Biology:Microprocessor complex
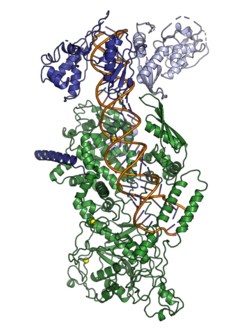
The microprocessor complex is a protein complex involved in the early stages of processing microRNA (miRNA) and RNA interference (RNAi) in animal cells.[2][3] The complex is minimally composed of the ribonuclease enzyme Drosha and the dimeric RNA-binding protein DGCR8 (also known as Pasha in non-human animals), and cleaves primary miRNA substrates to pre-miRNA in the cell nucleus.[4][5][6] Microprocessor is also the smaller of the two multi-protein complexes that contain human Drosha.[7]
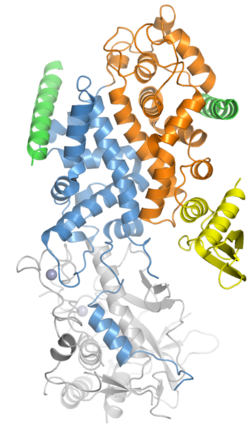
Composition
The microprocessor complex consists minimally of two proteins: Drosha, a ribonuclease III enzyme; and DGCR8, a double-stranded RNA binding protein.[4][5][6] (DGCR8 is the name used in mammalian genetics, abbreviated from "DiGeorge syndrome critical region 8"; the homologous protein in model organisms such as flies and worms is called Pasha, for Partner of Drosha.) The stoichiometry of the minimal complex was at one point experimentally difficult to determine, but it has been demonstrated to be a heterotrimer of two DGCR8 proteins and one Drosha.[1][8][9][10]
In addition to the minimal catalytically active microprocessor components, other cofactors such as DEAD box RNA helicases and heterogeneous nuclear ribonucleoproteins may be present in the complex to mediate the activity of Drosha.[4] Some miRNAs are processed by microprocessor only in the presence of specific cofactors.[11]
Function
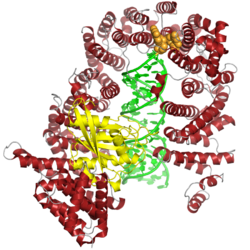
Located in the cell nucleus, the microprocessor complex cleaves primary miRNA (pri-miRNA) into precursor miRNA (pre-miRNA).[13] Its two subunits have been determined as necessary and sufficient for the mediation of the development of miRNAs from the pri-miRNAs.[7] These molecules of around 70 nucleotides contain a stem-loop or hairpin structure. Pri-miRNA substrates can be derived either from non-coding RNA genes or from introns. In the latter case, there is evidence that the microprocessor complex interacts with the spliceosome and that the pri-miRNA processing occurs prior to splicing.[5][14]
Microprocessor cleavage of pri-miRNAs typically occurs co-transcriptionally and leaves a characteristic RNase III single-stranded overhang of 2-3 nucleotides, which serves as a recognition element for the transport protein exportin-5.[15] Pre-miRNAs are exported from the nucleus to the cytoplasm in a RanGTP-dependent manner and are further processed, typically by the endoribonuclease enzyme Dicer.[4][5][6]
Hemin allows for the increased processing of pri-miRNAs through an induced conformational change of the DGCR8 subunit, and also enhances DGCR8's binding specificity for RNA.[16] DGCR8 recognizes the junctions between hairpin structures and single-stranded RNA and serves to orient Drosha to cleave around 11 nucleotides away from the junctions, and remains in contact with the pri-miRNAs following cleavage and dissociation of Drosha.[17]
Although the large majority of miRNAs undergo processing by microprocessor, a small number of exceptions called mirtrons have been described; these are very small introns which, after splicing, have the appropriate size and stem-loop structure to serve as a pre-miRNA.[18] The processing pathways for microRNA and for exogenously derived small interfering RNA converge at the point of Dicer processing and are largely identical downstream. Broadly defined, both pathways constitute RNAi.[5][18] Microprocessor is also found to be involved in ribosomal biogenesis specifically in the removal of R-loops and activating transcription of ribosomal protein encoding genes.[19]
Regulation
Gene regulation by miRNA is widespread across many genomes – by some estimates more than 60% of human protein-coding genes are likely to be regulated by miRNA,[20] though the quality of experimental evidence for miRNA-target interactions is often weak.[21] Because processing by microprocessor is a major determinant of miRNA abundance, microprocessor itself is then an important target of regulation.
