 229, 230, and 232 through 247.[96] There are two long-lived alpha-emitters; 243Am has a half-life of 7,350 years and is the most stable isotope, and 241Am has a half-life of 432.6 years. The most stable nuclear isomer is 242m1Am - generally called simply 242mAm - with a long half-life of 141 years. The half-lives of other isotopes and isomers are much shorter with a maximum of 50.8 hours for 240Am. As with most other actinides, the isotopes of americium with odd number of neutrons have relatively high fissionability with thermal neutrons and low critical mass.
Americium-241 decays to 237Np emitting alpha particles of several different energies, mostly at 5.486 MeV (85.2%) and 5.443 MeV (12.8%). Becausethe resulting states are metastable, gamma rays are also emitted at discrete energies between 26.3 and 158.5 keV, by far the strongest[97] is at 59.5 keV.
Americium-242 is a short-lived isotope with a half-life of 16.02 h. It mostly (82.7%) converts by β-decay to 242Cm, but also by electron capture to 242Pu (17.3%). Though both will join the uranium decay chain, they do not do so on any practical timescale because of the life of 238U generated by the former but not the latter.
Nearly all (99.55%) of 242mAm decays by internal conversion to 242Am and the remaining 0.45% by α-decay to 238Np. The latter subsequently decays to 238Pu and then to 234U, as with the main branch of the ground state.
Americium-243 transforms by α-emission into 239Np, which converts by β-decay to 239Pu, and further decay follows the actinium series.
Applications
Ionization-type smoke detector
Americium is used in the most common type of household smoke detector, which uses 241Am in the form of americium dioxide as its source of ionizing radiation.[98] This isotope is preferred over 226Ra because it emits 5 times more alpha particles and relatively little harmful gamma radiation.
The amount of americium in a typical new smoke detector is 1 microcurie (37 kBq) or 0.29 microgram. This amount declines slowly as the americium decays into neptunium-237, a different transuranic element with a much longer half-life (about 2.14 million years). With its half-life of 432.2 years, the americium in a smoke detector includes about 3% neptunium after 19 years, and about 5% after 32 years. The radiation passes through an ionization chamber, an air-filled space between two electrodes, and permits a small, constant current between the electrodes. Any smoke that enters the chamber absorbs the alpha particles, which reduces the ionization and affects this current, triggering the alarm. Compared to the alternative optical smoke detector, the ionization smoke detector is cheaper and can detect particles which are too small to produce significant light scattering; however, it is more prone to false alarms.[99][100][101][102]
Radionuclide
As 241Am has a half-life roughly similar to 238Pu (432.2 years vs. 87 years), it has been proposed as an active element of radioisotope thermoelectric generators, for example in spacecraft.[103] Although americium produces less heat and electricity – the power yield is 114.7 mW/g for 241Am and 6.31 mW/g for 243Am[45] (cf. 390 mW/g for 238Pu)[103] – and its radiation poses more threat to humans owing to neutron emission, the European Space Agency is considering using americium for its space probes.[104]
Another proposed space-related application of americium is a fuel for space ships with nuclear propulsion. It relies on the very high rate of nuclear fission of 242mAm, which can be maintained even in a micrometer-thick foil. Small thickness avoids the problem of self-absorption of emitted radiation. This problem is pertinent to uranium or plutonium rods, in which only surface layers provide alpha-particles.[105][106] The fission products of 242mAm can either directly propel the spaceship or they can heat a thrusting gas. They can also transfer their energy to a fluid and generate electricity through a magnetohydrodynamic generator.[107]
One more proposal which utilizes the high nuclear fission rate of 242mAm is a nuclear battery. Its design relies not on the energy of the emitted by americium alpha particles, but on their charge, that is the americium acts as the self-sustaining "cathode". A single 3.2 kg 242mAm charge of such battery could provide about 140 kW of power over a period of 80 days.[108] Even with all the potential benefits, the current applications of 242mAm are as yet hindered by the scarcity and high price of this particular nuclear isomer.[107]
In 2019, researchers at the UK National Nuclear Laboratory and the University of Leicester demonstrated the use of heat generated by americium to illuminate a small light bulb. This technology could lead to systems to power missions with durations up to 400 years into interstellar space, where solar panels do not function.[109][110]
