Chemistry:Flumazenil
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Anexate, Lanexat, Mazicon, Romazicon |
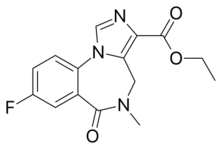
| Other names | ethyl 8-fluoro- 5,6-dihydro- 5-methyl- 6-oxo- 4H- imidazo [1,5-a] [1,4] benzodiazepine- 3-carboxylate |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | IV |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 7–15 min (initial) 20–30 min (brain) 40–80 min (terminal) |
| Excretion | Urine 90–95% Feces 5–10% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C15H14FN3O3 |
| Molar mass | 303.293 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Flumazenil (also known as flumazepil, code name Ro 15-1788[1]) is a selective GABAA receptor antagonist[2] administered via injection, otic insertion, or intranasally. Therapeutically, it acts as both an antagonist and antidote to benzodiazepines (particularly in cases of overdose), through competitive inhibition.
It was first characterized in 1981,[3] and was first marketed in 1987 by Hoffmann-La Roche under the trade name Anexate. However, it did not receive FDA approval until December 20, 1991. The developer lost its exclusive patent rights in 2008; so at present, generic formulations of this drug are available. Intravenous flumazenil is primarily used to treat benzodiazepine overdoses and to help reverse anesthesia. Administration of flumazenil by sublingual lozenge and topical cream has also been tested.[4][5]
Medical uses
Flumazenil benefits patients who become excessively drowsy after use of benzodiazepines for either diagnostic or therapeutic procedures.[6]
The drug has been used as an antidote in the treatment of benzodiazepine overdoses.[6] It reverses the effects of benzodiazepines by competitive inhibition at the benzodiazepine (BZ) recognition site on the GABA/benzodiazepine receptor complex. There are many complications that must be taken into consideration when used in the acute care setting.[6] These include lowered seizure threshold, agitation, and anxiousness. Flumazenil's short half-life requires multiple doses. Because of the potential risks of withdrawal symptoms and the drug's short half-life, patients must be carefully monitored to prevent recurrence of overdose symptoms or adverse side effects.
Flumazenil is also sometimes used after surgery to reverse the sedative effects of benzodiazepines. This is similar to naloxone's application to reverse the effect of opiates and opioids following surgery. Administration of the drug requires careful monitoring by an anesthesiologist due to potential side effects and serious risks associated with over-administeration. Likewise, post-surgical monitoring is also necessary because flumazenil can mask the apparent metabolization ("wearing off") of the drug after removal of patient life-support and monitoring equipment.
Flumazenil has been effectively used to treat overdoses of non-benzodiazepine hypnotics, such as zolpidem, zaleplon and zopiclone (also known as "Z-drugs").[7]
It may also be effective in reducing excessive daytime sleepiness while improving vigilance in primary hypersomnias, such as idiopathic hypersomnia.[4]
The drug has also been used in hepatic encephalopathy. It may have beneficial short‐term effects in people with cirrhosis, but there is no evidence for long-term benefits.[8]
The onset of action is rapid, and effects are usually seen within one to two minutes. The peak effect is seen at six to ten minutes. The recommended dose for adults is 200 μg every 1–2 minutes until the effect is seen, up to a maximum of 3 mg per hour. It is available as a clear, colourless solution for intravenous injection, containing 500 μg in 5 mL.[citation needed] Additional doses may be needed within 20 to 30 min if evidence of oversedation reappears.[9]
Many benzodiazepines (including midazolam) have longer half-lives than flumazenil. Therefore, in cases of overdose, repeated doses of flumazenil may be required to prevent recurrent symptoms once the initial dose of flumazenil wears off.[citation needed]
It is hepatically metabolised to inactive compounds which are excreted in the urine. Individuals who are physically dependent on benzodiazepines may experience benzodiazepine withdrawal symptoms, including seizures, upon rapid administration of flumazenil.
