Chemistry:Aluminium borohydride
| |
| Names | |
|---|---|
| IUPAC name
Aluminium borohydride
| |
| Other names
Aluminium borohydride, aluminium tetrahydroborate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| UN number | 2870 |
| |
| |
| Properties | |
| AlB3H12 | |
| Molar mass | 71.51 g·mol−1 |
| Appearance | colorless liquid |
| Melting point | −64.5 °C (−84.1 °F; 208.7 K) |
| Boiling point | 44.5 °C (112.1 °F; 317.6 K) |
| reacts | |
| Hazards | |
| Flash point | Spontaneously ignites |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
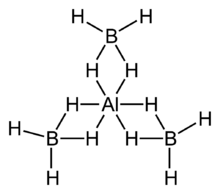
Aluminium borohydride, also known as aluminium tetrahydroborate, is the chemical compound with the formula Al(BH4)3. It is a volatile pyrophoric liquid which is used as a reducing agent in laboratories. Unlike most other metal–borohydrides, which are ionic structures, aluminium borohydride is a covalent compound.[2][3]
Preparation
Aluminium borohydride is formed by the reaction between sodium borohydride with aluminium chloride:[4]
- 3 NaBH4 + AlCl3 → Al(BH4)3 + 3 NaCl
or as the non-pyrophoric tetrahydrofuran (THF) adduct, by the analogous reaction of calcium borohydride and aluminium chloride in THF:[2]
- 3 Ca(BH4)2 + 2 AlCl3 → 3 CaCl2 + 2 Al(BH4)3
Reactions
Like most borohydrides, this compound is a reducing agent and hydride donor. It reacts with water to give elemental hydrogen gas,[4] and reduces carboxylic esters, aldehydes, and ketones to alcohols.[2]
It is used to prepare metal complexes of borohydride:[5]
- AnF4 + 2 Al(BH4)3 → An(κ3-BH4)4 + 2AIF2BH4 (An = actinide metal)
When aluminium borohydride is heated, diborane is released, and a dialuminium dihydride forms:[6]
- 2 Al(BH4)3 → Al2H2(BH4)4 + B2H6
References
- ↑ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–39. ISBN 0-8493-0594-2.
- ↑ 2.0 2.1 2.2 J. Kollonitsch & O. Fuchs (1955). "Preparation of Aluminium Borohydride and its Applications in Organic Reductions". Nature 176 (4492): 1081. doi:10.1038/1761081a0. Bibcode: 1955Natur.176.1081K.
- ↑ Miwa, K.; Ohba, N.; Towata, S.; Nakamori, Y.; Züttel, A.; Orimo, S. (2007). "First-principles study on thermodynamical stability of metal borohydrides: Aluminum borohydride Al(BH4)3". J. Alloys Compd. 446–447: 310–314. doi:10.1016/j.jallcom.2006.11.140.
- ↑ 4.0 4.1 Perry, Dale L.; Phillips, Sidney L. (1995). Handbook of Inorganic Compounds. CRC Press. pp. 3–4. ISBN 0-8493-8671-3. https://books.google.com/books?id=0fT4wfhF1AsC. Retrieved 2007-12-09.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 168. ISBN 978-0-08-037941-8.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 230. ISBN 978-0-08-037941-8.
Further reading
- Fletcher, Edward; Foster, Hampton; Straight, David (1959). "Aluminum Borohydride and Mixtures with Hydrocarbons in Jet Engine Combustor Ignition". Industrial & Engineering Chemistry 51 (11): 1389. doi:10.1021/ie50599a044.
- Hinkamp, James B.; Hnizda, Vincent (1955). "Aluminum Borohydride Preparation". Industrial & Engineering Chemistry 47 (8): 1560. doi:10.1021/ie50548a032.
 |
