Physics:Nuclear isomer
| Nuclear physics |
|---|
 |
| Nucleus · Nucleons (p, n) · Nuclear matter · Nuclear force · Nuclear structure · Nuclear reaction |
A nuclear isomer is a metastable state of an atomic nucleus in which one or more nucleons (protons or neutrons) occupy excited state levels (higher energy levels). "Metastable" describes nuclei whose excited states have half-lives of 10−9 seconds or longer,[1] 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half-life (ordinarily on the order of 10−12 seconds). Some references recommend using a threshold of 5×10−9 seconds to distinguish the metastable half-life from the normal "prompt" gamma-emission half-life.[2]
The half-lives of a number of isomers are far longer than this and may be minutes, hours, or years. An extreme example is the 180m73Ta nuclear isomer, which survives so long (at least 2.9×1017 years[3]) that it has never been observed to decay spontaneously, and occurs naturally as a primordial nuclide, though uncommonly at only 1/8000 of all tantalum. The second most stable isomer is 210m83Bi, which does not occur naturally; its half-life is 3.04×106 years to alpha decay. The half-life of a nuclear isomer can exceed that of the ground state of the same nuclide, as with the two above, as well as, for example, 186m75Re, 192m277Ir, 212m84Po, 242m95Am and multiple holmium isomers.
The gamma decay from a metastable state is referred to as isomeric transition (IT), or internal transition, though it resembles shorter-lived "prompt" gamma decays in all external aspects with the exception of the longer life. This is generally associated with a high nuclear spin change, or "forbiddenness", which would be required in gamma emission to reach the ground state; this is even more true of beta decays. A low transition energy both slows the transition rate and makes it more likely that only highly forbidden decays are available, so most long-lived isomers have a relatively low excitation energy above the ground state (in the extreme case of thorium-229m, low excitation alone causes the measurably long life). In 210m83Bi, the forbiddenness of available beta and gamma decays is so high that alpha decay is observed exclusively, though even that is slower than for the ground state. For most lighter isomers including 180m73Ta, alpha decay is not practically available, but others are not quite so forbidden as those two.
The first nuclear isomer and decay-daughter system (uranium X2/uranium Z, now known as 234m91Pa/23491Pa) was discovered by Otto Hahn in 1921.[4]
Metastable isomers can be produced through any nuclear reaction, including radioactive decay, neutron capture, nuclear fission, and bombardment by accelerated charged particles. A nucleus produced this way generally starts its existence in an excited state that loses its excess energy through the emission of one or more gamma rays or conversion electrons. This is normally a "prompt" process, but sometimes does not rapidly reach to the nuclear ground state, in which case a metastable isomer has formed. This usually occurs as a spin isomer when the formation of an intermediate excited state has a spin far different from that of the ground state. Gamma-ray emission is hindered if the spin of the post-emission state differs greatly from that of the emitting state, and if the excitation energy is low; such excited states will generally have long lives and be considered metastable.
After fission, several of the fission fragments may be produced in a metastable isomeric state, after their prompt de-excitation. At the end of this process, the nuclei can populate both the ground and the isomeric states. If the half-life of an isomer is long enough, it is possible to measure its production rate, and comparing it to that of the ground state gives the so-called isomeric yield ratio.[5]
A particular kind of metastable isomer is the fission isomer or shape isomer. Most actinide nuclei in their ground states are not spherical, but rather prolate spheroidal, with an axis of symmetry longer than the other axes, similar to an American football or rugby ball. This geometry can result in quantum-mechanical states where the distribution of protons and neutrons is so much further from spherical geometry that de-excitation to the nuclear ground state is strongly hindered. In general, these states either de-excite to the ground state far more slowly than a "usual" excited state, or they undergo spontaneous fission with half-lives of the order of nanoseconds or microseconds—a very short time, but many orders of magnitude longer than the half-life of a more usual nuclear excited state. Fission isomers may be denoted with a postscript or superscript "f" rather than "m", so that a fission isomer, e.g. of plutonium-240, can be denoted as plutonium-240f or 240f94Pu.
Nomenclature
Metastable isomers of a particular isotope are usually designated with an "m". This designation is placed after the mass number of the atom; for example, cobalt-58m1 is abbreviated 58m127Co, where 27 is the atomic number of cobalt. For isotopes with more than one metastable isomer, "indices" are placed after the designation, and the labeling becomes m1, m2, m3, and so on. Increasing indices, m1, m2, etc., correlate with increasing levels of excitation energy stored in each of the isomeric states (e.g., hafnium-178m2, or 178m272Hf). The index may be omitted if only one isomer is relevant.
