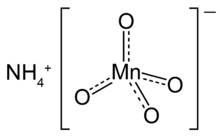
Chemistry:Ammonium permanganate
| |
| Names | |
|---|---|
| IUPAC name
Ammonium manganate(VII)
| |
| Other names
Ammonium permanganate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UN number | 3085, 1482 |
| |
| |
| Properties | |
| NH4MnO4 | |
| Molar mass | 136.974 g/mol |
| Appearance | rhombic needle crystals or powder with rich violet-brown or dark purple metallic sheen, become steel-gray in storage; magenta–rose in solution |
| Density | 2.2g/cm3, solid |
| Melting point | decomposes |
| 8.0 g/100 ml at 15 °C | |
| Structure | |
| Orthorhombic | |
| Thermochemistry | |
Std molar
entropy (S |
J.K−1.mol−1 |
| Hazards | |
| Main hazards | Oxidant (O), Harmful (Xn), Dangerous for the environment (N) |
| Safety data sheet | [ External MSDS] |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
Ammonium perrhenate |
Other cations
|
Sodium permanganate; Potassium permanganate |
Related compounds
|
Potassium manganate (K2MnO4); Manganese heptoxide; |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ammonium permanganate is the chemical compound NH4MnO4, or NH3·HMnO4. It is a water soluble, violet-brown or dark purple salt.
Preparation
Ammonium permanganate was first prepared by Eilhard Mitscherlich in 1824 by reaction of silver permanganate with equal molar amount of ammonium chloride, filtering the silver chloride and evaporating the water.
- AgMnO4 + NH4Cl → AgCl + NH4MnO4
It can also be prepared in a similar way from potassium permanganate and ammonium chloride.
- KMnO4 +NH4Cl → KCl + NH4MnO4
Properties
Ammonium permanganate is a strong oxidizer, owing to its permanganate anion, and it is a moderately strong explosive, owing to the combination of oxidizer permanganate anion and reducing ammonium cation. Dry ammonium permanganate can detonate by heat, shock, or friction, and it may explode at temperatures above 140 °F (60 °C).[1]
Ammonium permanganate decomposes explosively to manganese dioxide, nitrogen, and water:[2]
- 2 NH4MnO4 → 2 MnO2 + N2 + 4 H2O
Ammonium permanganate decomposes slowly in storage even at normal temperatures. A sample stored for 3 months was only 96% pure, after 6 months it assumed color of iodine and had strong smell of nitrogen oxides. It emits toxic fumes when decomposed by heat.[3]
Quaternary ammonium permanganate compounds can be prepared, such as tetrabutylammonium permanganate[4] and benzyltriethylammonium permanganate.[5]
References
- ↑ "MSDS Chemical Information File: Ammonium Permanganate". February 1988. http://www.textfiles.com/science/CHEMICALS/ammonium_permanganate.txt.
- ↑ Chisholm, Hugh, ed (1911). "Manganese". Encyclopædia Britannica. 17 (11th ed.). Cambridge University Press. pp. 569–571.
- ↑ Seymour M. Kaye (1978). "Permanganates - Ammonium Permanganate". Encyclopedia of Explosives and Related Items. 8. p. P178.
- ↑ Sala, Tony; Sargent, Melvyn V. (1978). "Tetrabutylammonium permanganate: an efficient oxidant for organic substrates". Journal of the Chemical Society, Chemical Communications (6): 253–254. doi:10.1039/C39780000253.
- ↑ Schmidt, H.-Jürgen; Schäfer, Hans J. (January 1979). "Oxidation of Hydrocarbons with Benzyl(triethyl)ammonium Permanganate". Angewandte Chemie International Edition in English 18 (1): 68–69. doi:10.1002/anie.197900681.
 |