Neutron source
The oxide of 241Am pressed with beryllium is an efficient neutron source. Here americium acts as the alpha source, and beryllium produces neutrons owing to its large cross-section for the (α,n) nuclear reaction:
The most widespread use of 241AmBe neutron sources is a neutron probe – a device used to measure the quantity of water present in soil, as well as moisture/density for quality control in highway construction. 241Am neutron sources are also used in well logging applications, as well as in neutron radiography, tomography and other radiochemical investigations.[111]
Production of other elements
Americium is a starting material for the production of other transuranic elements and transactinides – for example, 82.7% of 242Am decays to 242Cm and 17.3% to 242Pu. In the nuclear reactor, 242Am is also up-converted by neutron capture to 243Am and 244Am, which transforms by β-decay to 244Cm:
Irradiation of 241Am by 12C or 22Ne ions yields the isotopes 247Es (einsteinium) or 260Db (dubnium), respectively.[111] Furthermore, the element berkelium (243Bk isotope) had been first intentionally produced and identified by bombarding 241Am with alpha particles, in 1949, by the same Berkeley group, using the same 60-inch cyclotron. Similarly, nobelium was produced at the Joint Institute for Nuclear Research, Dubna, Russia, in 1965 in several reactions, one of which included irradiation of 243Am with 15N ions. Besides, one of the synthesis reactions for lawrencium, discovered by scientists at Berkeley and Dubna, included bombardment of 243Am with 18O.[7]
Spectrometer
Americium-241 has been used as a portable source of both gamma rays and alpha particles for a number of medical and industrial uses. The 59.5409 keV gamma ray emissions from 241Am in such sources can be used for indirect analysis of materials in radiography and X-ray fluorescence spectroscopy, as well as for quality control in fixed nuclear density gauges and nuclear densometers. For example, the element has been employed to gauge glass thickness to help create flat glass.[24] Americium-241 is also suitable for calibration of gamma-ray spectrometers in the low-energy range, since its spectrum consists of nearly a single peak and negligible Compton continuum (at least three orders of magnitude lower intensity).[112] Americium-241 gamma rays were also used to provide passive diagnosis of thyroid function. This medical application is however obsolete.
Health concerns
As a highly radioactive element, americium and its compounds must be handled only in an appropriate laboratory under special arrangements. Although most americium isotopes predominantly emit alpha particles which can be blocked by thin layers of common materials, many of the daughter products emit gamma-rays and neutrons which have a long penetration depth.[113]
If consumed, most of the americium is excreted within a few days, with only 0.05% absorbed in the blood, of which roughly 45% goes to the liver and 45% to the bones, and the remaining 10% is excreted. The uptake to the liver depends on the individual and increases with age. In the bones, americium is first deposited over cortical and trabecular surfaces and slowly redistributes over the bone with time. The biological half-life of 241Am is 50 years in the bones and 20 years in the liver, whereas in the gonads (testicles and ovaries) it remains permanently; in all these organs, americium promotes formation of cancer cells as a result of its radioactivity.[19][114][115]
Americium often enters landfills from discarded smoke detectors. The rules associated with the disposal of smoke detectors are relaxed in most jurisdictions. In 1994, 17-year-old David Hahn extracted the americium from about 100 smoke detectors in an attempt to build a breeder nuclear reactor.[116][117][118][119] There have been a few cases of exposure to americium, the worst case being that of chemical operations technician Harold McCluskey, who at the age of 64 was exposed to 500 times the occupational standard for americium-241 as a result of an explosion in his lab. McCluskey died at the age of 75 of unrelated pre-existing disease.[120][121]
See also
Notes
- ↑ The "metastable" state is marked by the letter m.
References
- ↑ Seaborg, Glenn T. (1946). "The Transuranium Elements". Science 104 (2704): 379–386. doi:10.1126/science.104.2704.379. PMID 17842184. Bibcode: 1946Sci...104..379S. https://archive.org/details/sim_science_1946-10-25_104_2704/page/n1.
- ↑ Kostecka, Keith (2008). "Americium – From Discovery to the Smoke Detector and Beyond". Bull. Hist. Chem. 33 (2): 89–93. doi:10.70359/bhc2008v033p089. http://acshist.scs.illinois.edu/bulletin_open_access/v33-2/v33-2%20p89-93.pdf.
- ↑ "C&En: It's Elemental: The Periodic Table - Americium". http://pubsapp.acs.org/cen/80th/print/americiumprint.html?.
