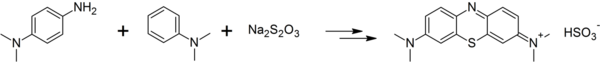Chemistry:Methylene blue
 Molecular structure | |
 A volumetric flask of a methylene blue solution | |
| Clinical data | |
|---|---|
| Trade names | Urelene blue, Provayblue, Proveblue, others[1][2] |
| Other names | CI 52015, basic blue 9[3] |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 5–24 hours (IV)[5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C16H18ClN3S |
| Molar mass | 319.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Methylthioninium chloride, commonly called methylene blue, is a salt used as a dye and as a medication. As a medication, it is mainly used to treat methemoglobinemia. It has previously been used for treating cyanide poisoning and urinary tract infections, but this use is no longer recommended.[5] It has also been used to treat cases of malaria for over a century.[8]
Methylene blue is typically given by injection into a vein.[5] Common side effects include headache, nausea, and vomiting.
Methylene blue was first prepared in 1876, by Heinrich Caro.[9] It is on the World Health Organization's List of Essential Medicines.[10]
In recent years, methylene blue has been promoted for various health effects, but outside its established medical use, its safety and effectiveness are unproven; it is potentially toxic and should only be used under a doctor's prescription.[11]
Medical uses
Methemoglobinemia
Methylene blue is used to treat methemoglobinemia by chemically reducing the ferric iron in hemoglobin to ferrous iron.[5][2] Methemoglobinemia can arise from ingestion of certain pharmaceuticals, toxins, or broad beans in those susceptible.[12] Specifically, it is used to treat methemoglobin levels that are greater than 30% or in which there are symptoms despite oxygen therapy.[2] Normally, through the NADH- or NADPH-dependent methemoglobin reductase enzymes, methemoglobin is reduced back to hemoglobin. When large amounts of methemoglobin occur secondary to toxins, methemoglobin reductases are overwhelmed. Methylene blue, when injected intravenously as an antidote, is itself first reduced to leucomethylene blue, which then reduces the heme group from methemoglobin to hemoglobin. Methylene blue can reduce the half-life of methemoglobin from hours to minutes.[13] At high doses, however, methylene blue actually induces methemoglobinemia, reversing this pathway.[13]
Isobutyl nitrite toxicity
Isobutyl nitrite is one of the compounds used as poppers, an inhalant drug that induces a brief euphoria.
Isobutyl nitrite is known to cause methemoglobinemia.[14] Severe methemoglobinemia may be treated with methylene blue.[15]
In a combination drug: Methylphen
Cyanide poisoning
Since its reduction potential is similar to that of oxygen and can be reduced by components of the electron transport chain, large doses of methylene blue are sometimes used as an antidote for cyanide poisoning, a method first successfully tested in 1933 by Matilda Moldenhauer Brooks in San Francisco,[16] although first demonstrated by Bo Sahlin of Lund University, in 1926.[16][17]
Shock
Methylene blue increases blood pressure in people with vasoplegic syndrome (redistributive shock). It does not improve delivery of oxygen to tissues or decrease mortality.[18][19]
Methylene blue has been used in calcium channel blocker toxicity as a possible rescue therapy for distributive shock unresponsive to first-line agents. Limited to case reports, a 2024 review found low-quality evidence that methylene blue may reduce short-term mortality, duration of the need for vasopressors, and length of hospital stay.[20]
Dye or stain


Methylene blue is used in endoscopic polypectomy as an adjunct to saline or epinephrine, and is used for injection into the submucosa around the polyp to be removed. This allows the submucosal tissue plane to be identified after the polyp is removed, which is useful in determining if more tissue needs to be removed or if there is a high risk for perforation. Methylene blue is also used as a dye in chromoendoscopy, and is sprayed onto the mucosa of the gastrointestinal tract to identify dysplasia, or pre-cancerous lesions. Intravenously injected methylene blue is readily released into the urine.[21]
It is sometimes used in cytopathology, in mixtures including Wright-Giemsa and Diff-Quik. It confers a blue color to both nuclei and cytoplasm, and makes the nuclei more visible.[22] When methylene blue is "polychromed" (oxidized in solution or "ripened" by fungal metabolism,[23] as originally noted in the thesis of Dr. D. L. Romanowsky in the 1890s), it gets serially demethylated and forms all the tri-, di-, mono- and non-methyl intermediates, which are Azure B, Azure A, Azure C, and thionine, respectively.[24] This is the basis of the basophilic part of the spectrum of Romanowski-Giemsa effect. If only synthetic Azure B and Eosin Y is used, it may serve as a standardized Giemsa stain; but, without methylene blue, the normal neutrophilic granules tend to overstain and look like toxic granules. On the other hand, if methylene blue is used it might help to give the normal look of neutrophil granules and may also enhance the staining of nucleoli and polychromatophilic RBCs (reticulocytes).[25]
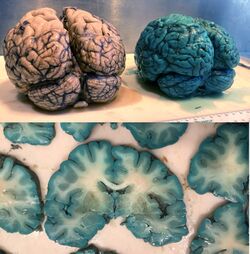