Nuclei of nuclear isomers
The nucleus of a nuclear isomer occupies a higher energy state than the non-excited nucleus existing in the ground state. In an excited state, one or more of the protons or neutrons in a nucleus occupy a nuclear orbital of higher energy than an available nuclear orbital. These states are analogous to excited states of electrons in atoms.
When excited atomic states decay, energy is released by fluorescence. In electronic transitions, this process usually involves emission of light near the visible range. The amount of energy released is related to bond-dissociation energy or ionization energy and is usually in the range of a few to few tens of eV per bond. However, a much stronger type of binding energy, the nuclear binding energy, is involved in nuclear processes. Due to this, most nuclear excited states decay by gamma ray emission. For example, a well-known nuclear isomer used in various medical procedures is 99m43Tc, which decays with a half-life of about 6 hours by emitting a gamma ray of 140.5 keV energy; this is similar to the energy of medical diagnostic X-rays.
Nuclear isomers have long half-lives because their decay to the ground state is highly "forbidden" from the large change in nuclear spin required. For example, 180m73Ta has a spin of 9 and the lower states have spins 1 and 2. Similarly, 99m43Tc has a spin of 1/2 and the lower states 7/2 and 9/2.[6] Clearly, the latter is less "forbidden" and, as expected, much faster.
Nuclear transitions, including the 'isomeric' variety, occur not only through gamma-ray emission, but also internal conversion where the transition energy instead ejects an electron from the atom. The two processes always compete, with gamma emission normally the most common, but as the proportion converted increases with lower energy and also with forbiddenness, it often becomes important for metastable isomers. In fact, the usual decay of 99m43Tc involves conversion to the spin-7/2 state, then prompt gamma emission to the spin-9/2 ground state; similarly, 180m73Ta could decay through conversion to the spin-2 state, followed by a gamma decay to the ground state. This gamma was looked for in,[3] which assumed that to be the likely decay scheme, and not found.
In isotopes whose ground state is unstable, isomers can decay by the same modes rather than going to the ground state. Often both are seen, but rates can differ so much that only one is. Both isomers discussed just above have unstable ground states: 9943Tc undergoes beta decay, though slowly (half-life 211 ky) due to forbiddenness, and the isomer, which is less so, beta-decays over 10,000 times faster (though still a small minority of decays); 18073Ta can fall to either beta decay or electron capture, and quickly (half-life 8.15 h) as it is not forbidden, there the isomer is much more so to either as well as to isomeric transition, explaining its stability.
Artificial de-excitation
It was first reported in 1988 by C. B. Collins[7] that theoretically The element Chemistry:Ta does not exist. can be forced to release its energy by weaker X-rays, although at that time this de-excitation mechanism had never been observed. However, the de-excitation of The element Chemistry:Ta does not exist. by resonant photo-excitation of intermediate high levels of this nucleus (E ≈ 1 MeV) was observed in 1999 by Belic and co-workers in the Stuttgart nuclear physics group.[8]
178m272Hf is another reasonably stable nuclear isomer, with a half-life of 31 years and a remarkably high excitation energy for that life. In the natural decay of The element Chemistry:Hf does not exist., the energy is released as gamma rays with a total energy of 2.45 MeV. As with The element Chemistry:Ta does not exist., it is thought that The element Chemistry:Hf does not exist. can be stimulated into releasing its energy. Due to this, the substance has been studied as a possible source for gamma-ray lasers, and reports have indicated that the energy could be released very quickly, so that The element Chemistry:Hf does not exist. can produce extremely high powers (on the order of exawatts).
Other isomers have also been investigated as possible media for gamma-ray stimulated emission.[2][9]
Other notable isomers
Holmium's nuclear isomer 166m167Ho has a half-life of 1,133 years, which is nearly the longest half-life of any holmium radionuclide. Only The element Chemistry:Holmium does not exist., with a half-life of 4,570 years, is more stable. Both the excitation energy of the former, and the decay energy of the latter, are less than 10 keV.
