Biology:Siroheme
From HandWiki
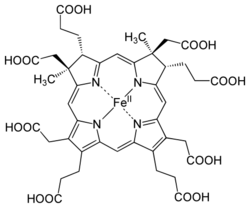
Siroheme (or sirohaem) is a heme-like prosthetic group at the active sites of some enzymes to accomplish the six-electron reduction of sulfur and nitrogen.[1] It is a cofactor at the active site of sulfite reductase, which plays a major role in sulfur assimilation pathway, converting sulfite into sulfide, which can be incorporated into the organic compound homocysteine.[2]
Biosynthesis
Like all tetrapyrroles, the macrocyclic ligand in siroheme is derived from uroporphyrinogen III. This porphyrinogen is methylated at two adjacent pyrrole rings to give dihydrosirohydrochlorin, which is subsequently oxidized to give sirohydrochlorin. A ferrochelatase then inserts iron into the macrocycle to give siroheme.[3]
See also
- Ferredoxin-nitrite reductase
- Hydrogensulfite reductase
- Nitrite reductase (NAD(P)H)
References
- ↑ Matthew J. Murphy (1974). "Siroheme: A New Prosthetic Group Participating in Six-Electron Reduction Reactions Catalyzed by Both Sulfite and Nitrite Reductases". PNAS 71 (3): 612–616. doi:10.1073/pnas.71.3.612. PMID 4595566. Bibcode: 1974PNAS...71..612M.
- ↑ Dominique Thomas; Yolande Surdin-Kerjan (1997). "Metabolism of sulfur amino acids in Saccharomyces cerevisiae". Microbiology and Molecular Biology Reviews 61 (4): 503–532. doi:10.1128/mmbr.61.4.503-532.1997. PMID 9409150.
- ↑ Kaushik Saha; Michaël Moulin; Alison G. Smith (2009). "Wiley Encyclopedia of Chemical Biology". Encyclopedia of Chemical Biology. John Wiley & Sons. doi:10.1002/9780470048672.wecb454. ISBN 978-0-470-04867-2.
Further reading
- Jorgen Hansen (1997). "Siroheme biosynthesis in Saccharomyces cerevisiae requires the products of both MET1 and MET8 genes". FEBS Letters 401 (1): 20–24. doi:10.1016/S0014-5793(96)01423-8. PMID 9003798.
 |