It is not recommended for routine use in those with a decreased level of consciousness.[10]
In terms of drug enforcement initiatives, diversion control programs and required post-marketing surveillance of adverse events, orders for flumazenil may trigger a prescription audit to the search for benzodiazepine misuse and for clinically significant adverse reactions related to their use.[11]
PET radioligand
Radiolabeled with the radioactive isotope carbon-11, flumazenil may be used as a radioligand in neuroimaging with positron emission tomography to visualize the distribution of GABAA receptors in the human brain.[12]
Treatment for benzodiazepine dependence & tolerance
Epileptic patients who have become tolerant to the anti-seizure effects of the benzodiazepine clonazepam became seizure-free for several days after treatment with 1.5 mg of flumazenil.[13] Similarly, patients who were dependent on high doses of benzodiazepines (median dosage 333 mg diazepam-equivalent) were able to be stabilised on a low dose of clonazepam after 7–8 days of treatment with flumazenil.[14]
Flumazenil has been tested against placebo in benzo-dependent subjects. Results showed that typical benzodiazepine withdrawal effects were reversed with few to no symptoms.[15] Flumazenil was also shown to produce significantly fewer withdrawal symptoms than saline in a randomized, placebo-controlled study with benzodiazepine-dependent subjects. Additionally, relapse rates were much lower during subsequent follow-up.[16]
In vitro studies of tissue cultured cell lines have shown that chronic treatment with flumazenil enhanced the benzodiazepine binding site where such receptors have become more numerous and uncoupling/down-regulation of GABAA has been reversed.[17][18][19] After long-term exposure to benzodiazepines, GABAA receptors become down-regulated and uncoupled. Growth of new receptors and recoupling after prolonged flumazenil exposure has also been observed. It is thought this may be due to increased synthesis of receptor proteins.[20]
Flumazenil was found to be more effective than placebo in reducing feelings of hostility and aggression in patients who had been free of benzodiazepines for 4–266 weeks.[21] This may suggest a role for flumazenil in treating protracted benzodiazepine withdrawal symptoms.
Low-dose, slow subcutaneous flumazenil administration is a safe procedure for patients withdrawing from long-term, high-dose benzodiazepine dependency.[22] It has a low risk of seizures even amongst those who have experienced convulsions when previously attempting benzodiazepine withdrawal.[23]
In Italy, the gold standard for treatment of high-dose benzodiazepine dependency is 8–10 days of low-dose, slowly infused flumazenil.[24] One addiction treatment centre in Italy has used flumazenil to treat over 300 patients who were dependent on high doses of benzodiazepines (up to 70 times higher than conventionally prescribed) with physicians being among the clinic's most common patients.[25]
Clinical pharmacology
Flumazenil, an imidazobenzodiazepine derivative, antagonizes the actions of benzodiazepines on the central nervous system. Flumazenil competitively inhibits the activity at the benzodiazepine recognition site on the GABA/benzodiazepine receptor complex.[26] It also exhibits weak partial agonism of GABAA receptor complexes that contain α6-type monomers; the clinical relevance of this is unknown.[27]
Flumazenil does not antagonize all of the central nervous system effects of drugs affecting GABA-ergic neurons by means other than the benzodiazepine receptor (including ethanol, barbiturates, and most anesthetics) and does not reverse the effects of opioids. It will however antagonize the action of non-benzodiazepine z-drugs, such as zolpidem and zopiclone, because they act via the benzodiazepine site of the GABA receptor[28] - it has been used to successfully treat z-drug overdose.[28][29][30]
Pharmacodynamics
Intravenous flumazenil has been shown to antagonize sedation, impairment of recall, psychomotor impairment and ventilatory depression produced by benzodiazepines in healthy human volunteers.
The duration and degree of reversal of sedative benzodiazepine effects are related to the dose and plasma concentrations of flumazenil.
Availability
Flumazenil is sold under a wide variety of brand names worldwide like Anexate, Lanexat, Mazicon, Romazicon. In India it is manufactured by Roche Bangladesh Pharmaceuticals and USAN Pharmaceuticals.
See also
- Benzodiazepine overdose
- Benzodiazepine
- Bretazenil
- Imidazenil
- Ro15-4513
- GABAA receptor negative allosteric modulators
References
- ↑ "Selective antagonists of benzodiazepines". Nature 290 (5806): 514–516. April 1981. doi:10.1038/290514a0. PMID 6261143. Bibcode: 1981Natur.290..514H.
- ↑ "Pharmacology of flumazenil". Acta Anaesthesiologica Scandinavica. Supplementum 108: 3–14. 1995-01-01. doi:10.1111/j.1399-6576.1995.tb04374.x. PMID 8693922.
- ↑ "Flumazenil: a benzodiazepine antagonist". BMJ 297 (6655): 999–1000. October 1988. doi:10.1136/bmj.297.6655.999. PMID 2903780.
- ↑ 4.0 4.1 "Modulation of vigilance in the primary hypersomnias by endogenous enhancement of GABAA receptors". Science Translational Medicine 4 (161): 161ra151. November 2012. doi:10.1126/scitranslmed.3004685. PMID 23175709.