- ↑ Obituary of Dr. Leon Owen (Tom) Morgan (1919–2002), Retrieved 28 November 2010
- ↑ Seaborg, G. T.; James, R.A. and Morgan, L. O.: "The New Element Americium (Atomic Number 95)", THIN PPR (National Nuclear Energy Series, Plutonium Project Record), Vol 14 B The Transuranium Elements: Research Papers, Paper No. 22.1, McGraw-Hill Book Co., Inc., New York, 1949. Abstract; Full text (January 1948), Retrieved 28 November 2010
- ↑ Street, K.; Ghiorso, A.; Seaborg, G. (1950). "The Isotopes of Americium". Physical Review 79 (3): 530. doi:10.1103/PhysRev.79.530. Bibcode: 1950PhRv...79..530S. http://repositories.cdlib.org/cgi/viewcontent.cgi?article=7073&context=lbnl.
- ↑ 7.0 7.1 Greenwood, p. 1252
- ↑ "Americium (Am) | AMERICAN ELEMENTS ®" (in en). https://www.americanelements.com/am.html.
- ↑ 9.0 9.1 Pepling, Rachel Sheremeta (2003). "Chemical & Engineering News: It's Elemental: The Periodic Table – Americium". http://pubs.acs.org/cen/80th/americium.html.
- ↑ Robert E. Krebs (2006). The History and Use of Our Earth's Chemical Elements: A Reference Guide (Second ed.). Greenwood Publishing Group. p. 322. ISBN 978-0-313-33438-2. https://books.google.com/books?id=yb9xTj72vNAC&pg=PA322.
- ↑ 11.0 11.1 Audi, Georges; Bersillon, Olivier; Blachot, Jean; Wapstra, Aaldert Hendrik (1997). "The NUBASE evaluation of nuclear and decay properties". Nuclear Physics A 624 (1): 1–124. doi:10.1016/S0375-9474(97)00482-X. Bibcode: 1997NuPhA.624....1A. http://www.nndc.bnl.gov/amdc/nubase/Nubase2003.pdf.
- ↑ Seaborg, Glenn T. U.S. Patent 3,156,523 "Element", Filing date: 23 August 1946, Issue date: 10 November 1964
- ↑ 13.0 13.1 Westrum, Edgar F.; Eyring, Leroy (1951). "The Preparation and Some Properties of Americium Metal". Journal of the American Chemical Society 73 (7): 3396. doi:10.1021/ja01151a116. Bibcode: 1951JAChS..73.3396W.
- ↑ Earth, Rachel Ross 2017-05-23T02:31:00Z Planet (23 May 2017). "Facts About Americium" (in en). https://www.livescience.com/39874-facts-about-americium.html.
- ↑ "Americium - Element information, properties and uses | Periodic Table". http://www.rsc.org/periodic-table/element/95/americium.
- ↑ 16.0 16.1 Thornton, Brett F.; Burdette, Shawn C. (2019). "Neutron stardust and the elements of Earth". Nature Chemistry 11 (1): 4–10. doi:10.1038/s41557-018-0190-9. PMID 30552435. Bibcode: 2019NatCh..11....4T. https://www.nature.com/articles/s41557-018-0190-9. Retrieved 19 February 2022.
- ↑ Fields, P. R.; Studier, M. H.; Diamond, H. et al. (1956). "Transplutonium Elements in Thermonuclear Test Debris". Physical Review 102 (1): 180–182. doi:10.1103/PhysRev.102.180. Bibcode: 1956PhRv..102..180F. https://archive.org/details/sim_physical-review_1956-04-01_102_1/page/180.
- ↑ Eriksson, Mats (April 2002). On Weapons Plutonium in the Arctic Environment. Risø National Laboratory, Roskilde, Denmark: Lund University. p. 28. http://www.risoe.dk/rispubl/NUK/nukpdf/ris-r-1321.pdf. Retrieved 15 November 2008.
- ↑ 19.0 19.1 Human Health Fact Sheet on Americium , Los Alamos National Laboratory, Retrieved 28 November 2010
- ↑ Hoffmann, Klaus Kann man Gold machen? Gauner, Gaukler und Gelehrte. Aus der Geschichte der chemischen Elemente (Can you make gold? Crooks, clowns, and scholars. From the history of the chemical elements), Urania-Verlag, Leipzig, Jena, Berlin 1979, no ISBN, p. 233
- ↑ Baetslé, L. Application of Partitioning/Transmutation of Radioactive Materials in Radioactive Waste Management , Nuclear Research Centre of Belgium Sck/Cen, Mol, Belgium, September 2001, Retrieved 28 November 2010
- ↑ Fioni, Gabriele; Cribier, Michel and Marie, Frédéric Can the minor actinide, americium-241, be transmuted by thermal neutrons? , Department of Astrophysics, CEA/Saclay, Retrieved 28 November 2010
- ↑ Emsley, John (2011). Nature's Building Blocks: An A-Z Guide to the Elements (New ed.). New York, NY: Oxford University Press. ISBN 978-0-19-960563-7.