A traditional application of methylene blue is the intravital or supravital staining of nerve fibers, an effect first described by Paul Ehrlich in 1887.[26] A dilute solution of the dye is either injected into tissue or applied to small freshly removed pieces. The selective blue coloration develops with exposure to air (oxygen) and can be fixed by immersion of the stained specimen in an aqueous solution of ammonium molybdate. Vital methylene blue was formerly much used for examining the innervation of muscle, skin, and internal organs.[27][28][29] The mechanism of selective dye uptake is incompletely understood; vital staining of nerve fibers in skin is prevented by ouabain, a drug that inhibits the Na/K-ATPase of cell membranes.[30]
Placebo
Methylene blue has been used as a placebo; physicians would tell their patients to expect their urine to change color and view this as a sign that their condition had improved.[31] This same side effect makes methylene blue difficult to use in traditional placebo-controlled clinical studies, including those testing for its efficacy as a treatment. One approach is to use a low dose, just enough to turn urine blue, as the placebo group.[32] However, a low dose does not guarantee inertness.[33]
Side effects
| Cardiovascular[34][35] | Central nervous system[34][35] | Dermatologic[34][35] | Gastrointestinal[34][35] | Genito-urinary[34][35] | Hematologic[34][35] |
|---|---|---|---|---|---|
|
|
Methylene blue is a monoamine oxidase inhibitor (MAOI)[36] and, if infused intravenously at doses exceeding 5 mg/kg, may result in serotonin syndrome if combined with any selective serotonin reuptake inhibitors (SSRIs) or other serotonergic drugs (e.g., duloxetine, sibutramine, venlafaxine, clomipramine, imipramine).[37]
It causes hemolytic anemia in carriers of the G6PD enzymatic deficiency (favism).[38] The actual degree of this danger is a subject of controversy as the association was made based on very few cases. A 2018 meta-analysis on clinical trials against malaria in Africa, where the moderate A minus type of G6PD deficiency is prevalent, shows no association between MB and hemolysis in such patients. There was, however, a clinically insignificant reduction in hemoglobin.[39]
Pregnancy
While use during pregnancy may harm the fetus, not using it in methemoglobinemia is likely more dangerous.[5][2]
Pharmacokinetics
After intravenous administration in humans, methylene blue shows a multiphasic change in concentration, with a terminal half-life of 5.25 hours. The initial disappearance from blood actually reflects its movement into organs, with brain, liver, and bile all showing significantly higher concentrations than blood in rats. The overall area under the curve in oral (dry gelatin capsule) administration is only 6.5% of the AUC for IV administration; judging from rat studies, the significantly altered organ distribution plays a key role in this difference.[40]
Administration as an oral solution (500 mg in 200 mL) greatly increases the bioavailability to 72.3±23.9%. In this newer study, the terminal half-lives were reported as 18.5±11.8 hours for IV use and 18.3±7.2 hours for oral use. The tmax for oral use is 2.2 hours, compared to 0.5 hours for iv use.[41]
Chemistry
Methylene blue is a formal derivative of phenothiazine. It is a dark green powder that yields a blue solution in water. The hydrated form has 3 molecules of water per unit of methylene blue.
Preparation
This compound is prepared by oxidation of 4-aminodimethylaniline in the presence of sodium thiosulfate to give the quinonediiminothiosulfonic acid, reaction with dimethylaniline, oxidation to the indamine, and cyclization to give the thiazine:[42]
A green electrochemical procedure, using only dimethyl-4-phenylenediamine and sulfide ions has been proposed.[43]
Light absorption properties

The maximum absorption of light is near 670 nm. The specifics of absorption depend on several factors, including protonation, adsorption to other materials, and metachromasy – the formation of dimers and higher-order aggregates depending on concentration and other interactions:[44]
| Species | Absorption peak | Extinction coefficient (dm3/mol·cm) |
|---|---|---|
| MB+ (solution) | 664 | 95000 |
| MBH2+ (solution) | 741 | 76000 |
| (MB+)2 (solution) | 605 | 132000 |
| (MB+)3 (solution) | 580 | 110000 |
| MB+ (adsorbed on clay) | 673 | 116000 |
| MBH2+ (adsorbed on clay) | 763 | 86000 |
| (MB+)2 (adsorbed on clay) | 596 | 80000 |
| (MB+)3 (adsorbed on clay) | 570 | 114000 |
Redox properties

Under reducing conditions, the blue-colored methylene blue cation (MB+) gains 1H+ and 2e− to become the electrically neutral and colorless leucomethylene blue (LMB).[45] The redox midpoint potential E0' is +0.01 V.[46]
The redox properties can be seen in a classical demonstration of chemical kinetics in general chemistry, the "blue bottle" experiment. Typically, a solution is made of glucose (dextrose), methylene blue, and sodium hydroxide. Upon shaking the bottle, oxygen oxidizes methylene blue, and the solution turns blue. The dextrose will gradually reduce the methylene blue to its colorless, reduced form. Hence, when the dissolved dextrose is entirely consumed, the solution will turn blue again.[47]
