Chemistry:Aluminium fluoride
 Anhydrous AlF
3 | |
| Names | |
|---|---|
| Other names
Aluminium(III) fluoride
Aluminum trifluoride | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| AlF 3 | |
| Molar mass |
|
| Appearance | Colorless to white crystalline solid |
| Odor | Odorless |
| Density |
|
| Melting point | 1,290 °C (2,350 °F; 1,560 K)[4] (anhydrous) (sublimes) |
| |
| −13.4×10−6 cm3/mol[2] | |
Refractive index (nD)
|
1.3767 (visible range)[3] |
| Structure | |
| Rhombohedral, hR24 | |
| R3c, No. 167[5] | |
a = 0.49254 nm, c = 1.24477 nm
| |
Lattice volume (V)
|
0.261519 |
Formula units (Z)
|
6 |
| Thermochemistry | |
Heat capacity (C)
|
75.1 J/(mol·K)[6] |
Std molar
entropy (S |
66.5 J/(mol·K)[6] |
Std enthalpy of
formation (ΔfH⦵298) |
−1510.4 kJ/mol[6] |
Gibbs free energy (ΔfG˚)
|
−1431.1 kJ/mol[6] |
| Hazards[7][8][9] | |
| Safety data sheet | InChem MSDS |
| GHS pictograms |   
|
| GHS Signal word | DANGER |
| H301, H302, H314, H315, H319, H335, H361, H372 | |
| P260, P261, P264, P270, P271, P280, P301+310, P301+312, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P312, P321, P330, P332+313, P337+313, P362, P363, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
none |
REL (Recommended)
|
2 mg/m3 |
IDLH (Immediate danger)
|
N.D. |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Aluminium fluoride is an inorganic compound with the formula AlF
3. It forms hydrates AlF
3 · xH
2O. Anhydrous AlF
3 and its hydrates are all colorless solids. Anhydrous AlF
3 is used in the production of aluminium. Several occur as minerals.
Occurrence and production
Aside from anhydrous AlF
3, several hydrates are known. With the formula AlF
3 · xH
2O, these compounds include monohydrate (x = 1), two polymorphs of the trihydrate (x = 3), a hexahydrate (x = 6), and a nonahydrate (x = 9).[10]
The majority of aluminium fluoride is produced by treating alumina with hydrogen fluoride at 700 °C:[4] Hexafluorosilicic acid may also be used make aluminium fluoride.[11]
- H
2[SiF
6] + Al
2O
3 + 3 H
2O → 2 AlF
3 + SiO
2 + 4 H
2O
Alternatively, it is manufactured by thermal decomposition of ammonium hexafluoroaluminate.[12] For small scale laboratory preparations, AlF
3 can also be prepared by treating aluminium hydroxide or aluminium with hydrogen fluoride.
Aluminium fluoride trihydrate is found in nature as the rare mineral rosenbergite.
The anhydrous form appears as the relatively recently (as of 2020) recognized mineral óskarssonite.[13][14] A related, exceedingly rare mineral, is zharchikhite, AlF(OH)
2.[15][14]
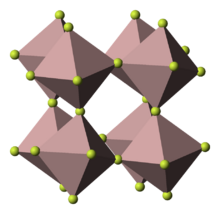
Structure
According to X-ray crystallography, anhydrous AlF
3 adopts the rhenium trioxide motif, featuring distorted AlF
6 octahedra. Each fluoride is connected to two Al centers. Because of its three-dimensional polymeric structure, AlF
3 has a high melting point. The other trihalides of aluminium in the solid state differ, AlCl
3 has a layer structure and AlBr
3 and AlI
3, are molecular dimers.[16] Also they have low melting points and evaporate readily to give dimers.[17]{{page needed|date=January 2020} ride exists as trigonal molecules of D3h symmetry. The Al–F bond lengths of this gaseous molecule are 163 pm.

3 adopts a planar structure upon evaporation.
Applications
Aluminium fluoride is an important additive for the production of aluminium by electrolysis.[4] Together with cryolite, it lowers the melting point to below 1000 °C and increases the conductivity of the solution. It is into this molten salt that aluminium oxide is dissolved and then electrolyzed to give bulk Al metal.[12]
Aluminium fluoride complexes are used to study the mechanistic aspects of phosphoryl transfer reactions in biology, which are of fundamental importance to cells, as phosphoric acid anhydrides such as adenosine triphosphate and guanosine triphosphate control most of the reactions involved in metabolism, growth and differentiation.[18] The observation that aluminium fluoride can bind to and activate heterotrimeric G proteins has proven to be useful for the study of G protein activation in vivo, for the elucidation of three-dimensional structures of several GTPases, and for understanding the biochemical mechanism of GTP hydrolysis, including the role of GTPase-activating proteins.[19]
Niche uses
Together with zirconium fluoride, aluminium fluoride is an ingredient for the production of fluoroaluminate glasses.
It is also used to inhibit fermentation.
Like magnesium fluoride it is used as a low-index optical thin film, particularly when far UV transparency is required. Its deposition by physical vapor deposition, particularly by evaporation, is favorable.