Both Drosha and DGCR8 are subject to regulation by post-translational modifications modulating stability, intracellular localization, and activity levels. Activity against particular substrates may be regulated by additional protein cofactors interacting with the microprocessor complex. The loop region of the pri-miRNA stem-loop is also a recognition element for regulatory proteins, which may up- or down-regulate microprocessor processing of the specific miRNAs they target.[11]
Microprocessor itself is auto-regulated by negative feedback through association with a pri-miRNA-like hairpin structure found in the DGCR8 mRNA, which when cleaved reduces DGCR8 expression. The structure in this case is located in an exon and is unlikely to itself function as miRNA in its own right.[11]
Evolution
Drosha shares striking structural similarity with the downstream ribonuclease Dicer, suggesting an evolutionary relationship, though Drosha and related enzymes are found only in animals while Dicer relatives are widely distributed, including among protozoans.[8] Both components of the microprocessor complex are conserved among the vast majority of metazoans with known genomes. Mnemiopsis leidyi, a ctenophore, lacks both Drosha and DGCR8 homologs, as well as recognizable miRNAs, and is the only known metazoan with no detectable genomic evidence of Drosha.[22] In plants, the miRNA biogenesis pathway is somewhat different; neither Drosha nor DGCR8 has a homolog in plant cells, where the first step in miRNA processing is usually executed by a different nuclear ribonuclease, DCL1, a homolog of Dicer.[11][23]
It has been suggested based on phylogenetic analysis that the key components of RNA interference based on exogenous substrates were present in the ancestral eukaryote, likely as an immune mechanism against viruses and transposable elements. Elaboration of this pathway for miRNA-mediated gene regulation is thought to have evolved later.[24]
Clinical significance
The involvement of miRNAs in diseases has led scientists to become more interested in the role of additional protein complexes, like microprocessor, that have the ability to influence or modulate the function and expression of miRNAs.[25] Microprocessor complex component, DGCR8, is affected through the micro-deletion of 22q11.2, a small portion of chromosome 22. This deletion causes irregular processing of miRNAs which leads to DiGeorge Syndrome.[26]
References
- ↑ 1.0 1.1 Partin, Alexander C.; Zhang, Kaiming; Jeong, Byung-Cheon; Herrell, Emily; Li, Shanshan; Chiu, Wah; Nam, Yunsun (May 2020). "Cryo-EM Structures of Human Drosha and DGCR8 in Complex with Primary MicroRNA". Molecular Cell 78 (3): 411–422.e4. doi:10.1016/j.molcel.2020.02.016. PMID 32220646.
- ↑ "The Microprocessor complex mediates the genesis of microRNAs". Nature 432 (7014): 235–40. November 2004. doi:10.1038/nature03120. PMID 15531877. Bibcode: 2004Natur.432..235G.
- ↑ "Processing of primary microRNAs by the Microprocessor complex". Nature 432 (7014): 231–5. November 2004. doi:10.1038/nature03049. PMID 15531879. Bibcode: 2004Natur.432..231D.
- ↑ 4.0 4.1 4.2 4.3 "Posttranscriptional regulation of microRNA biogenesis in animals". Molecular Cell 38 (3): 323–32. May 2010. doi:10.1016/j.molcel.2010.03.013. PMID 20471939.
- ↑ 5.0 5.1 5.2 5.3 5.4 "Molecular mechanisms of RNA interference". Annual Review of Biophysics 42: 217–39. 2013. doi:10.1146/annurev-biophys-083012-130404. PMID 23654304.
- ↑ 6.0 6.1 6.2 "Cellular functions of the microprocessor". Biochemical Society Transactions 41 (4): 838–43. August 2013. doi:10.1042/BST20130011. PMID 23863141.
- ↑ 7.0 7.1 "The Microprocessor complex mediates the genesis of microRNAs". Nature 432 (7014): 235–40. November 2004. doi:10.1038/nature03120. PMID 15531877. Bibcode: 2004Natur.432..235G.
- ↑ 8.0 8.1 8.2 "Structure of Human DROSHA". Cell 164 (1–2): 81–90. January 2016. doi:10.1016/j.cell.2015.12.019. PMID 26748718.