In the mitochondrial electron transport chain, reduced methylene blue (MBH2) directly reduces cytochrome c rather than to oxygen, limiting the formation of superoxide.[48][49] Methylene blue has been shown to directly accept electrons from NADH, NADPH, and FADH2.[49]
Other uses
Redox indicator
Methylene blue is widely used as a redox indicator in analytical chemistry.[47][50][51] Solutions of this substance are blue when in an oxidizing environment, but will turn colorless if exposed to a reducing agent.[52][50]
Photosensitizer
With the help of light, methylene blue can be used to kill some viruses and some bacteria.[53] This kind of photo-disinfection has also been done inside of human bodies (antimicrobial photodynamic therapy).[54] The same process can also be used to disinfect blood plasma.[55]
Methylene blue is theoretically also applicable to other forms of photodynamic therapy, i.e., the use of oxygen, light, and a photosensitizer to kill cells. Research on using it to kill cancer cells locally is in a preclinical stage.[56] Its cytotoxicity may be related to its ability to inhibit tubulin polymerization.[57]
Sulfide analysis
The formation of methylene blue after the reaction of hydrogen sulfide with dimethyl-p-phenylenediamine and iron(III) at pH 0.4 – 0.7 is used to determine by photometric measurements sulfide concentration in the range 0.020 to 1.50 mg/L (20 ppb to 1.5 ppm).[58] The test is very sensitive and the blue coloration developing upon contact of the reagents with dissolved H2S is stable for 60 min. Ready-to-use kits such as the Spectroquant sulfide test[59] facilitate routine analyses. The methylene blue sulfide test is a convenient method often used in soil microbiology to quickly detect in water the metabolic activity of sulfate reducing bacteria (SRB). In this colorimetric test, methylene blue is a product formed by the reaction and not a reagent added to the system.[58]
The addition of a strong reducing agent, such as ascorbic acid, to a sulfide-containing solution is sometimes used to prevent sulfide oxidation from atmospheric oxygen. Although it is certainly a sound precaution for the determination of sulfide with an ion selective electrode, it might however hamper the development of the blue color if the freshly formed methylene blue is also reduced, as described here above in the paragraph on redox indicator.[50]
Test for milk freshness
Methylene blue is a dye behaving as a redox indicator that is commonly used in the food industry to test the freshness of milk and dairy products.[5][60] A few drops of methylene blue solution added to a sample of milk should remain blue (oxidized form in the presence of enough dissolved O2), otherwise (discoloration caused by the reduction of methylene blue into its colorless reduced form) the dissolved O2 concentration in the milk sample is low indicating that the milk is not fresh (already abiotically oxidized by O2 whose concentration in solution decreases) or could be contaminated by bacteria also consuming the atmospheric O2 dissolved in the milk.[60] In other words, aerobic conditions should prevail in fresh milk, and methylene blue is simply used as an indicator of the dissolved oxygen remaining in the milk.[52]
Water testing
The adsorption of methylene blue serves as an indicator defining the adsorptive capacity of granular activated carbon in water filters. Adsorption of methylene blue is very similar to adsorption of pesticides from water; this quality makes methylene blue serve as a good predictor for filtration qualities of carbon. It is also a quick method of comparing different batches of activated carbon of the same quality. A color reaction in an acidified, aqueous methylene blue solution containing chloroform can detect anionic surfactants in a water sample. Such a test is known as an MBAS assay (methylene blue active substances assay).
Methylene blue value of fine aggregate
The methylene blue value is defined as the number of milliliters of standard methylene blue solution decolorized 0.1 g of activated carbon (dry basis).[61] Methylene blue value reflects the amount of clay minerals in aggregate samples.[62] In materials science, methylene blue solution is successively added to fine aggregate which is being agitated in water. The presence of free dye solution can be checked with a stain test on a filter paper.[63]
Biological staining
Aquaculture
- Methylene blue is used in aquaculture and by tropical fish hobbyists as a treatment for fungal infections.[64] With the help of light, it is also effective against bacteria and viruses.
- It can also be effective in treating fish infected with the parasitic protozoa Ichthyophthirius multifiliis (ich), although a combination of malachite green and formaldehyde is far more effective against it.[64]
- Methylene blue also works against nitrite poisoning as it treats the resulting methemoglobinemia. Like in humans, it also treats cyanide poisoning.
- Non-professional sources also claim that it works for ammonia poisoning,[65] but there is little medical literature to back this up.
It is usually used to protect newly laid fish eggs from being infected by fungus. This is useful when the hobbyist wants to artificially hatch the fish eggs.[64] For poisoning, injury (prevention of infection), or sickness, methylene blue is given as a "medicated bath" for the fish.