- ↑ Clinical trial number NCT01183312 for "Flumazenil for the Treatment of Primary Hypersomnia" at ClinicalTrials.gov
- ↑ 6.0 6.1 6.2 Goldfrank's toxicologic emergencies. New York: McGraw-Hill Medical Publ. Division. 2002. ISBN 978-0-07-136001-2. https://books.google.com/books?id=HVYyRsuUEc0C&q=flumazenil+not+to+be+used+in+overdose&pg=PA948.
- ↑ Goldfrank's toxicologic emergencies. New York: McGraw-Hill, Medical Pub. Division. 2006. ISBN 978-0-07-147914-1.
- ↑ "Flumazenil versus placebo or no intervention for people with cirrhosis and hepatic encephalopathy". The Cochrane Database of Systematic Reviews 2017 (8): CD002798. August 2017. doi:10.1002/14651858.CD002798.pub4. PMID 28796283.
- ↑ "Hypnotics and sedatives.". Goodman & Gilman's: The Pharmacological Basis of Therapeutics (14th ed.). McGraw Hill. 2023. ISBN 978-1-264-25807-9. https://accessanesthesiology.mhmedical.com/content.aspx?bookid=3191§ionid=269719266.
- ↑ Principles of critical care. McGraw-Hill Professional. 2005. ISBN 978-0-07-141640-5. https://books.google.com/books?id=Ss7gzGwk78gC&q=flumazenil+not+recommended+in+overdose&pg=RA11-PA1505.
- ↑ "Midazolam-related drug interactions: detection of risk situations to the patient safety in a brazilian teaching hospital". Journal of Patient Safety 5 (2): 69–74. June 2009. doi:10.1097/PTS.0b013e3181a5dafa. PMID 19920444.
- ↑ "Grey and white matter flumazenil binding in neocortical epilepsy with normal MRI. A PET study of 44 patients". Brain 126 (Pt 6): 1300–1318. June 2003. doi:10.1093/brain/awg138. PMID 12764053.
- ↑ "Feasibility of reversing benzodiazepine tolerance with flumazenil". Lancet 337 (8734): 133–137. January 1991. doi:10.1016/0140-6736(91)90799-U. PMID 1670787.
- ↑ "High dose benzodiazepine dependence: description of 29 patients treated with flumazenil infusion and stabilised with clonazepam". Psychiatry Research 198 (3): 457–462. August 2012. doi:10.1016/j.psychres.2012.02.008. PMID 22424905.
- ↑ "Intravenous flumazenil following prolonged exposure to lormetazepam in humans: lack of precipitated withdrawal". International Clinical Psychopharmacology 11 (2): 81–88. June 1996. doi:10.1097/00004850-199611020-00002. PMID 8803645.
- ↑ "Intravenous flumazenil versus oxazepam tapering in the treatment of benzodiazepine withdrawal: a randomized, placebo-controlled study". Addiction Biology 7 (4): 385–395. October 2002. doi:10.1080/1355621021000005973. PMID 14578014.
- ↑ "Chronic treatment with flumazenil enhances binding sites for convulsants at recombinant alpha(1)beta(2)gamma(2S) GABA(A) receptors". Biomedicine & Pharmacotherapy 59 (7): 408–414. August 2005. doi:10.1016/j.biopha.2005.02.003. PMID 16084060.
- ↑ "Enhancement of benzodiazepine binding sites following chronic treatment with flumazenil". European Journal of Pharmacology 507 (1–3): 7–13. January 2005. doi:10.1016/j.ejphar.2004.10.057. PMID 15659288.
- ↑ "Chronic exposure of cells expressing recombinant GABAA receptors to benzodiazepine antagonist flumazenil enhances the maximum number of benzodiazepine binding sites". Life Sciences 76 (3): 303–317. December 2004. doi:10.1016/j.lfs.2004.07.013. PMID 15531382.
- ↑ "The role of transcriptional and translational mechanisms in flumazenil-induced up-regulation of recombinant GABA(A) receptors". Neuroscience Research 61 (3): 234–241. July 2008. doi:10.1016/j.neures.2008.03.005. PMID 18453026.
- ↑ "Reduction of aggression during benzodiazepine withdrawal: effects of flumazenil". Pharmacology, Biochemistry, and Behavior 96 (2): 148–151. August 2010. doi:10.1016/j.pbb.2010.04.023. PMID 20451546.
- ↑ "Slow subcutaneous infusion of flumazenil for the treatment of long-term, high-dose benzodiazepine users: a review of 214 cases". Journal of Psychopharmacology 30 (10): 1047–1053. October 2016. doi:10.1177/0269881116647505. PMID 27166362.