- ↑ 24.0 24.1 Greenwood, p. 1262
- ↑ 25.0 25.1 Smoke detectors and americium , World Nuclear Association, January 2009, Retrieved 28 November 2010
- ↑ Hammond C. R. "The elements" in Lide, D. R., ed (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ↑ Emeleus, H. J.; Sharpe, A. G. (1987). Advances in Inorganic Chemistry. Academic Press. p. 2. ISBN 978-0-08-057880-4. https://books.google.com/books?id=K5_LSQqeZ_IC&pg=PA2.
- ↑ BREDL Southern Anti-Plutonium Campaign, Blue Ridge Environmental Defense League, Retrieved 28 November 2010
- ↑ Sasahara, A. (2004). "Neutron and Gamma Ray Source Evaluation of LWR High Burn-up UO2 and MOX Spent Fuels". Journal of Nuclear Science and Technology 41 (4): 448–456. doi:10.3327/jnst.41.448. article/200410/000020041004A0333355.php Abstract
- ↑ Penneman, pp. 34–48
- ↑ Hudson, M. J. (2003). "The coordination chemistry of 1,2,4-triazinyl bipyridines with lanthanide(III) elements – implications for the partitioning of americium(III)". Dalton Trans. (9): 1675–1685. doi:10.1039/b301178j.
- ↑ Geist, A. (11–13 December 2000). "Actinide(III)/Lanthanide(III) Partitioning Using n-Pr-BTP as Extractant: Extraction Kinetics and Extraction Test in a Hollow Fiber Module". 6th Information Exchange Meeting on Actinide and Fission Product Partitioning and Transmutation. OECD Nuclear Energy Agency. https://www.oecd-nea.org/pt/docs/iem/madrid00/Proceedings/Paper14.pdf.
- ↑ Hill, C.; Guillaneux, D. (24–26 October 2000). "Sanex-BTP Process Development Studies". Atalante 2000: Scientific Research on the Back-end of the Fuel Cycle for the 21st Century. Commissariat à l'énergie atomique. http://www-atalante2004.cea.fr/home/liblocal/docs/atalante2000/P3-26.pdf.
- ↑ Geist, A. (14–16 October 2002). "Effective Actinide(III)-Lanthanide(III) Separation in Miniature Hollow Fibre Modules". 7th Information Exchange Meeting on Actinide and Fission Product Partitioning and Transmutation. OECD Nuclear Energy Agency. http://www.nea.fr/html/pt/docs/iem/jeju02/session2/SessionII-15.pdf.
- ↑ Ensor, D.D.. "Separation Studies of f-Elements". Tennessee Tech University. http://www.tntech.edu/WRC/pdfs/Projects04_05/Ens_Elem.pdf.
- ↑ Magnusson D; Christiansen B; Foreman MRS; Geist A; Glatz JP; Malmbeck R; Modolo G; Serrano-Purroy D et al. (2009). "Demonstration of a SANEX Process in Centrifugal Contactors using the CyMe4-BTBP Molecule on a Genuine Fuel Solution". Solvent Extraction and Ion Exchange 27 (2): 97. doi:10.1080/07366290802672204.
- ↑ Penneman, p. 25
- ↑ 38.0 38.1 38.2 Gmelin Handbook of Inorganic Chemistry, System No. 71, transuranics, Part B 1, pp. 57–67.
- ↑ 39.0 39.1 Penneman, p. 3
- ↑ 40.0 40.1 Wade, W.; Wolf, T. (1967). "Preparation and some properties of americium metal". Journal of Inorganic and Nuclear Chemistry 29 (10): 2577. doi:10.1016/0022-1902(67)80183-0.
- ↑ 41.0 41.1 Benedict, U. (1984). "Study of actinide metals and actinide compounds under high pressures". Journal of the Less Common Metals 100: 153. doi:10.1016/0022-5088(84)90061-4.
- ↑ 42.0 42.1 42.2 42.3 McWhan, D. B.; Cunningham, B. B.; Wallmann, J. C. (1962). "Crystal structure, thermal expansion and melting point of americium metal". Journal of Inorganic and Nuclear Chemistry 24 (9): 1025. doi:10.1016/0022-1902(62)80246-2.