Methylene blue is not without side effects to fish.[66]
History
Methylene blue has been described as "the first fully synthetic drug used in medicine". Methylene blue was first prepared in 1876 by German chemist Heinrich Caro.[67]
It was discovered to be an antidote to carbon monoxide poisoning and cyanide poisoning in 1933 by Matilda Brooks.[68]
Methylene blue was the original prototype or lead compound for the design of many antimalarials including chloroquine, antihistamines, and antipsychotics including chlorpromazine.[69]
Research
Malaria
Antimalarial use of the drug has recently (2009) been revived.[70] It simultaneously targets many biological processes in the apicomplexan pathogen[71] though the main mechanism seems to be causing a lethal amount of redox cycling.[39]
A 2018 meta-analysis finds that it has proven effective against P. falciparum in Africa. It effectively reduces levels of the transmission-stage gametocyte and has synergy with the standard artemisinin-based combination therapy (ACT). Its effects against other malarial species and P. falciparum populations in other locations are unclear.[39]
Ifosfamide toxicity
Another use of methylene blue is to treat ifosfamide neurotoxicity. Methylene blue was first reported for treatment and prophylaxis of ifosfamide neuropsychiatric toxicity in 1994. A toxic metabolite of ifosfamide, chloroacetaldehyde (CAA), disrupts the mitochondrial respiratory chain, leading to an accumulation of nicotinamide adenine dinucleotide hydrogen (NADH). Methylene blue acts as an alternative electron acceptor, and reverses the NADH inhibition of hepatic gluconeogenesis while also inhibiting the transformation of chloroethylamine into chloroacetaldehyde, and inhibits multiple amine oxidase activities, preventing the formation of CAA.[72][73]
The dosing of methylene blue for treatment of ifosfamide neurotoxicity varies, depending upon its use simultaneously as an adjuvant in ifosfamide infusion, versus its use to reverse psychiatric symptoms that manifest after completion of an ifosfamide infusion. Reports suggest that methylene blue, up to six doses a day, has resulted in improvement of symptoms within 10 minutes to several days.[74] Alternatively, it has been suggested that intravenous methylene blue every six hours for prophylaxis during ifosfamide treatment in people with history of ifosfamide neuropsychiatric toxicity.[75] Prophylactic administration of methylene blue the day before initiation of ifosfamide, and three times daily during ifosfamide chemotherapy has been recommended to lower the occurrence of ifosfamide neurotoxicity.[76]
Neuropsychiatric disorders
Methylene blue inhibits monoamine oxidase, inhibits the glutamatergic system (via inhibition of NO synthase and soluble guanylate cyclase), modulates mitochondrial function (by acting as an electron acceptor), and decreases the activation of inflammasomes NLRP3 and NLRC4. As a result, it's been considered potentially useful in neuropsychiatric disorders. In humans it has been tried for (listed in decreasing order of evidence quality): bipolar disorder (especially depressive symptoms), Alzheimer's disease, claustrophobia, ifosfamide encephalopathy, and schizophrenia. With methylene blue, a higher dose does not necessarily work better than a lower dose.[33]
Society and culture
In the late 2010s and early 2020s, a social media trend emerged promoting the use of methylene blue for various medical purposes, including anti-aging, metabolism enhancement, cognitive improvement, cancer treatment, and COVID-19 treatment.[77][11][78][79] Currently there is no scientific consensus on, and no FDA approval for, its effectiveness and safety for these purposes.[77][11][79] Medical experts cautioned that methylene blue can be toxic in high doses and may interact with other medications, potentially reducing their effectiveness or causing unforeseen side effects. Therefore, it should only be used under a doctor's prescription.[77][11]
This trend probably started following the publication of a few scientific papers exploring the potential of methylene blue for treating some medical conditions,[77] such as progeria,[80] and skin aging.[81] It was also explored as part of anticancer photodynamic therapy using lasers.[82] One systematic review of the studies expresses optimism but emphasizes the need for more extensive research to confirm methylene blue's clinical applications.[83] Another review takes a more critical stance, stating that "it is obvious that the clinical use of [methylene blue] represents a rather controversial problem given the heterogeneity of available data and the lack of preclinical data, which is in conflict with standards of safe use of such substances in human medicinal practice".[84]
In January 2025, Robert F. Kennedy Jr, then the U.S. health secretary nominee, was filmed adding droplets of an unidentified blue liquid to his drink during a flight.[85] While many have speculated that it was methylene blue, Kennedy has not addressed the claims.[78][86][87]
References
- ↑ Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. 2015. p. 471. ISBN 978-1-284-05756-0.
- ↑ 2.0 2.1 2.2 2.3 British national formulary: BNF 69 (69 ed.). British Medical Association. 2015. p. 34. ISBN 978-0-85711-156-2.
- ↑ H. J. Conn's Biological stains (9th ed.). Baltimore: Williams & Wilkins. 1977. pp. 692p.
- ↑ "Provayblue- methylene blue injection". 29 June 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=4f6848e5-35ed-4046-b13c-3032b5ba3232.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 "Methylene Blue". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/methylene-blue.html.
- ↑ "Lumeblue EPAR". 19 June 2020. https://www.ema.europa.eu/en/medicines/human/EPAR/lumeblue.
- ↑ "Lumeblue Product information". https://ec.europa.eu/health/documents/community-register/html/h1470.htm.
- ↑ MPH, Leslie Goldman (2025-04-24). "What to know about methylene blue" (in en). https://www.health.harvard.edu/diseases-and-conditions/what-to-know-about-methylene-blue.
