Biology:Protamine
| Protamine 1 | |
|---|---|
| Identifiers | |
| Symbol | PRM1 |
| NCBI gene | 5619 |
| HGNC | 9447 |
| OMIM | 182880 |
| RefSeq | NM_002761 |
| UniProt | P04553 |
| Other data | |
| Locus | Chr. 16 p13.13 |
| Protamine 2 | |
|---|---|
| Identifiers | |
| Symbol | PRM2 |
| NCBI gene | 5620 |
| HGNC | 9448 |
| OMIM | 182890 |
| RefSeq | NM_002762 |
| UniProt | P04554 |
| Other data | |
| Locus | Chr. 16 p13.13 |
Protamines are small, arginine-rich, nuclear proteins that replace histones late in the haploid phase of spermatogenesis and are believed essential for sperm head condensation via genomic DNA compaction and stabilization. They may allow for denser packaging of DNA in the spermatozoon than histones, but they must be decompressed before the genetic data can be used for protein synthesis. However, part of the sperm's genome is packaged by histones (10-15% in humans and other primates) thought to bind genes that are essential for early embryonic development.[1]
Protamine and protamine-like (PL) proteins are collectively known as the sperm-specific nuclear basic proteins (SNBPs). The PL proteins are intermediate in structure between protamine and Histone H1. The C-terminal domain of PL could be the precursor of vertebrate protamine.[2]
Spermatogenesis
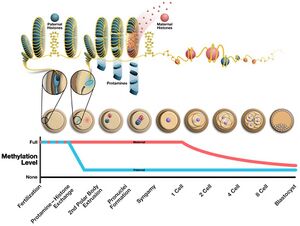
During the formation of sperm, protamine binds to the phosphate backbone of DNA using the arginine-rich domain as an anchor. DNA is then folded into a toroid, an O-shaped structure, although the mechanism is not known. A sperm cell can contain up to 50,000 toroid-shaped structures in its nucleus with each toroid containing about 50 kilobases.[4] Before the toroid is formed, histones are removed from the DNA by transition nuclear proteins, so that protamine can condense it. The effects of this change are 1) an increase in sperm hydrodynamics for better flow through liquids by reducing the head size 2) decrease in the occurrence of DNA damage 3) removal of the epigenetic markers that occur with histone modifications.[5]
The structure of the sperm head is also related to protamine levels. The ratio of protamine 2 to protamine 1 and transition nuclear proteins has been found to change the sperm head shape in various species of mice, by altering the expression of protamine 2 via mutations in its promoter region. A decrease in the ratio has been found to increase the competitive ability of sperm in Mus species. However, further testing is required to determine how this ratio influences the shape of the head and whether monogamy influences this selection. In humans, studies show that men who have unbalanced Prm1/Prm2 are subfertile or infertile.[6] Protamine 2 is encoded as a longer protein that needs its N-terminal cleaved before becoming functional. Human and chimp protamine has undergone rapid evolution.[7]
Medical uses
When mixed with insulin, protamines slow down the onset and increase the duration of insulin action (see NPH insulin).[8]
Protamine is used in cardiac surgery, vascular surgery, and interventional radiology procedures to neutralize the anti-clotting effects of heparin. Adverse effects include increased pulmonary artery pressure and decrease peripheral blood pressure, myocardial oxygen consumption, cardiac output, and heart rate.[9]
Protamine sulfate is an antidote for heparin overdose, but severe allergy may occur.[10] A chain shortened version of protamine also acts as a potent heparin antagonist, but with markedly reduced antigenicity. It was initially produced as a mixture made by thermolysin digestion of protamine,[11] but the actual effective peptide portion VSRRRRRRGGRRRR has since been isolated.[12] An analogue of this peptide has also been produced.[13]
In gene therapy, protamine sulfate's ability to condense plasmid DNA along with its approval by the U.S. Food and Drug Administration (FDA) have made it an appealing candidate to increase transduction rates by both viral[14] and nonviral (e.g. utilizing cationic liposomes)[15] mediated delivery mechanisms.