229m90Th is a remarkably low-lying metastable isomer only 8.355733554021(8) eV above the ground state.[10][11][12] This low energy produces "gamma rays" at a wavelength of 148.3821828827(15) nm, in the far ultraviolet, which allows for direct nuclear laser spectroscopy. Such ultra-precise spectroscopy, however, could not begin without a sufficiently precise initial estimate of the wavelength, something that was only achieved in 2024 after two decades (from 2003 to 2024) of effort.[13][14][15][16][17][11] The energy is so low that the ionization state of the atom affects its half-life. Neutral 229m90Th decays by internal conversion with a half-life of 7±1 μs, but because the isomeric energy is less than thorium's second ionization energy of 11.5 eV, this channel is forbidden in thorium cations and 229m90Th+ decays by gamma emission with a half-life of 1740±50 s.[10] This conveniently moderate lifetime allows the development of a nuclear clock of unprecedented accuracy.[18][19][12]
Mechanism of suppression of decay
The most common mechanism for suppression of gamma decay of excited nuclei, and thus the existence of a metastable isomer, is lack of a decay route for the excited state that will change nuclear angular momentum in any given step by 0 or 1 quantum unit (ħ) of spin angular momentum. This change is necessary to emit a gamma photon in an (electric dipole) allowed transition, as the photon has a spin of 1 unit. Changes of 2 or more units (any possible change is always integer) in angular momentum are possible, but the emitted photon must carry off the additional angular momentum. Changes of more than 1 unit are known as forbidden transitions. Each additional unit of spin larger than 1 that the emitted gamma ray must carry inhibits decay rate by about 5 orders of magnitude,[20] but this again increases at lower energies, and finally IC takes over, as can be seen in figures 14.61 and 14.62 of 'Quantum Mechanics for Engineers' by Leon van Dommelen.[20] From that it should be seen that the spin change of no less than 7 units that would occur in the hypothetical gamma decay of 180mTa should result in essentially total suppression and replacement by IC, in agreement with the above.
Gamma emission is impossible when the nucleus begins and ends in a zero-spin state, as such an emission would not conserve angular momentum. Internal conversion remains possible for such transitions.[20]
Applications
Hafnium[21][22] isomers (mainly 178m2Hf) have been considered as weapons that could be used to circumvent the Nuclear Non-Proliferation Treaty, since it is claimed that they can be induced to emit very strong gamma radiation. This claim is generally discounted.[23] DARPA had a program to investigate this use of both nuclear isomers.[24] The potential to trigger an abrupt release of energy from nuclear isotopes, a prerequisite to their use in such weapons, is disputed. Nonetheless a 12-member Hafnium Isomer Production Panel (HIPP) was created in 2003 to assess means of mass-producing the isotope.[25]
Technetium isomers 99m43Tc (with a half-life of 6.01 hours) and 95m43Tc (with a half-life of 61 days) are used in medical and industrial applications.
Nuclear batteries
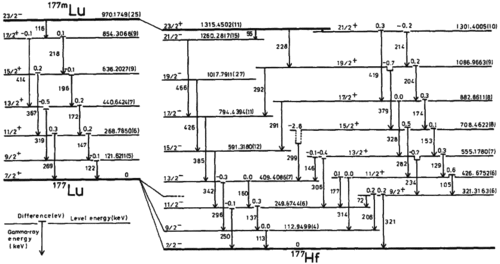
Nuclear batteries use small amounts (milligrams and microcuries) of radioisotopes with high energy densities. In one betavoltaic device design, radioactive material sits atop a device with adjacent layers of P-type and N-type silicon. Ionizing radiation directly penetrates the junction and creates electron–hole pairs. Nuclear isomers could replace other isotopes, and with further development, it may be possible to turn them on and off by triggering decay as needed. Current candidates for such use include 108Ag, 166Ho, 177Lu, and 242Am. As of 2004, the only successfully triggered isomer was 180mTa, which required more photon energy to trigger than was released.[26]
An isotope such as 177Lu releases gamma rays by decay through a series of internal energy levels within the nucleus, and it is thought that by learning the triggering cross sections with sufficient accuracy, it may be possible to create energy stores that are 106 times more concentrated than high explosive or other traditional chemical energy storage.[26]
See also
- Induced gamma emission
- Isomeric shift
- Mössbauer effect
References
- ↑ The standard reference Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties". Chinese Physics C 45 (3). doi:10.1088/1674-1137/abddae. https://www-nds.iaea.org/amdc/ame2020/NUBASE2020.pdf. uses approximately 10−7 seconds
- ↑ 2.0 2.1 Walker, Philip M.; Carroll, James J. (2007). "Nuclear Isomers: Recipes from the Past and Ingredients for the Future". Nuclear Physics News 17 (2): 11–15. doi:10.1080/10506890701404206. http://epubs.surrey.ac.uk/137/1/fulltext.pdf.