- ↑ (in en) New Strategies Combating Bacterial Infection. John Wiley & Sons. 2008. p. 91. ISBN 978-3-527-62294-8. https://books.google.com/books?id=Hrdzorisyp8C&pg=PA91.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 11.0 11.1 11.2 11.3 "When Medicines Go Rogue, Part 1: Methylene Blue" (in en-US). 2022-04-20. https://skepticalinquirer.org/exclusive/when-medicines-go-rogue-methylene-blue/.
- ↑ "Hemolytic Anemia: Hereditary and Acquired". Manual of Clinical Hematology (3rd ed.). Philadelphia: Lippincott Williams & Wilkins. 2002. p. 102. ISBN 978-0-7817-2980-2. https://books.google.com/books?id=NzMKMPzbxZwC&pg=PA102.
- ↑ 13.0 13.1 Critical care toxicology: diagnosis and management of the critically poisoned patient.. Elsevier Health Sciences. 2005.
- ↑ "Severe methemoglobinemia secondary to isobutyl nitrite toxicity: the case of the 'Gold Rush'". Oxford Medical Case Reports 2021 (2). February 2021. doi:10.1093/omcr/omaa136. PMID 33614047.
- ↑ "Methylene blue: a treatment for severe methaemoglobinaemia secondary to misuse of amyl nitrite". Emergency Medicine Journal 19 (3): 270–271. May 2002. doi:10.1136/emj.19.3.270. PMID 11971852.
- ↑ 16.0 16.1 "Methylene blue as an antidote for cyanide and carbon monoxide poisoning". The Scientific Monthly 43 (6): 585–586. 1936. Bibcode: 1936SciMo..43..585M.
- ↑ "Methylene Blue As Antidote for Cyanide Poisoning". JAMA 100 (5): 357. 4 February 1933. doi:10.1001/jama.1933.02740050053028.
- ↑ "Methylene Blue: Magic Bullet for Vasoplegia?". Anesthesia and Analgesia 122 (1): 194–201. January 2016. doi:10.1213/ANE.0000000000001045. PMID 26678471.
- ↑ "Methylene blue and vasoplegia: who, when, and how?". Mini Reviews in Medicinal Chemistry 8 (5): 472–490. May 2008. doi:10.2174/138955708784223477. PMID 18473936. http://www.bentham-direct.org/pages/content.php?MRMC/2008/00000008/00000005/0006N.SGM. Retrieved 2020-04-01.
- ↑ "Methylene Blue in Septic Shock: A Systematic Review and Meta-Analysis". Critical Care Explorations 6 (7). July 2024. doi:10.1097/CCE.0000000000001110. PMID 38904978.
- ↑ "Efficacy and safety of methylene blue in the treatment of malaria: a systematic review". BMC Medicine 16 (1). April 2018. doi:10.1186/s12916-018-1045-3. PMID 29690878.
- ↑ "A Simple and Efficient Method for Preparing Cell Slides and Staining without Using Cytocentrifuge and Cytoclips". International Journal of Cell Biology 2015. 2015. doi:10.1155/2015/813216. PMID 26664363.
- ↑ "Chapter 19: On Chemical Reactions and Staining Mechanisms". Special Stains and H & E. Education Guide (Second ed.). Carpinteria, California: Dako North America, Inc.. 2010. p. 172. http://www.dako.com/08066_12may10_webchapter19.pdf. "What is Giemsa's stain and how does it color blood cells, bacteria and chromosomes?"
- ↑ "On the Chemistry and Staining Properties of Certain Derivatives of the Methylene Blue Group When Combined With Eosin". The Journal of Experimental Medicine 9 (6): 645–670. November 1907. doi:10.1084/jem.9.6.645. PMID 19867116.
- ↑ Dacie and Lewis practical haematology (10th ed.). Philadelphia, PA: Churchill Livingstone/Elsevier. 2006. p. 61. ISBN 978-0-443-06660-3.
- ↑ "Ueber die Methylenblau Reaktion der lebenden Nerven Substanz." (in German). Biologisches Zentralblatt 6: 214–224. 1887. https://www.pei.de/SharedDocs/Downloads/DE/institut/veroeffentlichungen-von-paul-ehrlich/1886-1896/1887-ueber-methylenblaureaktion-lebender-nervensubstanz.pdf?__blob=publicationFile&v=2. cited by Principles of biological microtechnique. A study of fixation and dyeing.. London: Methuen. 1958.
- ↑ "Intra vitam staining with methylene blue.". The Anatomical Record 4 (7): 267–277. July 1910. doi:10.1002/ar.1090040705. https://zenodo.org/record/2478243.
- ↑ "Untersuchungen zur Methodik der Methylenblaufärbung des vegetativen Nervensystems." (in German). Zeitschrift für Zellforschung und Mikroskopische Anatomie 10 (2): 221–243. January 1930. doi:10.1007/BF02450696.
- ↑ The Motor Endplate (2nd ed.). Huntington, NY: Krieger. 1973.
- ↑ "Effects of metabolic inhibitors on vital staining with methylene blue". Histochemistry 40 (1): 51–57. June 1974. doi:10.1007/BF00490273. PMID 4136702.
- ↑ "The ethics of deception in medicine". Science Based Medicine. 23 January 2008. http://www.sciencebasedmedicine.org/?p=29.