- ↑ "Low risk of seizures with slow flumazenil infusion and routine anticonvulsant prophylaxis for high-dose benzodiazepine dependence". Journal of Psychopharmacology 31 (10): 1369–1373. October 2017. doi:10.1177/0269881117714050. PMID 28613124.
- ↑ "Agonist substitution for high-dose benzodiazepine-dependent patients: let us not forget the importance of flumazenil". Addiction 106 (4): 853. April 2011. doi:10.1111/j.1360-0443.2010.03327.x. PMID 21320225.
- ↑ "What is stopping us from using flumazenil?". Addiction 107 (7): 1359. July 2012. doi:10.1111/j.1360-0443.2012.03851.x. PMID 22509854.
- ↑ "Benzodiazepine dependence and its treatment with low dose flumazenil". British Journal of Clinical Pharmacology 77 (2): 285–294. February 2014. doi:10.1111/bcp.12023. PMID 23126253.
- ↑ "Cloning of cDNAs encoding the human gamma-aminobutyric acid type A receptor alpha 6 subunit and characterization of the pharmacology of alpha 6-containing receptors". Molecular Pharmacology 49 (2): 253–259. February 1996. PMID 8632757.
- ↑ 28.0 28.1 "The clinical and forensic toxicology of Z-drugs". Journal of Medical Toxicology 9 (2): 155–162. June 2013. doi:10.1007/s13181-013-0292-0. PMID 23404347.
- ↑ "Pediatric zolpidem ingestion demonstrating zero-order kinetics treated with flumazenil". Pediatric Emergency Care 29 (11): 1204–1206. November 2013. doi:10.1097/PEC.0b013e3182aa139c. PMID 24196090.
- ↑ "Zolpidem intoxication mimicking narcotic overdose: response to flumazenil". Human & Experimental Toxicology 9 (2): 105–107. March 1990. doi:10.1177/096032719000900209. PMID 2111156.
Other
- Romazicon product information, Roche USA
External links
{{Navbox | name = GABA receptor modulators | title = GABA receptor modulators | state = collapsed | bodyclass = hlist | groupstyle = text-align:center;
| group1 = Ionotropic | list1 = {{Navbox|subgroup | groupstyle = text-align:center | groupwidth = 5em
| group1 = GABAA | list1 =
- Agonists: (+)-Catechin
- Bamaluzole
- Barbiturates (e.g., phenobarbital)
- BL-1020
- DAVA
- Dihydromuscimol
- GABA
- Gabamide
- GABOB
- Gaboxadol (THIP)
- Homotaurine (tramiprosate, 3-APS)
- Ibotenic acid
- iso-THAZ
- iso-THIP
- Isoguvacine
- Isomuscimol
- Isonipecotic acid
- Kojic amine
- Lignans (e.g., honokiol)
- Methylglyoxal
- Monastrol
- Muscimol
- Nefiracetam
- Neuroactive steroids (e.g., allopregnanolone)
- Org 20599
- PF-6372865
- Phenibut
- Picamilon
- P4S
- Progabide
- Propofol
- Quisqualamine
- SL-75102
- TACA
- TAMP
- Terpenoids (e.g., borneol)
- Thiomuscimol
- Tolgabide
- ZAPA
- Positive modulators (abridged; see here for a full list): α-EMTBL
- Alcohols (e.g., ethanol)
- Anabolic steroids
- Avermectins (e.g., ivermectin)
- Barbiturates (e.g., phenobarbital)
- Benzodiazepines (e.g., diazepam)
- Bromide compounds (e.g., potassium bromide)
- Carbamates (e.g., meprobamate)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Dihydroergolines (e.g., ergoloid (dihydroergotoxine))
- Etazepine
- Etifoxine
- Fenamates (e.g., mefenamic acid)
- Flavonoids (e.g., apigenin, hispidulin)
- Fluoxetine
- Flupirtine
- Imidazoles (e.g., etomidate)
- Kava constituents (e.g., kavain)<!--PMID: 9776662-->
- Lanthanum
- Loreclezole
- Monastrol
- Neuroactive steroids (e.g., allopregnanolone, [[Chemistry:Cholecholesterol]], THDOC)
- Niacin