Protamine may be used as a drug to prevent obesity. Protamine has been shown to deter increases in body weight and low-density lipoprotein in high-fat diet rats. This effect occurs through the inhibition of lipase activity, an enzyme responsible for triacylglycerol digestion and absorption, resulting in a decrease in the absorption of dietary fat. No liver damage was found when the rats were treated with protamine. However, emulsification of long-chain fatty acids for digestion and absorption in the small intestine is less constant in humans than rats, which will vary the effectiveness of protamine as a drug. Furthermore, human peptidases may degrade protamine at different rates, thus further tests are required to determine protamine's ability to prevent obesity in humans.[16]
Species distribution and isoforms
Mice, humans[1] and certain fish have two or more different protamines, whereas the sperm of bull and boar,[17] have one form of protamine due to a mutation in the PRM2 gene. In the rat, although the gene for PRM2 is present, expression of this protein is extremely small because of limited transcription due to an inefficient promoter in addition to altered processing of the mRNA transcript.[18]
Mammals
The 2 human protamines are denoted PRM1 and PRM2. In mice and humans, PRM1, PRM2, and TNP2 are co-located in a conserved gene cluster.[19]
Eutherian mammals generally have both PRM1 and PRM2. Metatherians on the other hand only have a homolog to P1.[20]
Fish
Examples of protamines from fish are:
- salmine and protamine sulfate from salmon
- clupeine from herring sperm (Clupea)
- iridine from rainbow trout
- thinnine from tunafish (Thunnus)
- stelline from starry sturgeon (Acipenser stellatus)
- scylliorhinine from dogfish (Scylliorhinus)
Fish protamine are generally shorter than that of mammals, with a higher amount of arginine.[20]
Sequence
| Protamine P1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Protamine_P1 | ||||||||
| Pfam | PF00260 | ||||||||
| InterPro | IPR000221 | ||||||||
| PROSITE | PDOC00047 | ||||||||
| |||||||||
| Protamine P2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Protamine_P2 | ||||||||
| Pfam | PF00841 | ||||||||
| InterPro | IPR000492 | ||||||||
| |||||||||
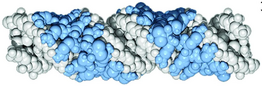
The primary structure of protamine P1, the protamine used for packaging DNA in sperm cells, in placental mammals is usually 49 or 50 amino acids long. This sequence is divided into three separate domains: an arginine-rich domain for DNA binding flanked by shorter peptide sequences containing mostly cysteine residues. The arginine-rich domain consists of 3-11 arginine residues and is conserved between fish protamine and mammalian protamine 1 sequences at about 60-80% sequence identity.[1]
Structure
After translation, the protamine P1 structure is immediately phosphorylated at all three of the above-mentioned domains. Another round of phosphorylation occurs when the sperm enters the egg, but the function of these phosphorylations is uncertain.[1]
The exact secondary and tertiary structure of protamine is not known with certainty, but several proposals have been published since the 1970s.[22][23][1][24][25][20][26] The broad consensus is that protamine forms beta strand structures that then crosslink through disulfide bonds (and potentially dityrosine and cysteine-tyrosine bonds).[25][20] When protamine P1 binds to DNA, cysteine from the amino terminal of one protamine P1 forms disulfide bonds with the cysteine from the carboxy-terminal of another protamine P1. By neutralizing the backbone charge protamine enables the DNA to more tightly coil.[3][26] The disulfide bonds function to prevent the dissociation of protamine P1 from DNA until the bonds are reduced when the sperm enters the egg.[1] These long protamine polymers may then wrap around the DNA within the major groove.[1][23]
The positively charged protamine has the capacity to condense various polyanions. Recent findings indicate that salmon protamine forms a wide range of liquid and solid condensates or aggregates with both single-stranded and double-stranded DNA, as well as with glycosaminoglycans.[27]
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "The protamine family of sperm nuclear proteins". Genome Biology 8 (9): 227. 2007. doi:10.1186/gb-2007-8-9-227. PMID 17903313.
- ↑ "Origin and evolution of chromosomal sperm proteins". BioEssays 31 (10): 1062–70. October 2009. doi:10.1002/bies.200900050. PMID 19708021.
- ↑ 3.0 3.1 "Dynamic alterations in the paternal epigenetic landscape following fertilization" (in English). Frontiers in Genetics 3: 143. 2012. doi:10.3389/fgene.2012.00143. PMID 23024648.
- ↑ "Protamine-induced condensation and decondensation of the same DNA molecule". Science 286 (5437): 120–3. October 1999. doi:10.1126/science.286.5437.120. PMID 10506559. https://zenodo.org/record/1231163.
- ↑ "Optimizing Tethered Particle Motion to Measure DNA Compaction by Protamine". Biophysical Journal 108 (2): 393a. January 2015. doi:10.1016/j.bpj.2014.11.2156. Bibcode: 2015BpJ...108..393W.
- ↑ "Sexual selection on protamine and transition nuclear protein expression in mouse species". Proceedings. Biological Sciences 281 (1783). May 2014. doi:10.1098/rspb.2013.3359. PMID 24671975.
- ↑ "Rapid evolution of male reproductive genes in the descent of man". Nature 403 (6767): 304–9. January 2000. doi:10.1038/35002070. PMID 10659848. Bibcode: 2000Natur.403..304W.
- ↑ "Insulin preparations with prolonged effect". Diabetes Technology & Therapeutics 13 (Suppl 1): S5-14. June 2011. doi:10.1089/dia.2011.0068. PMID 21668337.