- ↑ 3.0 3.1 Arnquist, I. J.; Avignone III, F. T.; Barabash, A. S.; Barton, C. J.; Bhimani, K. H.; Blalock, E.; Bos, B.; Busch, M. et al. (13 October 2023). "Constraints on the Decay of 180mTa". Phys. Rev. Lett. 131 (15). doi:10.1103/PhysRevLett.131.152501. PMID 37897780.
- ↑ Hahn, Otto (1921). "Über ein neues radioaktives Zerfallsprodukt im Uran". Die Naturwissenschaften 9 (5): 84. doi:10.1007/BF01491321. Bibcode: 1921NW......9...84H. https://zenodo.org/record/2482506.
- ↑ Rakopoulos, V.; Lantz, M.; Solders, A.; Al-Adili, A.; Mattera, A.; Canete, L.; Eronen, T.; Gorelov, D. et al. (2018-08-13). "First isomeric yield ratio measurements by direct ion counting and implications for the angular momentum of the primary fission fragments" (in en). Physical Review C 98 (2). doi:10.1103/PhysRevC.98.024612. ISSN 2469-9985. Bibcode: 2018PhRvC..98b4612R. https://link.aps.org/doi/10.1103/PhysRevC.98.024612.
- ↑ ENSDF data as compiled at National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory. http://www.nndc.bnl.gov/nudat2/.
- ↑ C. B. Collins (1988). "Depopulation of the isomeric state 180Tam by the reaction 180Tam(γ,γ′)180Ta". Physical Review C 37 (5): 2267–2269. doi:10.1103/PhysRevC.37.2267. PMID 9954706. Bibcode: 1988PhRvC..37.2267C. http://www.hafniumisomer.org/isomer/180ta.pdf.
- ↑ D. Belic (1999). "Photoactivation of 180Tam and Its Implications for the Nucleosynthesis of Nature's Rarest Naturally Occurring Isotope". Physical Review Letters 83 (25): 5242–5245. doi:10.1103/PhysRevLett.83.5242. Bibcode: 1999PhRvL..83.5242B.
- ↑ "UNH researchers search for stimulated gamma ray emission". 1997. http://einstein.unh.edu/nuclear/NucNews/graser_news.html.
- ↑ 10.0 10.1 Tiedau, J.; Okhapkin, M. V.; Zhang, K.; Thielking, J.; Zitzer, G.; Peik, E.; Schaden, F.; Pronebner, T. et al. (2024-04-29). "Laser Excitation of the Th-229 Nucleus". Physical Review Letters 132 (18). doi:10.1103/PhysRevLett.132.182501. PMID 38759160. Bibcode: 2024PhRvL.132r2501T.
- ↑ 11.0 11.1 Zhang, Chuankun; Ooi, Tian; Higgins, Jacob S.; Doyle, Jack F.; von der Wense, Lars; Beeks, Kjeld; Leitner, Adrian; Kazakov, Georgy et al. (4 September 2024). "Frequency ratio of the 229mTh nuclear isomeric transition and the 87Sr atomic clock". Nature 633 (8028): 63–70. doi:10.1038/s41586-024-07839-6. PMID 39232152. Bibcode: 2024Natur.633...63Z. "The transition frequency between the I = 5/2 ground state and the I = 3/2 excited state is determined as: 𝜈Th = 1/6 (𝜈a + 2𝜈b + 2𝜈c + 𝜈d) = 2020407384335(2) kHz.".
- ↑ 12.0 12.1 "A nuclear clock prototype hints at ultraprecise timekeeping". ScienceNews. 4 September 2024. https://www.sciencenews.org/article/nuclear-clock-ultraprecise-timekeeping.
- ↑ von der Wense, Lars; Seiferle, Benedict; Laatiaoui, Mustapha; Neumayr, Jürgen B.; Maier, Hans-Jörg; Wirth, Hans-Friedrich; Mokry, Christoph; Runke, Jörg et al. (2016-05-05). "Direct detection of the 229Th nuclear clock transition". Nature 533 (7601): 47–51. doi:10.1038/nature17669. PMID 27147026. Bibcode: 2016Natur.533...47V. http://www.2physics.com/2016/06/direct-detection-of-229-th-nuclear.html.
- ↑ "Results on 229mThorium published in "Nature"" (Press release). LMU Munich. 2016-05-06. Archived from the original on 27 August 2016. Retrieved 1 August 2016.
