Biology:Succinate dehydrogenase complex subunit C
 Generic protein structure example |
Succinate dehydrogenase complex subunit C, also known as succinate dehydrogenase cytochrome b560 subunit, mitochondrial, is a protein that in humans is encoded by the SDHC gene.[1] This gene encodes one of four nuclear-encoded subunits that comprise succinate dehydrogenase, also known as mitochondrial complex II, a key enzyme complex of the tricarboxylic acid cycle and aerobic respiratory chains of mitochondria. The encoded protein is one of two integral membrane proteins that anchor other subunits of the complex, which form the catalytic core, to the inner mitochondrial membrane. There are several related pseudogenes for this gene on different chromosomes. Mutations in this gene have been associated with pheochromocytomas and paragangliomas. Alternatively spliced transcript variants have been described.[2]
Structure
The gene that codes for the SDHC protein is nuclear, even though the protein is located in the inner membrane of the mitochondria. The location of the gene in humans is on the first chromosome at q21. The gene is partitioned in 6 exons. The SDHC gene produces an 18.6 kDa protein composed of 169 amino acids.[3][4]
The SDHC protein is one of the two transmembrane subunits of the four-subunit succinate dehydrogenase (Complex II) protein complex that resides in the inner mitochondrial membrane. The other transmembrane subunit is SDHD. The SDHC/SDHD dimer is connected to the SDHB electron transport subunit which, in turn, is connected to the SDHA subunit.[5]
Function
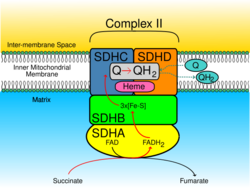
The SDHC protein is one of four nuclear-encoded subunits that comprise succinate dehydrogenase, also known as Complex II of the electron transport chain, a key enzyme complex of the citric acid cycle and aerobic respiratory chains of mitochondria. The encoded protein is one of two integral membrane proteins that anchor other subunits of the complex, which form the catalytic core, to the inner mitochondrial membrane.[2]
SDHC forms part of the transmembrane protein dimer with SDHD that anchors Complex II to the inner mitochondrial membrane. The SDHC/SDHD dimer provides binding sites for ubiquinone and water during electron transport at Complex II. Initially, SDHA oxidizes succinate via deprotonation at the FAD binding site, forming FADH2 and leaving fumarate, loosely bound to the active site, free to exit the protein. The electrons derived from succinate tunnel along the [Fe-S] relay in the SDHB subunit until they reach the [3Fe-4S] iron sulfur cluster. The electrons are then transferred to an awaiting ubiquinone molecule at the Q pool active site in the SDHC/SDHD dimer. The O1 carbonyl oxygen of ubiquinone is oriented at the active site (image 4) by hydrogen bond interactions with Tyr83 of SDHD. The presence of electrons in the [3Fe-4S] iron sulphur cluster induces the movement of ubiquinone into a second orientation. This facilitates a second hydrogen bond interaction between the O4 carbonyl group of ubiquinone and Ser27 of SDHC. Following the first single electron reduction step, a semiquinone radical species is formed. The second electron arrives from the [3Fe-4S] cluster to provide full reduction of the ubiquinone to ubiquinol.[6]
Clinical significance
Mutations in this gene have been associated with paragangliomas.[2][7] More than 30 mutations in the SDHC gene have been found to increase the risk of hereditary paraganglioma-pheochromocytoma type 3. People with this condition have paragangliomas, pheochromocytomas, or both. An inherited SDHC gene mutation predisposes an individual to the condition, and a somatic mutation that deletes the normal copy of the SDHC gene is needed to cause hereditary paraganglioma-pheochromocytoma type 3. Most of the inherited SDHC gene mutations change single amino acids in the SDHC protein sequence or result in a shortened protein. As a result, there is little or no SDH enzyme activity. Because the mutated SDH enzyme cannot convert succinate to fumarate, succinate accumulates in the cell. The excess succinate abnormally stabilizes hypoxia-inducible factors (HIF), which also builds up in cells. Excess HIF stimulates cells to divide and triggers the production of blood vessels when they are not needed. Rapid and uncontrolled cell division, along with the formation of new blood vessels, can lead to the development of tumors in people with hereditary paraganglioma-pheochromocytoma.[8]
Interactive pathway map
References
- ↑ "Cytochrome b in human complex II (succinate-ubiquinone oxidoreductase): cDNA cloning of the components in liver mitochondria and chromosome assignment of the genes for the large (SDHC) and small (SDHD) subunits to 1q21 and 11q23". Cytogenetics and Cell Genetics 79 (1–2): 132–8. 1997. doi:10.1159/000134700. PMID 9533030.
- ↑ 2.0 2.1 2.2 "Entrez Gene: succinate dehydrogenase complex". https://www.ncbi.nlm.nih.gov/sites/entrez?Db=gene&Cmd=ShowDetailView&TermToSearch=6391.
- ↑ "Integration of cardiac proteome biology and medicine by a specialized knowledgebase". Circulation Research 113 (9): 1043–53. October 2013. doi:10.1161/CIRCRESAHA.113.301151. PMID 23965338.
- ↑ "SDHC - Succinate dehydrogenase cytochrome b560 subunit, mitochondrial". Cardiac Organellar Protein Atlas Knowledgebase (COPaKB). https://amino.heartproteome.org/web/protein/Q99643.
- ↑ "Crystal structure of mitochondrial respiratory membrane protein complex II". Cell 121 (7): 1043–57. July 2005. doi:10.1016/j.cell.2005.05.025. PMID 15989954.