- ↑ Young, D. A. (1991). Phase diagrams of the elements. University of California Press. p. 226. ISBN 978-0-520-91148-2. https://books.google.com/books?id=F2HVYh6wLBcC&pg=PA226.
- ↑ Benedict, U.; Dufour, C. (1980). "Low temperature lattice expansion of americium dioxide". Physica B+C 102 (1): 303. doi:10.1016/0378-4363(80)90178-3. Bibcode: 1980PhyBC.102..303B.
- ↑ 45.0 45.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedres - ↑ Kanellakopulos, B.; Blaise, A.; Fournier, J. M.; Müller, W. (1975). "The magnetic susceptibility of Americium and curium metal". Solid State Communications 17 (6): 713. doi:10.1016/0038-1098(75)90392-0. Bibcode: 1975SSCom..17..713K.
- ↑ Mondal, J. U.; Raschella, D. L.; Haire, R. G.; Petereson, J. R. (1987). "The enthalpy of solution of 243Am metal and the standard enthalpy of formation of Am3+(aq)". Thermochimica Acta 116: 235. doi:10.1016/0040-6031(87)88183-2.
- ↑ 48.0 48.1 48.2 Penneman, p. 4
- ↑ Americium , Das Periodensystem der Elemente für den Schulgebrauch (The periodic table of elements for schools) chemie-master.de (in German), Retrieved 28 November 2010
- ↑ Greenwood, p. 1265
- ↑ Penneman, pp. 10–14
- ↑ 52.0 52.1 Asprey, L. B.; Penneman, R. A. (1961). "First Observation of Aqueous Tetravalent Americium1". Journal of the American Chemical Society 83 (9): 2200. doi:10.1021/ja01470a040. Bibcode: 1961JAChS..83.2200A.
- ↑ 53.0 53.1 Coleman, J. S.; Keenan, T. K.; Jones, L. H.; Carnall, W. T.; Penneman, R. A. (1963). "Preparation and Properties of Americium(VI) in Aqueous Carbonate Solutions". Inorganic Chemistry 2: 58. doi:10.1021/ic50005a017.
- ↑ 54.0 54.1 Asprey, L. B.; Stephanou, S. E.; Penneman, R. A. (1951). "Hexavalent Americium". Journal of the American Chemical Society 73 (12): 5715. doi:10.1021/ja01156a065. Bibcode: 1951JAChS..73.5715A.
- ↑ Wiberg, p. 1956
- ↑ Werner, L. B.; Perlman, I. (1951). "The Pentavalent State of Americium". Journal of the American Chemical Society 73 (1): 495. doi:10.1021/ja01145a540. Bibcode: 1951JAChS..73..495W.
- ↑ Hall, G.; Markin, T. L. (1957). "The self-reduction of americium(V) and (VI) and the disproportionation of americium(V) in aqueous solution". Journal of Inorganic and Nuclear Chemistry 4 (5–6): 296. doi:10.1016/0022-1902(57)80011-6.
- ↑ Coleman, James S. (1963). "The Kinetics of the Disproportionation of Americium(V)". Inorganic Chemistry 2: 53. doi:10.1021/ic50005a016.
- ↑ Greenwood, p. 1275
- ↑ Asprey, L. B.; Stephanou, S. E.; Penneman, R. A. (1950). "A New Valence State of Americium, Am(Vi)1". Journal of the American Chemical Society 72 (3): 1425. doi:10.1021/ja01159a528. Bibcode: 1950JAChS..72.1425A. https://digital.library.unt.edu/ark:/67531/metadc1020623/.
- ↑ Akimoto, Y. (1967). "A note on AmN and AmO". Journal of Inorganic and Nuclear Chemistry 29 (10): 2650–2652. doi:10.1016/0022-1902(67)80191-X.
- ↑ Wiberg, p. 1972
- ↑ Greenwood, p. 1267
- ↑ Penneman, p. 5
- ↑ 65.0 65.1 Wiberg, p. 1969
- ↑ 66.0 66.1 Asprey, L. B.; Keenan, T. K.; Kruse, F. H. (1965). "Crystal Structures of the Trifluorides, Trichlorides, Tribromides, and Triiodides of Americium and Curium". Inorganic Chemistry 4 (7): 985. doi:10.1021/ic50029a013. https://digital.library.unt.edu/ark:/67531/metadc1035960/.
- ↑ Baybarz, R. D. (1973). "The preparation and crystal structures of americium dichloride and dibromide". Journal of Inorganic and Nuclear Chemistry 35 (2): 483. doi:10.1016/0022-1902(73)80560-3.