- ↑ Clinical trial number NCT00214877 for "Methylene blue for cognitive dysfunction in bipolar disorder" at ClinicalTrials.gov
- ↑ 33.0 33.1 "Methylene Blue in the Treatment of Neuropsychiatric Disorders". CNS Drugs 33 (8): 719–725. August 2019. doi:10.1007/s40263-019-00641-3. PMID 31144270.
- ↑ 34.0 34.1 34.2 34.3 34.4 34.5 "Adult toxicology in critical care: Part II: specific poisonings". Chest 123 (3): 897–922. March 2003. doi:10.1378/chest.123.3.897. PMID 12628894.
- ↑ 35.0 35.1 35.2 35.3 35.4 35.5 "Studies of the efficacy and potential hazards of methylene blue therapy in aniline-induced methaemoglobinaemia". British Journal of Haematology 54 (1): 29–41. May 1983. doi:10.1111/j.1365-2141.1983.tb02064.x. PMID 6849836.
- ↑ "Methylene blue and serotonin toxicity: inhibition of monoamine oxidase A (MAO A) confirms a theoretical prediction". British Journal of Pharmacology 152 (6): 946–951. November 2007. doi:10.1038/sj.bjp.0707430. PMID 17721552.
- ↑ "Methylene blue implicated in potentially fatal serotonin toxicity". Anaesthesia 61 (10): 1013–1014. October 2006. doi:10.1111/j.1365-2044.2006.04808.x. PMID 16978328.
- ↑ "Failure of methylene blue treatment in toxic methemoglobinemia. Association with glucose-6-phosphate dehydrogenase deficiency". Annals of Internal Medicine 75 (1): 83–86. July 1971. doi:10.7326/0003-4819-75-1-83. PMID 5091568.
- ↑ 39.0 39.1 39.2 "Efficacy and safety of methylene blue in the treatment of malaria: a systematic review". BMC Medicine 16 (1). April 2018. doi:10.1186/s12916-018-1045-3. PMID 29690878.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedpmid10952480 - ↑ "High absolute bioavailability of methylene blue given as an aqueous oral formulation". European Journal of Clinical Pharmacology 65 (2): 179–189. February 2009. doi:10.1007/s00228-008-0563-x. PMID 18810398. https://hal.archives-ouvertes.fr/hal-00477926/file/PEER_stage2_10.1007%252Fs00228-008-0563-x.pdf.
- ↑ Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2012. doi:10.1002/14356007.a03_213.pub3. ISBN 978-3-527-30673-2.
- ↑ "An efficient electrochemical method for the synthesis of methylene blue" (in en). Electrochemistry Communications 11 (12): 2261–2264. December 2009. doi:10.1016/j.elecom.2009.09.040. https://linkinghub.elsevier.com/retrieve/pii/S1388248109004846.
- ↑ "Visible spectroscopy of methylene blue on hectorite, laponite B, and barasym in aqueous suspension". Clays and Clay Minerals 36 (3): 214–224. 1988. doi:10.1346/ccmn.1988.0360302. Bibcode: 1988CCM....36..214C.
- ↑ "Methylene Blue". IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 108. 2016. pp. 155–183 (166). https://publications.iarc.fr/_publications/media/download/6707/581359b111699ead293649c15000080016dce9f3.pdf.
- ↑ "Chapter 8: Oxidation/Phosphorylation B: Oxidative Enzymes". Biochemistry Online. 2016. https://employees.csbsju.edu/hjakubowski/classes/ch331/oxphos/oloxidativeenzymes.html.
- ↑ 47.0 47.1 "The Blue Bottle experiment revisited: How Blue? How Sweet?". Journal of Chemical Education 71 (2): 160. February 1994. doi:10.1021/ed071p160. Bibcode: 1994JChEd..71..160C.
- ↑ "Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways". The FASEB Journal 22 (3): 703–712. 2008. doi:10.1096/fj.07-9610com. PMID 17928358. https://d1wqtxts1xzle7.cloudfront.net/41715377/fj.07-9610com.full-libre.pdf?1454057116=&response-content-disposition=inline%3B+filename%3DMethylene_blue_delays_cellular_senescenc.pdf&Expires=1743199680&Signature=ItIIdTkGebvWdx5Xp7QM4EAb1FWD6ytz5jmUrmR6r25dQwDj53ZGRMgXjUvkX7xhMJCwYSS8LNqQ0TT5oiLJWgvdQyEAiyMoTkdCLInanRGNP6xW5GhYlLWfwIwILUbKZyKOMF-fjc-PTTtjvZNgyiR3yJIhxLBOfooWCg9i10w4-6qZu5oTulqcwUQmBbSBzdtETJBxRemlydYjJDSMqnpzOUPDfdJsF1OhJJ0kTbWJ3nh18RQvH68~9InEZsCMgP5EYTd64K5dF~ss-uYwnDoDTTDIdr48nWxYo1hfprrS9aLymKlk6OpPW1IZ9Pk4oDS4xg1TPCQ4zK7InhD0RQ__&Key-Pair-Id=APKAJLOHF5GGSLRBV4ZA.
