Chemistry:Monomagnesium phosphate
From HandWiki
Revision as of 18:04, 16 July 2022 by imported>TextAI2 (update)
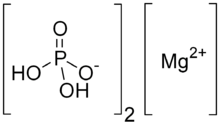
| |
| Names | |
|---|---|
| Other names
Monomagnesium orthophosphate;
Magnesium dihydrogen phosphate; Magnesium phosphate monobasic; Magnesium biphosphate; Acid magnesium phosphate | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| H4MgO8P2 | |
| Molar mass | 218.277 g·mol−1 |
| Appearance | White, odorless, crystalline powder |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Monomagnesium phosphate is one of the forms of magnesium phosphate. It is a magnesium acid salt of phosphoric acid with the chemical formula Mg(H2PO4)2. Di- and tetrahydrates are known also. It dissolves in water, forming phosphoric acid and depositing a solid precipitate of Mg(HPO4).3H2O, dimagnesium phosphate.[2]
As a food additive, it is used as an acidity regulator and has the E number E343.
References
- ↑ Monomagnesium phosphate, FAO JECFA Monographs 5 (2008)
- ↑ Klaus Schrödter; Gerhard Bettermann; Thomas Staffel; Friedrich Wahl; Thomas Klein; Thomas Hofmann (2008). "Phosphoric Acid and Phosphates". Ullmann's Encyclopedia of Industrial Chemistry. Ullmann’s Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_465.pub3. ISBN 978-3527306732.
 |