- ↑ 68.0 68.1 Greenwood, p. 1272
- ↑ 69.0 69.1 Asprey, L. B. (1954). "New Compounds of Quadrivalent Americium, AmF4, KAmF5". Journal of the American Chemical Society 76 (7): 2019. doi:10.1021/ja01636a094. Bibcode: 1954JAChS..76.2019A.
- ↑ Greenwood, p. 1271
- ↑ Penneman, p. 6
- ↑ Burns, John H.; Peterson, Joseph Richard (1971). "Crystal structures of americium trichloride hexahydrate and berkelium trichloride hexahydrate". Inorganic Chemistry 10: 147. doi:10.1021/ic50095a029.
- ↑ 73.0 73.1 Damien, D.; Jove, J. (1971). "Americium disulfide and diselenide". Inorganic and Nuclear Chemistry Letters 7 (7): 685. doi:10.1016/0020-1650(71)80055-7.
- ↑ 74.0 74.1 Roddy, J. (1974). "Americium metallides: AmAs, AmSb, AmBi, Am3Se4, and AmSe2". Journal of Inorganic and Nuclear Chemistry 36 (11): 2531. doi:10.1016/0022-1902(74)80466-5.
- ↑ Damien, D. (1972). "Americium tritelluride and ditelluride". Inorganic and Nuclear Chemistry Letters 8 (5): 501. doi:10.1016/0020-1650(72)80262-9.
- ↑ Charvillat, J.; Damien, D. (1973). "Americium monoarsenide". Inorganic and Nuclear Chemistry Letters 9 (5): 559. doi:10.1016/0020-1650(73)80191-6.
- ↑ Weigel, F.; Wittmann, F.; Marquart, R. (1977). "Americium monosilicide and "disilicide"". Journal of the Less Common Metals 56: 47. doi:10.1016/0022-5088(77)90217-X.
- ↑ Lupinetti, A. J. et al. U.S. Patent 6,830,738 "Low-temperature synthesis of actinide tetraborides by solid-state metathesis reactions", Filed 4 Apr 2002, Issued 14 December 2004
- ↑ Eick, Harry A.; Mulford, R. N. R. (1969). "Americium and neptunium borides". Journal of Inorganic and Nuclear Chemistry 31 (2): 371. doi:10.1016/0022-1902(69)80480-X.
- ↑ Elschenbroich, Christoph (2008). Organometallchemie. Vieweg+teubner Verlag. p. 589. ISBN 978-3-8351-0167-8.
- ↑ Albrecht-Schmitt, Thomas E. (2008). Organometallic and Coordination Chemistry of the Actinides. Springer. p. 8. ISBN 978-3-540-77836-3. https://books.google.com/books?id=rgmnVSzFzXMC&pg=PA8.
- ↑ Dutkiewicz, Michał S.; Apostolidis, Christos; Walter, Olaf; Arnold, Polly L. (30 January 2017). "Reduction chemistry of neptunium cyclopentadienide complexes: from structure to understanding". Chemical Science 2017 (8): 2553–61. doi:10.1039/C7SC00034K. PMID 28553487.
- ↑ Girnt, Denise; Roesky, Peter W.; Geist, Andreas; Ruff, Christian M.; Panak, Petra J.; Denecke, Melissa A. (2010). "6-(3,5-Dimethyl-1H-pyrazol-1-yl)-2,2'-bipyridine as Ligand for Actinide(III)/Lanthanide(III) Separation". Inorganic Chemistry 49 (20): 9627–35. doi:10.1021/ic101309j. PMID 20849125. https://www.escholar.manchester.ac.uk/api/datastream?publicationPid=uk-ac-man-scw:209191&datastreamId=POST-PEER-REVIEW-PUBLISHERS.PDF. Retrieved 24 August 2019.
- ↑ Toeniskoetter, Steve; Dommer, Jennifer and Dodge, Tony The Biochemical Periodic Tables – Americium, University of Minnesota, Retrieved 28 November 2010
- ↑ Dodge, C.J. (1998). "Role of Microbes as Biocolloids in the Transport of Actinides from a Deep Underground Radioactive Waste Repository". Radiochim. Acta 82: 347–354. doi:10.1524/ract.1998.82.special-issue.347. Bibcode: 1998RadAc..82..347F. http://www.osti.gov/bridge/product.biblio.jsp?osti_id=2439.