- ↑ 49.0 49.1 "Neuroprotective actions of methylene blue and its derivatives". PLOS One 7 (10). 2012. doi:10.1371/journal.pone.0048279. PMID 23118969. Bibcode: 2012PLoSO...748279P.
- ↑ 50.0 50.1 50.2 "What is happening when the Blue Bottle bleaches: An Investigation of the methylene blue-catalyzed air oxidation of glucose". Journal of Chemical Education 89 (11): 1425–1431. 9 October 2012. doi:10.1021/ed200511d. Bibcode: 2012JChEd..89.1425A.
- ↑ "Rapid Blue Bottle experiment: Autoxidation of benzoin catalyzed by redox indicators". Journal of Chemical Education 93 (8): 1490–1494. 9 August 2016. doi:10.1021/acs.jchemed.6b00018. Bibcode: 2016JChEd..93.1490R.
- ↑ 52.0 52.1 "The relation between the oxygen concentration and rate of reduction of methylene blue by milk". The Journal of General Physiology 1 (4): 415–419. March 1919. doi:10.1085/jgp.1.4.415. PMID 19871757.
- ↑ "Methylene Blue: The Little-Known Disinfectant" (in en). https://asm.org/articles/2022/march/methylene-blue-the-little-known-disinfectant.
- ↑ "Methylene blue mediated antimicrobial photodynamic therapy in clinical human studies: The state of the art". Photodiagnosis and Photodynamic Therapy 31. September 2020. doi:10.1016/j.pdpdt.2020.101828. PMID 32473398.
- ↑ "Methods of pathogen inactivation in whole blood and red blood cells: current state of knowledge". Acta Haematologica Polonica 52 (4): 406–411. 31 August 2021. doi:10.5603/AHP.2021.0076. https://journals.viamedica.pl/acta_haematologica_polonica/article/download/AHP.2021.0076/63958.
- ↑ "Methylene blue in anticancer photodynamic therapy: systematic review of preclinical studies". Frontiers in Pharmacology 14. 28 September 2023. doi:10.3389/fphar.2023.1264961. PMID 37841915.
- ↑ "NanoDSF Screening for Anti-tubulin Agents Uncovers New Structure–Activity Insights". Journal of Medicinal Chemistry. 2025. doi:10.1021/acs.jmedchem.5c01008.
- ↑ 58.0 58.1 "A direct microdetermination for sulfide". Analytical Biochemistry 11 (1): 126–132. April 1965. doi:10.1016/0003-2697(65)90051-5. PMID 14328633.
- ↑ "Analytik und Probenvorbereitung". Darmstadt, Germany: Merck KGaA. http://photometry.merck.de/.
- ↑ 60.0 60.1 "Studies on Oxidation-Reduction in Milk: The Methylene Blue Reduction Test". Journal of Dairy Science (Elsevier Inc.) 13 (3): 221–245. May 1930. doi:10.3168/jds.S0022-0302(30)93520-5.
- ↑ "Test Methods for Activated Carbon". European Council of Chemical Manufacturers' Federations. April 1986. https://activatedcarbon.org/images/Test_method_for_Activated_Carbon_86.pdf.
- ↑ Standard Test Method for Rapid Determination of the Methylene Blue Value for Fine Aggregate or Mineral Filler Using a Colorimeter (Report). West Conshohocken, PA: ASTM (American Society for Testing and Material) International. 7 July 2020. doi:10.1520/C1777-20. ASTM C1777. http://www.astm.org/Standards/C1777.htm.
- ↑ Standing Committee on Concrete Technology (SCCT) (May 2013). "Construction Standard CS3:2013 – Aggregates for Concrete". The Government of the Hong Kong Special Administrative Region. https://www.cedd.gov.hk/filemanager/eng/content_78/cs3_2013_original.pdf.
- ↑ 64.0 64.1 64.2 "Methylene Blue". 3 Little Fish Sdn Bhd. Kelana Jaya, Selangor, Malaysia. 10 September 2021. https://www.3lfish.com/fish-medicants/#methylene-blue.
- ↑ "Methylene Blue as an Anti-Fungal & Anti-Parasitic Drug for Aquarium Fish" (in en). https://www.thesprucepets.com/methylene-blue-1379926.
- ↑ "Methylene blue at recommended concentrations alters metabolism in early zebrafish development". Communications Biology 8 (1). January 2025. doi:10.1038/s42003-025-07471-8. PMID 39856203.
- ↑ Heinrich Caro was an employee of the Badische Anilin- und Sodafabrik, BASF, of Mannheim, Germany, which received a patent for methylene blue in 1877:
- Badische Anilin- und Sodafabrik, BASF, of Mannheim, Germany, "Verfahren zur Darstellung blauer Farbstoffe aus Dimethylanilin und anderen tertiaren aromatischen Monaminen" [Method for preparation of blue dyes from dimethylaniline and other tertiary aromatic monoamines], Deutsches Reich Patent no. 1886 (issued: December 15, 1877).
- Available on-line at: (in German) Fortschritte der Theerfarbenfabrikation und verwandter Industriezweige. 1. Berlin, Germany: Julius Springer. 1888. pp. 247–249. https://books.google.com/books?id=3akMAAAAYAAJ&pg=PA247. Retrieved 2016-10-12.