- ↑ "Structural and computational analysis of the quinone-binding site of complex II (succinate-ubiquinone oxidoreductase): a mechanism of electron transfer and proton conduction during ubiquinone reduction". The Journal of Biological Chemistry 281 (11): 7309–16. March 2006. doi:10.1074/jbc.m508173200. PMID 16407191.
- ↑ "Autosomal dominant malignant and catecholamine-producing paraganglioma caused by a splice donor site mutation in SDHC". Human Genetics 113 (1): 92–4. July 2003. doi:10.1007/s00439-003-0938-0. PMID 12658451.
- ↑ "SDHC". U.S. National Library of Medicine. http://ghr.nlm.nih.gov/gene/SDHC.
Further reading
- "Molecular characterization of novel germline deletions affecting SDHD and SDHC in pheochromocytoma and paraganglioma patients". Endocrine-Related Cancer 16 (3): 929–37. September 2009. doi:10.1677/ERC-09-0084. PMID 19546167.
- "Clinical and molecular genetics of patients with the Carney-Stratakis syndrome and germline mutations of the genes coding for the succinate dehydrogenase subunits SDHB, SDHC, and SDHD". European Journal of Human Genetics 16 (1): 79–88. January 2008. doi:10.1038/sj.ejhg.5201904. PMID 17667967.
- "Isocitrate dehydrogenase mutations are rare in pheochromocytomas and paragangliomas". The Journal of Clinical Endocrinology and Metabolism 95 (3): 1274–8. March 2010. doi:10.1210/jc.2009-2170. PMID 19915015.
- "Development and validation of a comprehensive mutation and deletion detection assay for SDHB, SDHC, and SDHD". Clinical Biochemistry 43 (7–8): 700–4. May 2010. doi:10.1016/j.clinbiochem.2010.01.016. PMID 20153743.
- "Single nucleotide polymorphisms in succinate dehydrogenase subunits and citrate synthase genes: association results for impaired spermatogenesis". International Journal of Andrology 30 (3): 144–52. June 2007. doi:10.1111/j.1365-2605.2006.00730.x. PMID 17298551.
- "Rationalization of genetic testing in patients with apparently sporadic pheochromocytoma/paraganglioma". Hormone and Metabolic Research 41 (9): 672–5. September 2009. doi:10.1055/s-0029-1202814. PMID 19343621.
- "No association of an SDHC gene polymorphism with gastric cancer". Asian Pacific Journal of Cancer Prevention 7 (4): 525–8. 2006. PMID 17250422.
- "Genetics of pheochromocytoma and paraganglioma in Spanish patients". The Journal of Clinical Endocrinology and Metabolism 94 (5): 1701–5. May 2009. doi:10.1210/jc.2008-2756. PMID 19258401.
- "Malignant head and neck paragangliomas in SDHB mutation carriers". Otolaryngology–Head and Neck Surgery 137 (1): 126–9. July 2007. doi:10.1016/j.otohns.2007.01.015. PMID 17599579.
- "Immunohistochemistry for SDHB triages genetic testing of SDHB, SDHC, and SDHD in paraganglioma-pheochromocytoma syndromes". Human Pathology 41 (6): 805–14. June 2010. doi:10.1016/j.humpath.2009.12.005. PMID 20236688.
- "Germline SDHB mutations and familial renal cell carcinoma". Journal of the National Cancer Institute 100 (17): 1260–2. September 2008. doi:10.1093/jnci/djn254. PMID 18728283.
- "Familial gastrointestinal stromal tumors and germ-line mutations". The New England Journal of Medicine 357 (10): 1054–6. September 2007. doi:10.1056/NEJMc071191. PMID 17804857.
- "A role for mitochondrial enzymes in inherited neoplasia and beyond". Nature Reviews. Cancer 3 (3): 193–202. March 2003. doi:10.1038/nrc1013. PMID 12612654.
- "Relevance of germline mutation screening in both familial and sporadic head and neck paraganglioma for early diagnosis and clinical management". Cellular Oncology 32 (4): 275–83. January 2010. doi:10.3233/CLO-2009-0498. PMID 20208144.
- "Succinate dehydrogenase deficiency in human". Cellular and Molecular Life Sciences 62 (19–20): 2317–24. October 2005. doi:10.1007/s00018-005-5237-6. PMID 16143825.
- "Clinically guided genetic screening in a large cohort of italian patients with pheochromocytomas and/or functional or nonfunctional paragangliomas". The Journal of Clinical Endocrinology and Metabolism 94 (5): 1541–7. May 2009. doi:10.1210/jc.2008-2419. PMID 19223516. https://iris.unito.it/bitstream/2318/96662/4/Clinically%20Guided%20Genetic%20Screening.pdf.
- "A role for succinate dehydrogenase genes in low chemoresponsiveness to hypoxia?". Clinical Autonomic Research 19 (6): 335–42. December 2009. doi:10.1007/s10286-009-0028-z. PMID 19768395.
- "Should genetic testing be performed in each patient with sporadic pheochromocytoma at presentation?". European Journal of Endocrinology 160 (2): 227–31. February 2009. doi:10.1530/EJE-08-0574. PMID 19029228.
- "Genetic analyses of apparently sporadic pheochromocytomas: the Rotterdam experience". Annals of the New York Academy of Sciences 1073 (1): 138–48. August 2006. doi:10.1196/annals.1353.014. PMID 17102080. Bibcode: 2006NYASA1073..138K.
- "Polymorphisms in mitochondrial genes and prostate cancer risk". Cancer Epidemiology, Biomarkers & Prevention 17 (12): 3558–66. December 2008. doi:10.1158/1055-9965.EPI-08-0434. PMID 19064571.
External links
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
 |