- ↑ MacAskie, L. E.; Jeong, B. C.; Tolley, M. R. (1994). "Enzymically accelerated biomineralization of heavy metals: application to the removal of americium and plutonium from aqueous flows". FEMS Microbiology Reviews 14 (4): 351–67. doi:10.1111/j.1574-6976.1994.tb00109.x. PMID 7917422. Bibcode: 1994FEMMR..14..351M.
- ↑ Wurtz, E. A.; Sibley, T. H.; Schell, W. R. (1986). "Interactions of Escherichia coli and marine bacteria with 241Am in laboratory cultures". Health Physics 50 (1): 79–88. doi:10.1097/00004032-198601000-00007. PMID 3511007. Bibcode: 1986HeaPh..50...79W.
- ↑ Francis, A.J. (1998). "Role of Bacteria as Biocolloids in the Transport of Actinides from a Deep Underground Radioactive Waste Repository". Radiochimica Acta 82: 347–354. doi:10.1524/ract.1998.82.special-issue.347. Bibcode: 1998RadAc..82..347F.
- ↑ Liu, N.; Yang, Y.; Luo, S.; Zhang, T.; Jin, J.; Liao, J.; Hua, X. (2002). "Biosorption of 241Am by Rhizopus arrihizus: preliminary investigation and evaluation". Applied Radiation and Isotopes 57 (2): 139–43. doi:10.1016/s0969-8043(02)00076-3. PMID 12150270.
- ↑ Remick, Kaleigh; Helmann, John D. (30 January 2023). "The Elements of Life: A Biocentric Tour of the Periodic Table". Advances in Microbial Physiology (PubMed Central) 82: 1–127. doi:10.1016/bs.ampbs.2022.11.001. ISBN 978-0-443-19334-7. PMID 36948652.
- ↑ Pfennig, G.; Klewe-Nebenius, H and Seelmann Eggebert, W. (Eds.): Karlsruhe nuclide, 7 Edition 2006.
- ↑ Dias, H.; Tancock, N.; Clayton, A. (2003). "Critical Mass Calculations for 241Am, 242mAm and 243Am". Nippon Genshiryoku Kenkyujo JAERI: 618–623. http://typhoon.jaea.go.jp/icnc2003/Proceeding/paper/6.5_022.pdf. Abstract
- ↑ Institut de Radioprotection et de Sûreté Nucléaire, "Evaluation of nuclear criticality safety data and limits for actinides in transport", p. 16.
- ↑ Ronen, Y.; Aboudy, M.; Regev, D. (2000). "A novel method for energy production using 242mAm as a nuclear fuel". Nuclear Technology 129 (3): 407–417. doi:10.13182/nt00-a3071. Bibcode: 2000NucTe.129..407R. http://cat.inist.fr/?aModele=afficheN&cpsidt=1337515.
- ↑ Ronen, Y.; Aboudy, M.; Regev, D. (2001). "Homogeneous 242mAm-Fueled Reactor for Neutron Capture Therapy". Nuclear Science and Engineering 138 (3): 295–304. doi:10.13182/nse01-a2215. Bibcode: 2001NSE...138..295R.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedNUBASE2020 - ↑ National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory. http://www.nndc.bnl.gov/nudat2/.
- ↑ "Smoke Detectors and Americium", Nuclear Issues Briefing Paper 35, May 2002, http://www.uic.com.au/nip35.htm, retrieved 2015-08-26
- ↑ Residential Smoke Alarm Performance, Thomas Cleary. Building and Fire Research Laboratory, National Institute of Standards and Technology; UL Smoke and Fire Dynamics Seminar. November 2007
- ↑ Bukowski, R. W. et al. (2007) Performance of Home Smoke Alarms Analysis of the Response of Several Available Technologies in Residential Fire Settings , NIST Technical Note 1455-1
- ↑ "Smoke detectors and americium-241 fact sheet". Canadian Nuclear Society. http://media.cns-snc.ca/pdf_doc/ecc/smoke_am241.pdf.
- ↑ Gerberding, Julie Louise (2004). "Toxicological Profile For Americium". United States Department of Health and Human Services/Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/toxprofiles/tp156.pdf.
- ↑ 103.0 103.1 Basic elements of static RTGs , G.L. Kulcinski, NEEP 602 Course Notes (Spring 2000), Nuclear Power in Space, University of Wisconsin Fusion Technology Institute (see last page)
- ↑ Space agencies tackle waning plutonium stockpiles, Spaceflight now, 9 July 2010
- ↑ "Extremely Efficient Nuclear Fuel Could Take Man To Mars in Just Two Weeks". 3 January 2001. https://www.sciencedaily.com/releases/2001/01/010103073253.htm.
