Biology:Glycerol-3-phosphate dehydrogenase
| Glycerol-3-phosphate dehydrogenase (NAD+) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
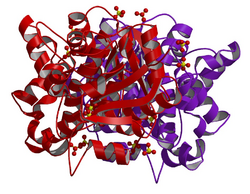 Crystallographic structure of human glycerol-3-phosphate dehydrogenase 1.[1] | |||||||||
| Identifiers | |||||||||
| EC number | 1.1.1.8 | ||||||||
| CAS number | 9075-65-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Glycerol-3-phosphate dehydrogenase (quinone) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 1.1.5.3 | ||||||||
| CAS number | 9001-49-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
| NAD-dependent glycerol-3-phosphate dehydrogenase N-terminus | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of the n-(1-d-carboxylethyl)-l-norvaline dehydrogenase from arthrobacter sp. strain 1c | |||||||||
| Identifiers | |||||||||
| Symbol | NAD_Gly3P_dh_N | ||||||||
| Pfam | PF01210 | ||||||||
| Pfam clan | CL0063 | ||||||||
| InterPro | IPR011128 | ||||||||
| PROSITE | PDOC00740 | ||||||||
| SCOP2 | 1m66 / SCOPe / SUPFAM | ||||||||
| |||||||||
| NAD-dependent glycerol-3-phosphate dehydrogenase C-terminus | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 structure of glycerol-3-phosphate dehydrogenase from archaeoglobus fulgidus | |||||||||
| Identifiers | |||||||||
| Symbol | NAD_Gly3P_dh_C | ||||||||
| Pfam | PF07479 | ||||||||
| Pfam clan | CL0106 | ||||||||
| InterPro | IPR006109 | ||||||||
| PROSITE | PDOC00740 | ||||||||
| SCOP2 | 1m66 / SCOPe / SUPFAM | ||||||||
| |||||||||
Glycerol-3-phosphate dehydrogenase (GPDH) is an enzyme that catalyzes the reversible redox conversion of dihydroxyacetone phosphate (a.k.a. glycerone phosphate, outdated) to sn-glycerol 3-phosphate.[2]
Glycerol-3-phosphate dehydrogenase serves as a major link between carbohydrate metabolism and lipid metabolism. It is also a major contributor of electrons to the electron transport chain in the mitochondria.
Older terms for glycerol-3-phosphate dehydrogenase include alpha glycerol-3-phosphate dehydrogenase (alphaGPDH) and glycerolphosphate dehydrogenase (GPDH). However, glycerol-3-phosphate dehydrogenase is not the same as glyceraldehyde 3-phosphate dehydrogenase (GAPDH), whose substrate is an aldehyde not an alcohol.
Metabolic function
GPDH plays a major role in lipid biosynthesis. Through the reduction of dihydroxyacetone phosphate into glycerol 3-phosphate, GPDH allows the prompt dephosphorylation of glycerol 3-phosphate into glycerol.[3] Additionally, GPDH is one of the enzymes involved in maintaining the redox potential across the inner mitochondrial membrane.[3]

Reaction
The NAD+/NADH coenzyme couple act as an electron reservoir for metabolic redox reactions, carrying electrons from one reaction to another.[5] Most of these metabolism reactions occur in the mitochondria. To regenerate NAD+ for further use, NADH pools in the cytosol must be reoxidized. Since the mitochondrial inner membrane is impermeable to both NADH and NAD+, these cannot be freely exchanged between the cytosol and mitochondrial matrix.[4]
One way to shuttle this reducing equivalent across the membrane is through the Glycerol-3-phosphate shuttle, which employs the two forms of GPDH:
- Cytosolic GPDH, or GPD1, is localized to the outer membrane of the mitochondria facing the cytosol, and catalyzes the reduction of dihydroxyacetone phosphate into glycerol-3-phosphate.