- ↑ "Methylene Blue As Antidote for Cyanide and Carbon Monoxide Poisoning". JAMA 100: 59. January 1933. doi:10.1001/jama.1933.02740010061028.
- ↑ "Dyes in the development of drugs and pharmaceuticals". Dyes and Pigments 76 (3): 582–589. January 2008. doi:10.1016/j.dyepig.2007.01.015.
- ↑ "Strong gametocytocidal effect of methylene blue-based combination therapy against falciparum malaria: a randomised controlled trial". PLOS ONE 4 (5). 2009. doi:10.1371/journal.pone.0005318. PMID 19415120. Bibcode: 2009PLoSO...4.5318C.
- ↑ "Methylene blue for treating malaria". Cochrane Database of Systematic Reviews 2022 (6). 20 October 2017. doi:10.1002/14651858.CD012837.
- ↑ "Ifosfamide neuropsychiatric toxicity in patients with cancer". Psycho-Oncology 16 (10): 956–960. October 2007. doi:10.1002/pon.1161. PMID 17278152.
- ↑ "Methylene blue and ifosfamide-induced encephalopathy: Myth or reality?". Journal of Oncology Pharmacy Practice 27 (1): 143–149. January 2021. doi:10.1177/1078155220971843. PMID 33153383.
- ↑ "Methylene blue for management of Ifosfamide-induced encephalopathy". The Annals of Pharmacotherapy 40 (2): 299–303. February 2006. doi:10.1345/aph.1G114. PMID 16391008.
- ↑ "[Ifosfamide induced encephalopathy: 15 observations]" (in fr). Archives de Pédiatrie 13 (2): 140–145. February 2006. doi:10.1016/j.arcped.2005.10.021. PMID 16364615.
- ↑ "Inhibition of (mono)amine oxidase activity and prevention of ifosfamide encephalopathy by methylene blue". Drug Metabolism and Disposition 24 (12): 1336–1339. December 1996. doi:10.1016/S0090-9556(25)08471-5. PMID 8971139.
- ↑ 77.0 77.1 77.2 77.3 "Medical expert issues warning about fish tank disinfectant craze" (in en). 2022-03-18. https://www.the-independent.com/life-style/health-and-families/fish-tank-cleaner-anti-aging-doctor-methylene-blue-b2038679.html.
- ↑ 78.0 78.1 "What was RFK Jr putting in his drink mid-flight?" (in en). 2025-02-06. https://www.independent.co.uk/news/world/americas/us-politics/rfk-jr-drinking-flight-methylene-blue-b2693495.html.
- ↑ 79.0 79.1 "From methylene blue to vitamin E, here's why health and wellness supplements are no silver bullet for cancer" (in en-US). 2025-01-27. https://theconversation.com/from-methylene-blue-to-vitamin-e-heres-why-health-and-wellness-supplements-are-no-silver-bullet-for-cancer-247847.
- ↑ "Methylene blue alleviates nuclear and mitochondrial abnormalities in progeria". Aging Cell 15 (2): 279–290. April 2016. doi:10.1111/acel.12434. PMID 26663466.
- ↑ "Anti-Aging Potentials of Methylene Blue for Human Skin Longevity". Scientific Reports 7 (1). May 2017. doi:10.1038/s41598-017-02419-3. PMID 28559565. Bibcode: 2017NatSR...7.2475X.
- ↑ "Methylene blue in anticancer photodynamic therapy: systematic review of preclinical studies". Frontiers in Pharmacology 14. 2023-09-28. doi:10.3389/fphar.2023.1264961. PMID 37841915.
- ↑ "The Potentials of Methylene Blue as an Anti-Aging Drug". Cells 10 (12): 3379. December 2021. doi:10.3390/cells10123379. PMID 34943887.
- ↑ "Methylene blue: a controversial diagnostic acid and medication?". Toxicology Research 11 (5): 711–717. October 2022. doi:10.1093/toxres/tfac050. PMID 36337249.
- ↑ "The Right Chemistry: TikTok is crazy for methylene blue, but not for the right reasons". https://www.montrealgazette.com/opinion/columnists/article887766.html.
- ↑ "Did RFK Jr really drink fish medicine? He definitely has weird ideas about 'making America healthy again'" (in en-GB). The Guardian. 2025-02-11. ISSN 0261-3077. https://www.theguardian.com/commentisfree/2025/feb/11/did-rfk-jr-really-drink-fish-medicine-definitely-has-weird-ideas-making-america-healthy-again.
- ↑ "Robert F. Kennedy Jr.'s viral video sparks mystery over blue liquid. What Is Methylene Blue?". The Economic Times. 2025-02-06. ISSN 0013-0389. https://economictimes.indiatimes.com/magazines/panache/rfk-jr-s-viral-video-sparks-mystery-over-blue-liquid-what-is-methylene-blue/articleshow/117987350.cms?from=mdr.
External links
| Wikimedia Commons has media related to Methylene blue. |
- "Methylene blue". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/methylthioninium%20chloride.
- "Methylene blue test". https://medlineplus.gov/ency/article/003412.htm.
 |