- ↑ Seiferle, B.; von der Wense, L.; Thirolf, P.G. (2017-01-26). "Lifetime measurement of the 229Th nuclear isomer". Phys. Rev. Lett. 118 (4). doi:10.1103/PhysRevLett.118.042501. PMID 28186791. Bibcode: 2017PhRvL.118d2501S.
- ↑ Thielking, J.; Okhapkin, M.V.; Przemyslaw, G.; Meier, D.M.; von der Wense, L.; Seiferle, B.; Düllmann, C.E.; Thirolf, P.G. et al. (2018). "Laser spectroscopic characterization of the nuclear-clock isomer 229mTh". Nature 556 (7701): 321–325. doi:10.1038/s41586-018-0011-8. PMID 29670266. Bibcode: 2018Natur.556..321T.
- ↑ Seiferle, B.; von der Wense, L.; Bilous, P.V.; Amersdorffer, I.; Lemell, C.; Libisch, F.; Stellmer, S.; Schumm, T. et al. (2019-09-12). "Energy of the 229Th nuclear clock transition". Nature 573 (7773): 243–246. doi:10.1038/s41586-019-1533-4. PMID 31511684. Bibcode: 2019Natur.573..243S.
- ↑ Peik, Ekkehard; Tamm, Christian (2003-01-15). "Nuclear laser spectroscopy of the 3.5 eV transition in 229Th". Europhysics Letters 61 (2): 181–186. doi:10.1209/epl/i2003-00210-x. Bibcode: 2003EL.....61..181P. http://www.ptb.de/cms/fileadmin/internet/fachabteilungen/abteilung_4/4.4_zeit_und_frequenz/pdf/th001.pdf. Retrieved 12 September 2019.
- ↑ Campbell, C.; Radnaev, A.G.; Kuzmich, A.; Dzuba, V.A.; Flambaum, V.V.; Derevianko, A. (22 March 2012). "A single ion nuclear clock for metrology at the 19th decimal place". Phys. Rev. Lett. 108 (12). doi:10.1103/PhysRevLett.108.120802. PMID 22540568. Bibcode: 2012PhRvL.108l0802C. https://link.aps.org/accepted/10.1103/PhysRevLett.108.120802.
- ↑ 20.0 20.1 20.2 Leon van Dommelen, Quantum Mechanics for Engineers, Section 14.20
- ↑ David Hambling (16 August 2003). "Gamma-ray weapons". New Scientist. http://www.eurekalert.org/pub_releases/2003-08/ns-gw081303.php.
- ↑ Jeff Hecht (19 June 2006). "A perverse military strategy". New Scientist. https://www.newscientist.com/article/mg19025562.200-a-perverse-military-strategy.html. Retrieved 12 December 2010.
- ↑ Davidson, Seay. "Superbomb Ignites Science Dispute". http://www.veteransforpeace.org/Superbomb_ignites_092903.htm.
- ↑ S. Weinberger (28 March 2004). "Scary things come in small packages". Washington Post. https://www.washingtonpost.com/ac2/wp-dyn?pagename=article&contentId=A22099-2004Mar24¬Found=true.
- ↑ "Superbomb ignites science dispute". San Francisco Chronicle. 2003-09-28. http://www.commondreams.org/headlines03/0928-07.htm.
- ↑ 26.0 26.1 "Controlled extraction of energy from nuclear isomers". December 2004. http://apps.dtic.mil/dtic/tr/fulltext/u2/a433348.pdf.
External links
- Research group which presented initial claims of hafnium nuclear isomer de-excitation control. – The Center for Quantum Electronics, The University of Texas at Dallas.
- JASON Defense Advisory Group report on high energy nuclear materials mentioned in the Washington Post story above
- Bertram Schwarzschild (May 2004). "Conflicting Results on a Long-Lived Nuclear Isomer of Hafnium Have Wider Implications". Physics Today 57 (5): 21–24. doi:10.1063/1.1768663. Bibcode: 2004PhT....57e..21S. http://scitation.aip.org/journals/doc/PHTOAD-ft/vol_57/iss_5/21_1.shtml.
- Confidence for Hafnium Isomer Triggering in 2006. – The Center for Quantum Electronics, The University of Texas at Dallas.
- Reprints of articles about nuclear isomers in peer reviewed journals. – The Center for Quantum Electronics, The University of Texas at Dallas.
 |