- ↑ Kammash, T. (10 January 1993). "An americium-fueled gas core nuclear rocket". Tenth symposium on space nuclear power and propulsion. 271. pp. 585–589. doi:10.1063/1.43073. https://deepblue.lib.umich.edu/bitstream/2027.42/87734/2/585_1.pdf.
- ↑ 107.0 107.1 Ronen, Y.; Shwageraus, E. (2000). "Ultra-thin 242mAm fuel elements in nuclear reactors". Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment 455 (2): 442. doi:10.1016/S0168-9002(00)00506-4. Bibcode: 2000NIMPA.455..442R.
- ↑ Genuth, Iddo Americium Power Source , The Future of Things, 3 October 2006, Retrieved 28 November 2010
- ↑ "UK scientists generate electricity from rare element to power future space missions". 3 May 2019. https://www.nnl.co.uk/2019/05/uk-scientists-generate-electricity-from-rare-element-to-power-future-space-missions/.
- ↑ "Rare element could power distant space missions". E&T Engineering and Technology (Institution of Engineering and Technology). 3 May 2019. https://eandt.theiet.org/content/articles/2019/05/rare-element-could-power-far-flung-space-missions. Retrieved 3 May 2019.
- ↑ 111.0 111.1 Binder, Harry H. (1999). Lexikon der chemischen Elemente: das Periodensystem in Fakten, Zahlen und Daten : mit 96 Abbildungen und vielen tabellarischen Zusammenstellungen. Hirzel. ISBN 978-3-7776-0736-8.
- ↑ Nuclear Data Viewer 2.4 , NNDC
- ↑ Public Health Statement for Americium Section 1.5., Agency for Toxic Substances and Disease Registry, April 2004, Retrieved 28 November 2010
- ↑ Division of Environmental Health, Office of Radiation Protection (November 2002). "Fact Sheet # 23. Americium-241". http://www.doh.wa.gov/ehp/rp/factsheets/factsheets-pdf/fs23am241.pdf.
- ↑ Frisch, Franz Crystal Clear, 100 x energy, Bibliographisches Institut AG, Mannheim 1977, ISBN 3-411-01704-X, p. 184
- ↑ Ken Silverstein, The Radioactive Boy Scout: When a teenager attempts to build a breeder reactor. Harper's Magazine, November 1998
- ↑ "'Radioactive Boy Scout' Charged in Smoke Detector Theft". Fox News. 4 August 2007. http://www.foxnews.com/story/0,2933,292111,00.html.
- ↑ "Man dubbed 'Radioactive Boy Scout' pleads guilty". Detroit Free Press. Associated Press. 27 August 2007. http://www.freep.com/apps/pbcs.dll/article?AID=/20070827/BUSINESS05/70827091.
- ↑ "'Radioactive Boy Scout' Sentenced to 90 Days for Stealing Smoke Detectors". Fox News. 4 October 2007. https://www.foxnews.com/story/radioactive-boy-scout-sentenced-to-90-days-for-stealing-smoke-detectors.
- ↑ Cary, Annette (25 April 2008). "Doctor remembers Hanford's 'Atomic Man'". Tri-City Herald. http://www.hanfordnews.com/news/2008/story/11403.html.
- ↑ AP wire (3 June 2005). "Hanford nuclear workers enter site of worst contamination accident". http://www.billingsgazette.com/index.php?id=1&display=rednews/2005/06/03/build/nation/94-contamination.inc.
Bibliography
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Penneman, R. A. and Keenan T. K. The radiochemistry of americium and curium, University of California, Los Alamos, California, 1960
- Wiberg, Nils (2007). Lehrbuch Der Anorganischen Chemie. De Gruyter. ISBN 978-3-11-017770-1.
Further reading
- Nuclides and Isotopes – 14th Edition, GE Nuclear Energy, 1989.
- Fioni, Gabriele; Cribier, Michel. "Can the minor actinide, americium-241, be transmuted by thermal neutrons?". Commissariat à l'énergie atomique. http://www.cea.fr/var/cea/storage/static/gb/library/Clefs46/pagesg/clefs46_30.html.
- Stwertka, Albert (1999). A Guide to the Elements. Oxford University Press, USA. ISBN 978-0-19-508083-4. https://archive.org/details/guidetoelements00stwe.
External links
| Wikimedia Commons has media related to Americium. |
- Americium at The Periodic Table of Videos (University of Nottingham)
- ATSDR – Public Health Statement: Americium
- World Nuclear Association – Smoke Detectors and Americium
 |