- In conjunction, Mitochondrial GPDH, or GPD2, is embedded on the outer surface of the inner mitochondrial membrane, overlooking the cytosol, and catalyzes the oxidation of glycerol-3-phosphate to dihydroxyacetone phosphate.[6]
The reactions catalyzed by cytosolic (soluble) and mitochondrial GPDH are as follows:
Variants
There are two forms of GPDH:
| Enzyme | Protein | Gene | |||||
|---|---|---|---|---|---|---|---|
| EC number | Name | Donor / Acceptor | Name | Subcellular location | Abbreviation | Name | Symbol |
| 1.1.1.8 | glycerol-3-phosphate dehydrogenase | NADH / NAD+ | Glycerol-3-phosphate dehydrogenase [NAD+] | cytoplasmic | GPDH-C | glycerol-3-phosphate dehydrogenase 1 (soluble) | GPD1 |
| 1.1.5.3 | glycerol-3-phosphate dehydrogenase | quinol / quinone | Glycerol-3-phosphate dehydrogenase | mitochondrial | GPDH-M | glycerol-3-phosphate dehydrogenase 2 (mitochondrial) | GPD2 |
The following human genes encode proteins with GPDH enzymatic activity:
|
| ||||||||||||||||||||||||||||||||||||||||||||
GPD1
Cytosolic Glycerol-3-phosphate dehydrogenase (GPD1), is an NAD+-dependent enzyme[8] that reduces dihydroxyacetone phosphate to glycerol-3-phosphate. Simultaneously, NADH is oxidized to NAD+ in the following reaction:
As a result, NAD+ is regenerated for further metabolic activity.
GPD1 consists of two subunits,[9] and reacts with dihydroxyacetone phosphate and NAD+ though the following interaction:
Figure 4. The putative active site. The phosphate group of DHAP is half-encircled by the side-chain of Arg269, and interacts with Arg269 and Gly268 directly by hydrogen bonds (not shown). The conserved residues Lys204, Asn205, Asp260 and Thr264 form a stable hydrogen bonding network. The other hydrogen bonding network includes residues Lys120 and Asp260, as well as an ordered water molecule (with a B-factor of 16.4 Å2), which hydrogen bonds to Gly149 and Asn151 (not shown). In these two electrostatic networks, only the ε-NH3+ group of Lys204 is the nearest to the C2 atom of DHAP (3.4 Å).[1]
GPD2
Mitochondrial glycerol-3-phosphate dehydrogenase (GPD2), catalyzes the irreversible oxidation of glycerol-3-phosphate to dihydroxyacetone phosphate and concomitantly transfers two electrons from FAD to the electron transport chain. GPD2 consists of 4 identical subunits.[10]
Response to environmental stresses
- Studies indicate that GPDH is mostly unaffected by pH changes: neither GPD1 or GPD2 is favored under certain pH conditions.
- At high salt concentrations (E.g. NaCl), GPD1 activity is enhanced over GPD2, since an increase in the salinity of the medium leads to an accumulation of glycerol in response.
- Changes in temperature do not appear to favor neither GPD1 nor GPD2.[11]
Glycerol-3-phosphate shuttle
The cytosolic together with the mitochondrial glycerol-3-phosphate dehydrogenase work in concert. Oxidation of cytoplasmic NADH by the cytosolic form of the enzyme creates glycerol-3-phosphate from dihydroxyacetone phosphate. Once the glycerol-3-phosphate has moved through the outer mitochondrial membrane it can then be oxidised by a separate isoform of glycerol-3-phosphate dehydrogenase that uses quinone as an oxidant and FAD as a co-factor. As a result, there is a net loss in energy, comparable to one molecule of ATP.[7]
The combined action of these enzymes maintains the NAD+/NADH ratio that allows for continuous operation of metabolism.
Role in disease
The fundamental role of GPDH in maintaining the NAD+/NADH potential, as well as its role in lipid metabolism, makes GPDH a factor in lipid imbalance diseases, such as obesity.
- Enhanced GPDH activity, particularly GPD2, leads to an increase in glycerol production. Since glycerol is a main subunit in lipid metabolism, its abundance can easily lead to an increase in triglyceride accumulation at a cellular level. As a result, there is a tendency to form adipose tissue leading to an accumulation of fat that favors obesity.[12]
- GPDH has also been found to play a role in Brugada syndrome. Mutations in the gene encoding GPD1 have been proven to cause defects in the electron transport chain. This conflict with NAD+/NADH levels in the cell is believed to contribute to defects in cardiac sodium ion channel regulation and can lead to a lethal arrythmia during infancy.[13]
Pharmacological target
The mitochondrial isoform of G3P dehydrogenase is thought to be inhibited by metformin, a first line drug for type 2 diabetes. [14]
Biological Research
Sarcophaga barbata was used to study the oxidation of L-3-glycerophosphate in mitochondria. It is found that the L-3-glycerophosphate does not enter the mitochondrial matrix, unlike pyruvate. This helps locate the L-3-glycerophosphate-flavoprotein oxidoreductase, which is on the inner membrane of the mitochondria.