- Nicotinamide (niacinamide)
- Nonbenzodiazepines (e.g., β-carbolines (e.g., [[abecarnil), cyclopyrrolones (e.g., zopiclone), imidazopyridines (e.g., zolpidem), pyrazolopyrimidines (e.g., zaleplon))
- Norfluoxetine
- Petrichloral
- Phenols (e.g., propofol)
- Phenytoin
- Piperidinediones (e.g., glutethimide)
- Propanidid
- Pyrazolopyridines (e.g., etazolate)
- Quinazolinones (e.g., methaqualone)
- Retigabine (ezogabine)
- ROD-188
- Skullcap constituents (e.g., baicalin)
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal))
- Topiramate
- Valerian constituents (e.g., valerenic acid)
- Volatiles/gases (e.g., chloral hydrate, chloroform, [[Chemistry:Diethyl diethyl ether, Parparaldehyde]], sevoflurane)
- Antagonists: Bicuculline
- Coriamyrtin
- Dihydrosecurinine
- Gabazine (SR-95531)
- Hydrastine
- Hyenachin (mellitoxin)
- PHP-501
- Pitrazepin
- Securinine
- Sinomenine
- SR-42641
- SR-95103
- Thiocolchicoside
- Tutin
- Negative modulators: 1,3M1B
- 3M2B
- 11-Ketoprogesterone
- 17-Phenylandrostenol
- α5IA (LS-193,268)
- β-CCB
- β-CCE
- β-CCM
- β-CCP
- β-EMGBL
- Anabolic steroids
- Amiloride
- Anisatin
- β-Lactams (e.g., penicillins, cephalosporins, carbapenems)
- Basmisanil
- Bemegride
- Bicyclic phosphates (TBPS, TBPO, IPTBO)
- BIDN
- Bilobalide
- Bupropion
- CHEB
- Chlorophenylsilatrane
- Cicutoxin
- Cloflubicyne
- Cyclothiazide
- DHEA
- DHEA-S
- Dieldrin
- (+)-DMBB
- DMCM
- DMPC
- EBOB
- Etbicyphat
- FG-7142 (ZK-31906)
- Fiproles (e.g., fipronil)
- Flavonoids (e.g., amentoflavone, oroxylin A)
- Flumazenil
- Fluoroquinolones (e.g., ciprofloxacin)
- Flurothyl
- Furosemide
- Golexanolone
- Iomazenil (123I)
- IPTBO
- Isopregnanolone (sepranolone)
- L-655,708
- Laudanosine
- Leptazol
- Lindane
- MaxiPost
- Morphine
- Morphine-3-glucuronide
- MRK-016
- Naloxone
- Naltrexone
- Nicardipine
- Nonsteroidal antiandrogens (e.g., [[apalutamide, [[Chemistry:Bicalutbicalutamide, Enzalutenzalutamide, Chemistry:Flutamide|flut]]amide]], nilutamide)
- Oenanthotoxin
- Pentylenetetrazol (pentetrazol)
- Phenylsilatrane
- Picrotoxin (i.e., picrotin, picrotoxinin and dihydropicrotoxinin)
- Pregnenolone sulfate
- Propybicyphat
- PWZ-029
- Radequinil
- Ro 15-4513
- Ro 19-4603
- RO4882224
- RO4938581
- Sarmazenil
- SCS
- Suritozole
- TB-21007
- TBOB
- TBPS
- TCS-1105
- Terbequinil
- TETS
- Thujone
- U-93631
- Zinc
- ZK-93426
| group2 = GABAA-ρ | list2 =
- Agonists: BL-1020
- CACA
- CAMP
- Homohypotaurine
- GABA
- GABOB
- Ibotenic acid
- Isoguvacine
- Muscimol
- N4-Chloroacetylcytosine arabinoside
- Picamilon
- Progabide
- TACA
- TAMP
- Thiomuscimol
- Tolgabide
- Positive modulators: Allopregnanolone
- Alphaxolone
- ATHDOC
- Lanthanides
- Antagonists: (S)-2-MeGABA
- (S)-4-ACPBPA
- (S)-4-ACPCA
- 2-MeTACA
- 3-APMPA
- 4-ACPAM
- 4-GBA
- cis-3-ACPBPA
- CGP-36742 (SGS-742)
- DAVA
- Gabazine (SR-95531)
- Gaboxadol (THIP)
- I4AA
- Isonipecotic acid
- Loreclezole
- P4MPA
- P4S
- SKF-97541
- SR-95318
- SR-95813
- TPMPA
- trans-3-ACPBPA
- ZAPA
- Negative modulators: 5α-Dihydroprogesterone
- Bilobalide
- Loreclezole
- Picrotoxin (picrotin, picrotoxinin)
- Pregnanolone
- ROD-188
- THDOC
- Zinc
}}
| group2 = Metabotropic
| list2 =
| below =
- See also
- Receptor/signaling modulators
- GABAA receptor positive modulators
- GABA metabolism/transport modulators
}}
 |
