Chemistry:Rhein (molecule)
| |

| |
| Names | |
|---|---|
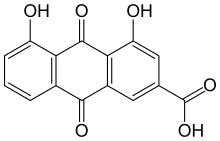
| Preferred IUPAC name
4,5-Dihydroxy-9,10-dioxo-9,10-dihydroanthracene-2-carboxylic acid | |
| Other names
Rhubarb Yellow
Monorhein Cassic acid Rheic acid Chrysazin-3-carboxylic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| KEGG | |
| MeSH | Rhein |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C15H8O6 | |
| Molar mass | 284.22 g/mol |
| Appearance | Orange crystals[1] |
| Density | 1.687 g/cm3 |
| Melting point | 350 to 352 °C (662 to 666 °F; 623 to 625 K)[1] |
| Boiling point | 597.8 °C (1,108.0 °F; 870.9 K) at 760 mmHg |
| Insoluble in water | |
| Hazards | |
| Main hazards | Irritant |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| Flash point | 329.4 °C (624.9 °F; 602.5 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Rhein, also known as cassic acid, is a substance in the anthraquinone group obtained from rhubarb.[2] Like all such substances, rhein is a cathartic, which is commonly found as a glycoside such as rhein-8-glucoside or glucorhein.[2] Rhein was first isolated in 1895.[3] It is found in rhubarb species like Rheum undulatum[4] and Rheum palmatum[5] as well as in Cassia reticulata.[6]
Originally the rhubarb plant which contains rhein was used as a laxative. It was believed that rhein along with other anthraquinone glycosides imparted this activity.[2]
Rhein has been reevaluated as an antibacterial agent against Staphylococcus aureus in 2008.[7] Synergy or partial synergy has been demonstrated between rhein and the antibiotics oxacillin and ampicillin.[8]
Rhein has been shown to inhibit the fat mass and obesity-associated protein, an enzyme responsible for removing the methylation from N6-methyladenosine in nucleic acids.[9][10]
The pharmacokinetics of rhein have not been intensively studied in humans, but at least one study in healthy male volunteers found that rhein was better absorbed from oral administration of rhubarb than from a retention enema.[11] Rhein (at an oral dose of 50 mg twice per day) was shown to be safe when administered for five days to elderly patients with chronic congestive heart failure.[12]
See also
References
- ↑ 1.0 1.1 Cassic acid on www.naturestandard.com
- ↑ 2.0 2.1 2.2 Pharmacognosy of Rhubarb | Chemical Constituents
- ↑ Hesse O (1895). "The Chemistry of rhubarb (EN).". Pharmaceutical Journal and Transactions 1: 325.
- ↑ "Pharmacokinetic analysis of rhein in Rheum undulatum L.". J Ethnopharmacol 84 (1): 5–9. 2003. doi:10.1016/S0378-8741(02)00222-2. PMID 12499069.
- ↑ "Neue Untersuchungen über die Inhaltsstoffe von Rheum palmatum L. 1. Mitteilung: Zur Analytik des Rheins" (in de). Archiv der Pharmazie 292 (64): 591–601. 1959. doi:10.1002/ardp.19592921105. PMID 14402302.
- ↑ Anchel M (1949). "Identification of the antibiotic substance from Cassia reticulata as 4,5-Dihydroxyanthraquinone-2-carboxylic acid". J Biol Chem 177 (1): 169–177. doi:10.1016/S0021-9258(18)57072-1. PMID 18123056. http://www.jbc.org/content/177/1/169.full.pdf.
- ↑ "Global transcriptional response of Staphylococcus aureus to rhein, a natural plant product". J Biotechnol 135 (3): 304–308. 2008. doi:10.1016/j.jbiotec.2008.04.010. PMID 18514345.
- ↑ "Synergistic effect of rhein in combination with ampicillin or oxacillin against methicillin-resistant Staphylococcus aureus". Exp Ther Med 3 (4): 608–612. 2012. doi:10.3892/etm.2012.459. PMID 22969937.
- ↑ Yu, Jun; Chen, Mengxian; Huang, Haijiao; Zhu, Junda; Song, Huixue; Zhu, Jian; Park, Jaewon; Ji, Sheng-Jian (2017-11-23). "Dynamic m6A modification regulates local translation of mRNA in axons" (in en). Nucleic Acids Research 46 (3): 1412–1423. doi:10.1093/nar/gkx1182. ISSN 0305-1048. PMID 29186567.
- ↑ Jia, Guifang; Fu, Ye; Zhao, Xu; Dai, Qing; Zheng, Guanqun; Yang, Ying; Yi, Chengqi; Lindahl, Tomas et al. (2011-10-16). "N6-Methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO" (in En). Nature Chemical Biology 7 (12): 885–887. doi:10.1038/nchembio.687. ISSN 1552-4450. PMID 22002720.
- ↑ "Pharmacokinetic of rhein in healthy male volunteers following oral and retention enema administration of rhubarb extract: a single dose study". Am J Chin Med 33 (6): 839–850. 2005. doi:10.1142/S0192415X05003508. PMID 16355440.
- ↑ "Effects of rhein on renal arachidonic acid metabolism and renal function in patients with congestive heart failure". Eur J Clin Pharmacol 37 (1): 1–5. 1989. doi:10.1007/bf00609415. PMID 2512175.
 |