- ↑ "The heparin-protamine interaction. A review". The Journal of Cardiovascular Surgery 40 (5): 659–66. October 1999. PMID 10596998.
- ↑ "Serious adverse reactions to protamine sulfate: are alternatives needed?". The Journal of Allergy and Clinical Immunology 75 (2): 297–303. February 1985. doi:10.1016/0091-6749(85)90061-2. PMID 2857186.
- ↑ "Low molecular weight protamine: a potent but nontoxic antagonist to heparin/low molecular weight protamine". ASAIO Journal 46 (4): 435–9. 2000. doi:10.1097/00002480-200007000-00013. PMID 10926141.
- ↑ "Low molecular weight protamine (LMWP): a nontoxic protamine substitute and an effective cell-penetrating peptide". Journal of Controlled Release 193: 63–73. November 2014. doi:10.1016/j.jconrel.2014.05.056. PMID 24943246.
- ↑ "Low molecular weight protamine (LMWP) as nontoxic heparin/low molecular weight heparin antidote (I): preparation and characterization". AAPS PharmSci 3 (3): 7–14. 1 September 2001. doi:10.1208/ps030317. PMID 11741268.
- ↑ "Protamine sulfate as an effective alternative to polybrene in retroviral-mediated gene-transfer: implications for human gene therapy". Journal of Virological Methods 23 (2): 187–94. February 1989. doi:10.1016/0166-0934(89)90132-8. PMID 2786000. https://zenodo.org/record/1258363.
- ↑ "Protamine sulfate enhances lipid-mediated gene transfer". Gene Therapy 4 (9): 961–8. September 1997. doi:10.1038/sj.gt.3300484. PMID 9349433.
- ↑ "Effect of protamine in obesity induced by high-fat diets in rats". International Journal of Obesity 33 (6): 687–92. June 2009. doi:10.1038/ijo.2009.78. PMID 19434066.
- ↑ "The lack of protamine 2 (P2) in boar and bull spermatozoa is due to mutations within the P2 gene". Nucleic Acids Research 18 (5): 1249–54. March 1990. doi:10.1093/nar/18.5.1249. PMID 2320417.
- ↑ "Expression of the rat protamine 2 gene is suppressed at the level of transcription and translation". Experimental Cell Research 188 (1): 147–52. May 1990. doi:10.1016/0014-4827(90)90290-q. PMID 2328773.
- ↑ "Conservation of the PRM1 → PRM2 → TNP2 domain". DNA Sequence 14 (5): 359–67. October 2003. doi:10.1080/10425170310001599453. PMID 14756422.
- ↑ 20.0 20.1 20.2 20.3 "Entropy based analysis of vertebrate sperm protamines sequences: evidence of potential dityrosine and cysteine-tyrosine cross-linking in sperm protamines". BMC Genomics 21 (1): 277. April 2020. doi:10.1186/s12864-020-6681-2. PMID 32245406.
- ↑ "The protamine family of sperm nuclear proteins". Genome Biology 8 (9): 227. 2007-09-26. doi:10.1186/gb-2007-8-9-227. PMID 17903313.
- ↑ "alpha-Helix-double helix interaction shown in the structure of a protamine-transfer RNA complex and a nucleoprotamine model". Nature 271 (5641): 130–5. January 1978. doi:10.1038/271130a0. PMID 622153. Bibcode: 1978Natur.271..130W.
- ↑ 23.0 23.1 "Evidence of novel secondary structure in DNA-bound protamine is revealed by Raman spectroscopy". Biochemistry 33 (24): 7528–35. June 1994. doi:10.1021/bi00190a005. PMID 8011618.
- ↑ "Nuclear matrix interactions at the human protamine domain: a working model of potentiation". The Journal of Biological Chemistry 279 (50): 51862–8. December 2004. doi:10.1074/jbc.M409415200. PMID 15452126.
- ↑ 25.0 25.1 "Formation of native-like mammalian sperm cell chromatin with folded bull protamine". The Journal of Biological Chemistry 279 (19): 20088–95. May 2004. doi:10.1074/jbc.M312777200. PMID 14990583.
- ↑ 26.0 26.1 "Protamine loops DNA in multiple steps". Nucleic Acids Research 48 (11): 6108–6119. June 2020. doi:10.1093/NAR/GKAA365. PMID 32392345.
- ↑ van der Harten, Florian J. F.; Sheikhhassani, Vahid; Mashaghi, Alireza (22 September 2025). "Protamine Drives Liquid and Solid Condensation of DNA and Glycosaminoglycans". Langmuir. doi:10.1021/acs.langmuir.5c01954.
External links
- Protamines at the US National Library of Medicine Medical Subject Headings (MeSH)
- V03AB14 (WHO)
 |
