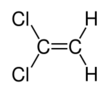
Chemistry:1,1-Dichloroethene
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,1-Dichloroethene | |||
| Other names
1,1-Dichloroethylene
1,1-DCE Vinylidene chloride Vinylidene dichloride Asymmetrical dichloroethene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C2H2Cl2 | |||
| Molar mass | 96.94 g/mol | ||
| Density | 1.213 g/cm3 | ||
| Melting point | −122 °C (−188 °F; 151 K) | ||
| Boiling point | 32 °C (90 °F; 305 K) | ||
| 2,240mg/L (25°C) | |||
| Vapor pressure | 500 mmHg (20°C)[1] | ||
| -49.2·10−6 cm3/mol | |||
| 1.3 D | |||
| Structure | |||
| C2v | |||
| Planar | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −22.8 °C (−9.0 °F; 250.3 K) | ||
| Explosive limits | 6.5–15.5%[1] | ||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
16,000 ppm (rat, 4 hr) 17,300 ppm (mouse, 2 hr) 16,000 ppm (rat, 8 hr)[2] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[1] | ||
REL (Recommended)
|
Ca[1] | ||
IDLH (Immediate danger)
|
Ca [N.D.][1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
1,1-Dichloroethene, commonly called 1,1-dichloroethylene or vinylidene chloride or 1,1-DCE, is an organochloride with the molecular formula C2H2Cl2. It is a colorless liquid with a sharp odor. Like most chlorocarbons, it is poorly soluble in water, but soluble in organic solvents. 1,1-DCE was the precursor to the original clingwrap, Saran, for food, but this application has been phased out.
Production
1,1-DCE is produced by dehydrochlorination of 1,1,2-trichloroethane, a relatively unwanted byproduct in the production of 1,1,1-trichloroethane and 1,2-dichloroethane. The conversion is a base-catalyzed reaction which uses either NaOH or Ca(OH)2 with temperature ca. 100 °C.[3]
- Cl2CHCH2Cl + NaOH → Cl2C=CH2 + NaCl + H2O
The gas phase reaction, without the base, would be more desirable but is less selective.[4]
Applications
1,1-DCE is mainly used as a comonomer in the polymerization of vinyl chloride, acrylonitrile, and acrylates. It is also used in semiconductor device fabrication for growing high purity silicon dioxide (SiO2) films.
Polyvinylidene chloride
As with many other alkenes, 1,1-DCE can be polymerised to form polyvinylidene chloride. A very widely used product, cling wrap, or Saran was made from this polymer. During the 1990s research suggested that, in common with many chlorinated carbon compounds, Saran posed a possible danger to health by leaching, especially on exposure to food in microwave ovens. Since 2004, therefore cling wrap's formulation has changed to a form of polyethylene.
Safety
The health effects from exposure to 1,1-DCE are primarily on the central nervous system, including symptoms of sedation, inebriation, convulsions, spasms, and unconsciousness at high concentrations.[5]
International Agency for Research on Cancer has put vinylidene chloride in Class 2B, meaning possibly carcinogenic to humans. National Institute for Occupational Safety and Health considers vinylidene chloride a potential occupational carcinogen.[6] It is also listed as a chemical known to the state of California to cause cancer and birth defects.[7]
See also
- 1,2-Dichloroethene
- Dichloroethane
References
- ↑ 1.0 1.1 1.2 1.3 1.4 NIOSH Pocket Guide to Chemical Hazards. "#0661". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0661.html.
- ↑ "1,1-Dichloroethane". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/75343.html.
- ↑ Course CEIC2004 Industrial Chemistry for Engineers. Chemistry Lecture Notes, UNSW
- ↑ Manfred Rossberg, Wilhelm Lendle, Gerhard Pfleiderer, Adolf Tögel, Eberhard-Ludwig Dreher, Ernst Langer, Heinz Rassaerts, Peter Kleinschmidt, Heinz Strack, Richard Cook, Uwe Beck, Karl-August Lipper, Theodore R. Torkelson, Eckhard Löser, Klaus K. Beutel, Trevor Mann "Chlorinated Hydrocarbons" in Ullmann's Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_233.pub2.
- ↑ epa.gov
- ↑ CDC - NIOSH Pocket Guide to Chemical Hazards
- ↑ "Chemical Listed Effective December 29, 2017 as Known to the State of California to Cause Cancer: Vinylidene Chloride (1,1-dichloroethylene) | OEHHA". https://oehha.ca.gov/proposition-65/crnr/chemical-listed-effective-december-29-2017-known-state-california-cause-cancer.
External links
- Agency for Toxic Substances and Disease Registry: 1,1-Dichloroethene
 |