Structure
Glycerol-3-phosphate dehydrogenase consists of two protein domains. The N-terminal domain is an NAD-binding domain, and the C-terminus acts as a substrate-binding domain.[15] However, dimer and tetramer interface residues are involved in GAPDH-RNA binding, as GAPDH can exhibit several moonlighting activities, including the modulation of RNA binding and/or stability.[16]
See also
- substrate pages: glycerol 3-phosphate, dihydroxyacetone phosphate
- related topics: glycerol phosphate shuttle, creatine kinase, glycolysis, gluconeogenesis
References
- ↑ 1.0 1.1 PDB: 1X0V; "Crystal structures of human glycerol 3-phosphate dehydrogenase 1 (GPD1)". Journal of Molecular Biology 357 (3): 858–69. Mar 2006. doi:10.1016/j.jmb.2005.12.074. PMID 16460752.
- ↑ "Crystal structures of human glycerol 3-phosphate dehydrogenase 1 (GPD1)". Journal of Molecular Biology 357 (3): 858–69. Mar 2006. doi:10.1016/j.jmb.2005.12.074. PMID 16460752.
- ↑ 3.0 3.1 "Role of glycerol 3-phosphate dehydrogenase in glyceride metabolism. Effect of diet on enzyme activities in chicken liver". The Biochemical Journal 146 (1): 223–9. Jan 1975. doi:10.1042/bj1460223. PMID 167714.
- ↑ 4.0 4.1 "Physiological and genetic engineering of cytosolic redox metabolism in Saccharomyces cerevisiae for improved glycerol production". Metabolic Engineering 8 (6): 532–42. Nov 2006. doi:10.1016/j.ymben.2006.06.004. PMID 16891140.
- ↑ "The two isoenzymes for yeast NAD+-dependent glycerol 3-phosphate dehydrogenase encoded by GPD1 and GPD2 have distinct roles in osmoadaptation and redox regulation". The EMBO Journal 16 (9): 2179–87. May 1997. doi:10.1093/emboj/16.9.2179. PMID 9171333.
- ↑ "Role of glycerol-3-phosphate dehydrogenase 2 in mouse sperm capacitation". Molecular Reproduction and Development 77 (9): 773–83. Sep 2010. doi:10.1002/mrd.21218. PMID 20602492.
- ↑ 7.0 7.1 Stryer, Lubert; Berg, Jeremy Mark; Tymoczko, John L. (2002). "Chapter 18.5: Glycerol 3-Phosphate Shuttle". Biochemistry. San Francisco: W.H. Freeman. ISBN 0-7167-4684-0. https://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=stryer&part=A2548&rendertype=figure&id=A2548.
- ↑ "The influence of obstructive sleep apnea on the expression of glycerol-3-phosphate dehydrogenase 1 gene". Experimental Biology and Medicine 235 (1): 52–6. Jan 2010. doi:10.1258/ebm.2009.009150. PMID 20404019. http://ebm.rsmjournals.com/cgi/content/full/235/1/52. Retrieved 2011-05-16.
- ↑ "Competition of electrons to enter the respiratory chain: a new regulatory mechanism of oxidative metabolism in Saccharomyces cerevisiae". The Journal of Biological Chemistry 280 (5): 3407–13. Feb 2005. doi:10.1074/jbc.M407746200. PMID 15557339.
- ↑ "Tyrosine phosphoproteome of hamster spermatozoa: role of glycerol-3-phosphate dehydrogenase 2 in sperm capacitation". Proteomics 9 (7): 1809–26. Apr 2009. doi:10.1002/pmic.200800519. PMID 19333995.
- ↑ "Differential response of the catalase, superoxide dismutase and glycerol-3-phosphate dehydrogenase to different environmental stresses in Debaryomyces nepalensis NCYC 3413". Current Microbiology 62 (2): 382–7. Feb 2011. doi:10.1007/s00284-010-9717-z. PMID 20644932.
- ↑ "Attenuating effect of casein glycomacropeptide on proliferation, differentiation, and lipid accumulation of in vitro Sprague-Dawley rat preadipocytes". Journal of Dairy Science 94 (2): 676–83. Feb 2011. doi:10.3168/jds.2010-3827. PMID 21257036.