- ↑ "A heterotrimer model of the complete Microprocessor complex revealed by single-molecule subunit counting". RNA 22 (2): 175–83. February 2016. doi:10.1261/rna.054684.115. PMID 26683315.
- ↑ "Functional Anatomy of the Human Microprocessor". Cell 161 (6): 1374–87. June 2015. doi:10.1016/j.cell.2015.05.010. PMID 26027739.
- ↑ 11.0 11.1 11.2 11.3 "Regulation of microRNA biogenesis". Nature Reviews. Molecular Cell Biology 15 (8): 509–24. August 2014. doi:10.1038/nrm3838. PMID 25027649.
- ↑ Okada, Chimari; Yamashita, Eiki; Lee, Soo Jae; Shibata, Satoshi; Katahira, Jun; Nakagawa, Atsushi; Yoneda, Yoshihiro; Tsukihara, Tomitake (2009-11-27). "A High-Resolution Structure of the Pre-microRNA Nuclear Export Machinery". Science 326 (5957): 1275–1279. doi:10.1126/science.1178705. PMID 19965479. Bibcode: 2009Sci...326.1275O.
- ↑ "Post-transcriptional control of miRNA biogenesis". RNA 25 (1): 1–16. January 2019. doi:10.1261/rna.068692.118. PMID 30333195.
- ↑ "Functional association of the Microprocessor complex with the spliceosome". Molecular and Cellular Biology 29 (12): 3243–54. June 2009. doi:10.1128/MCB.00360-09. PMID 19349299.
- ↑ "Primary microRNA transcripts are processed co-transcriptionally". Nature Structural & Molecular Biology 15 (9): 902–9. September 2008. doi:10.1038/nsmb.1475. PMID 19172742.
- ↑ "Heme enables proper positioning of Drosha and DGCR8 on primary microRNAs". Nature Communications 8 (1): 1737. November 2017. doi:10.1038/s41467-017-01713-y. PMID 29170488. Bibcode: 2017NatCo...8.1737P.
- ↑ "Microprocessor dynamics and interactions at endogenous imprinted C19MC microRNA genes". Journal of Cell Science 125 (Pt 11): 2709–20. June 2012. doi:10.1242/jcs.100354. PMID 22393237.
- ↑ 18.0 18.1 "Many roads to maturity: microRNA biogenesis pathways and their regulation". Nature Cell Biology 11 (3): 228–34. March 2009. doi:10.1038/ncb0309-228. PMID 19255566.
- ↑ "Control of ribosomal protein synthesis by the Microprocessor complex". Science Signaling 14 (671): eabd2639. February 2021. doi:10.1126/scisignal.abd2639. PMID 33622983.
- ↑ "Most mammalian mRNAs are conserved targets of microRNAs". Genome Research 19 (1): 92–105. January 2009. doi:10.1101/gr.082701.108. PMID 18955434.
- ↑ "Validated MicroRNA Target Databases: An Evaluation". Drug Development Research 76 (7): 389–96. November 2015. doi:10.1002/ddr.21278. PMID 26286669.
- ↑ "MicroRNAs and essential components of the microRNA processing machinery are not encoded in the genome of the ctenophore Mnemiopsis leidyi". BMC Genomics 13: 714. December 2012. doi:10.1186/1471-2164-13-714. PMID 23256903.
- ↑ "Vive la différence: biogenesis and evolution of microRNAs in plants and animals". Genome Biology 12 (4): 221. 2011. doi:10.1186/gb-2011-12-4-221. PMID 21554756.
- ↑ "On the origin and functions of RNA-mediated silencing: from protists to man". Current Genetics 50 (2): 81–99. August 2006. doi:10.1007/s00294-006-0078-x. PMID 16691418.
- ↑ "Microprocessor of microRNAs: regulation and potential for therapeutic intervention". Molecular Cancer 9 (1): 134. June 2010. doi:10.1186/1476-4598-9-134. PMID 20515486.
- ↑ "Deficiency of Dgcr8, a gene disrupted by the 22q11.2 microdeletion, results in altered short-term plasticity in the prefrontal cortex". Proceedings of the National Academy of Sciences of the United States of America 108 (11): 4447–52. March 2011. doi:10.1073/pnas.1101219108. PMID 21368174. Bibcode: 2011PNAS..108.4447F.
 |