Safety
The reported oral animal lethal dose (LD50) of aluminium fluoride is 100 mg/kg.[20] Repeated or prolonged inhalation exposure may cause asthma, and may have effects on the bone and nervous system, resulting in bone alterations (fluorosis), and nervous system impairment.[21]
Many of the neurotoxic effects of fluoride are due to the formation of aluminium fluoride complexes, which mimic the chemical structure of a phosphate and influence the activity of ATP phosphohydrolases and phospholipase D. Only micromolar concentrations of aluminium are needed to form aluminium fluoride.[22]
Human exposure to aluminium fluoride can occur in an industrial setting, such as emissions from aluminium reduction processes,[23] or when a person ingests both a fluoride source (e.g., fluoride in drinking water or residue of fluoride-based pesticides) and an aluminium source; sources of human exposure to aluminium include drinking water, tea, food residues, infant formula, aluminium-containing antacids or medications, deodorants, cosmetics, and glassware.[22] Fluoridation chemicals may also contain aluminium fluoride.[24] Data on the potential neurotoxic effects of chronic exposure to the aluminium species existing in water are limited.[25]
See also
References
- ↑ 1.0 1.1 Haynes, William M., ed (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.45. ISBN 1439855110.
- ↑ Haynes, William M., ed (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.131. ISBN 1439855110.
- ↑ Lide, David R. (2003-06-19). CRC Handbook of Chemistry and Physics. CRC Handbook (84th ed.). CRC Press. ISBN 9780849304842. https://books.google.com/books?id=kTnxSi2B2FcC.
- ↑ 4.0 4.1 4.2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 233. ISBN 978-0-08-037941-8.
- ↑ Hoppe, R.; Kissel, D. (1984). "Zur kenntnis von AlF3 und InF3 [1]". Journal of Fluorine Chemistry 24 (3): 327. doi:10.1016/S0022-1139(00)81321-4.
- ↑ 6.0 6.1 6.2 6.3 Haynes, William M., ed (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 5.5. ISBN 1439855110.
- ↑ Pohanish, Richard P. (2005-03-04) (in en). HazMat Data: For First Response, Transportation, Storage, and Security. John Wiley & Sons. ISBN 9780471726104. https://books.google.com/books?id=3Tbl2W8j-3oC&q=nfpa+704+aluminum+fluoride.
- ↑ "Aluminum Fluoride". National Institute of Health. https://pubchem.ncbi.nlm.nih.gov/compound/aluminum_fluoride.
- ↑ NIOSH Pocket Guide to Chemical Hazards. "#0024". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0024.html.
- ↑ Guangmei Wang; Anja-Verena Mudring (2016). "The missing Hydrate AlF3·6H2O [Al(H2O)6]F3: Ionothermal Synthesis, Crystal Structure and Characterization of Aluminum Fluoride Hexahydrate". Solid State Sciences 61: 61. doi:10.1016/j.solidstatesciences.2016.09.007.
- ↑ Dreveton, Alain (2012-01-01). "Manufacture of Aluminium Fluoride of High Density and Anhydrous Hydrofluoric Acid from Fluosilicic Acid". Procedia Engineering. SYMPHOS 2011 - 1st International Symposium on Innovation and Technology in the Phosphate Industry 46 (Supplement C): 255–265. doi:10.1016/j.proeng.2012.09.471.
- ↑ 12.0 12.1 Aigueperse, J.; Mollard, P.; Devilliers, D.; Chemla, M.; Faron, R.; Romano, R.; Cuer, J. P.. "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH.
- ↑ "Óskarssonite". https://www.mindat.org/min-43853.html.
- ↑ 14.0 14.1 "List of Minerals". 21 March 2011. https://www.ima-mineralogy.org/Minlist.htm.
- ↑ "Zharchikhite". https://www.mindat.org/min-4399.html.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego, CA: Academic Press. ISBN 0-12-352651-5..
- ↑ Wittinghofer, Alfred (1997-11-01). "Signaling mechanistics: Aluminum fluoride for molecule of the year". Current Biology 7 (11): R682–R685. doi:10.1016/S0960-9822(06)00355-1. PMID 9382787. Bibcode: 1997CBio....7R.682W.
- ↑ Vincent, Sylvie; Brouns, Madeleine; Hart, Matthew J.; Settleman, Jeffrey (1998-03-03). "Evidence for distinct mechanisms of transition state stabilization of GTPases by fluoride" (in en). Proceedings of the National Academy of Sciences 95 (5): 2210–2215. doi:10.1073/pnas.95.5.2210. ISSN 0027-8424. PMID 9482864. Bibcode: 1998PNAS...95.2210V.
- ↑ "ALUMINUM FLUORIDE, CASRN: 7784-18-1". CDC.gov. June 24, 2005. https://www.nlm.nih.gov/toxnet/index.html.
- ↑ "ALUMINIUM FLUORIDE (ANHYDROUS) International Chemical Safety Cards (ICSC)". July 22, 2015. https://www.cdc.gov/niosh/ipcsneng/neng1324.html.
- ↑ 22.0 22.1 (in en) Fluoride in Drinking Water: A Scientific Review of EPA's Standards. The National Academies Press. 2006. pp. 51–52, 219. doi:10.17226/11571. ISBN 978-0-309-10128-8. https://www.nap.edu/read/11571.
- ↑ TOXICOLOGICAL PROFILE FOR FLUORIDES, HYDROGEN FLUORIDE, AND FLUORINE. U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service Agency for Toxic Substances and Disease Registry. 2003. pp. 211. https://www.atsdr.cdc.gov/toxprofiles/tp11.pdf.
- ↑ Mullenix, Phyllis J (2014). "A new perspective on metals and other contaminants in fluoridation chemicals". International Journal of Occupational and Environmental Health 20 (2): 157–166. doi:10.1179/2049396714Y.0000000062. ISSN 1077-3525. PMID 24999851.
- ↑ Aluminum Compounds Review of Toxicological Literature Abridged Final Report. Prepared for National Institute of Environmental Health Sciences. NTP.gov Nomination Summary for Aluminum contaminants of drinking water (N20025). October 2001
External links
 |