- ↑ "Molecular and functional characterization of novel glycerol-3-phosphate dehydrogenase 1 like gene (GPD1-L) mutations in sudden infant death syndrome". Circulation 116 (20): 2253–9. Nov 2007. doi:10.1161/CIRCULATIONAHA.107.704627. PMID 17967976.
- ↑ "The target of metformin in type 2 diabetes". The New England Journal of Medicine 371 (16): 1547–8. Oct 2014. doi:10.1056/NEJMcibr1409796. PMID 25317875.
- ↑ "A potential target enzyme for trypanocidal drugs revealed by the crystal structure of NAD-dependent glycerol-3-phosphate dehydrogenase from Leishmania mexicana". Structure 8 (5): 541–52. May 2000. doi:10.1016/s0969-2126(00)00135-0. PMID 10801498.
- ↑ "A dimer interface mutation in glyceraldehyde-3-phosphate dehydrogenase regulates its binding to AU-rich RNA" (in en). The Journal of Biological Chemistry 290 (3): 1770–85. Jan 2015. doi:10.1074/jbc.M114.618165. PMID 25451934.
Further reading
- Baranowski T (1963). "α-Glycerophosphate dehydrogenase". The Enzymes (2nd ed.). New York: Academic Press. pp. 85–96.
- "Comparative structural properties of honeybee and rabbit alpha-glycerophosphate dehydrogenases". Biochemistry 8 (5): 2095–105. May 1969. doi:10.1021/bi00833a047. PMID 4307630.
- "The -glycerophosphate cycle in Drosophila melanogaster. I. Biochemical and developmental aspects". Biochemical Genetics 7 (2): 141–61. Oct 1972. doi:10.1007/BF00486085. PMID 4340553.
- "Isolation and characterization of cytoplasmic L-glycerol-3-phosphate dehydrogenase from rabbit-renal-adipose tissue and its comparison with the skeletal-muscle enzyme". European Journal of Biochemistry 36 (1): 97–109. Jul 1973. doi:10.1111/j.1432-1033.1973.tb02889.x. PMID 4200180.
- "Purification and characterization of glycerol-3-phosphate dehydrogenase of Saccharomyces cerevisiae". FEBS Letters 308 (2): 130–2. Aug 1992. doi:10.1016/0014-5793(92)81259-O. PMID 1499720.
- "Isolation and characterization of adipose tissue glycerol-3-phosphate dehydrogenase". The International Journal of Biochemistry & Cell Biology 27 (6): 625–32. Jun 1995. doi:10.1016/1357-2725(95)00012-E. PMID 7671141.
- "Kinetic regulation of the mitochondrial glycerol-3-phosphate dehydrogenase by the external NADH dehydrogenase in Saccharomyces cerevisiae". The Journal of Biological Chemistry 277 (31): 27991–5. Aug 2002. doi:10.1074/jbc.M204079200. PMID 12032156. http://www.jbc.org/content/277/31/27991.full.
- "In vivo analysis of the mechanisms for oxidation of cytosolic NADH by Saccharomyces cerevisiae mitochondria". Journal of Bacteriology 182 (10): 2823–30. May 2000. doi:10.1128/JB.182.10.2823-2830.2000. PMID 10781551.
- "Reconstruction of the alpha-glycerolphosphate shuttle using rat kidney mitochondria". FEBS Letters 91 (2): 169–72. Jul 1978. doi:10.1016/0014-5793(78)81164-8. PMID 210038.
- "Localization of glycerol-3-phosphate oxidase in the mitochondrion and particulate NAD+-linked glycerol-3-phosphate dehydrogenase in the microbodies of the bloodstream form to Trypanosoma brucei". European Journal of Biochemistry 76 (1): 29–39. Jun 1977. doi:10.1111/j.1432-1033.1977.tb11567.x. PMID 142010.
- "Mechanism of action of a flavin-containing monooxygenase". Proceedings of the National Academy of Sciences of the United States of America 103 (26): 9832–7. Jun 2006. doi:10.1073/pnas.0602398103. PMID 16777962. Bibcode: 2006PNAS..103.9832E.
External links
- equivalent entries:
- alphaGPDH at the US National Library of Medicine Medical Subject Headings (MeSH)
- GPDH
- Yeast genome database GO term: GPDH
 |




