Chemistry:Bis(trimethylsilyl)mercury
From HandWiki
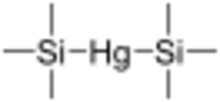
| |
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C6H18HgSi2 | |
| Molar mass | 346.972 g·mol−1 |
| Boiling point | 104 °C (377 K)(dec.) |
| Hazards | |
| GHS pictograms |   
|
| GHS Signal word | Danger |
| H300, H310, H330, H373, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+310, P302+350, P304+340, P310, P314, P320, P321, P322, P330, P361, P363, P391, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bis(trimethylsilyl)mercury is a chemical reagent with the formula (CH3)3-Si-Hg-Si-(CH3)3.
Synthesis
This compound was first synthesized by Wiberg et al. in 1963, by the reaction of trimethylsilyl bromide with sodium amalgam:[1]
- 2 Na + Hg + TMSBr → TMS2Hg + 2 NaBr
Reactions
On prolonged heating at 100-160 °C, or when stood under light as an ethereal solution, it decomposes to hexamethyldisilane:[1]
- TMS2Hg → (CH3)3Si-Si(CH3)3 + Hg
Reaction with hydrogen chloride gives trimethylsilane and trimethylsilyl chloride:[1]
- TMS2Hg + HCl → TMSH + TMSCl + Hg
References
- ↑ 1.0 1.1 1.2 Wiberg, E.; Stecher, O.; Andrascheck, H. J.; Kreuzbichler, L.; Staude, E. (1963). "Recent Developments in the Chemistry of Metal Silyls of the Type M(SiR3)n". Angew. Chem. Int. Ed. Engl. 2 (9): 507. doi:10.1002/anie.196305071.
Further reading
- Pickett, Nl; Just, O; Vanderveer, Dg; Rees Ws, Jr (Apr 2000). "Reinvestigation of bis(trimethylsilyl)mercury". Acta Crystallographica C 56 (4): 412–3. doi:10.1107/S0108270199016339. PMID 10815189.
 |

