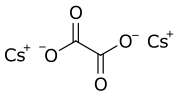
Chemistry:Caesium oxalate
| |
| Names | |
|---|---|
| IUPAC name
Caesium oxalate
| |
| Preferred IUPAC name
Dicaesium oxalate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| Cs 2C 2O 4 | |
| Molar mass | 353.829 g·mol−1 |
| Appearance | White solid |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302, H312 | |
| P264, P270, P280, P301+312, P302+352, P312, P322, P330, P363, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Caesium oxalate (standard IUPAC spelling), or dicesium oxalate, or cesium oxalate (American spelling) is a chemical compound with the chemical formula Cs
2C
2O
4. It is a caesium salt of oxalic acid. It consists of caesium cations Cs+
and oxalate anions C
2O2−
4.
Preparation
Caesium oxalate can be prepared by passing carbon monoxide and carbon dioxide over caesium carbonate at 380 °C:[1]
- Cs
2CO
3 + CO → Cs
2C
2O
4
Other alkali carbonates do not undergo transformation to oxalate.[1]
Caesium carbonate can react with oxalic acid in aqueous solution to give caesium oxalate.[2]
- Cs
2CO
3 + H
2C
2O
4 · 2H2O → Cs
2C
2O
4 · H2O + CO
2 + 2 H
2O
Chemical Reactions
Caesium oxalate can be reduced back into caesium carbonate and carbon monoxide by thermal decomposition.
- Cs
2C
2O
4 → Cs
2CO
3 + CO
Double salts
Compounds that contain caesium and another element in addition to the oxalate anion are double salts of caesium and oxalate. The oxalate may form a complex with a metal that can make a salt with caesium.
Examples include:
| name | formula | properties | reference |
|---|---|---|---|
| caesium bis(oxalato)oxotitanate(IV) trihydrate | Cs 4[TiO(C 2O 4) 2] 2 · 3H2O |
[3] | |
| caesium oxalatooxovanadate(IV) | Cs 2[VO(C 2O 4) 2] |
[4] | |
| caesium tris(oxalato) ferrate(III) dihydrate | Cs 3[Fe(C 2O 4) 3] · 2H2O |
[5] | |
| caesium bis(oxalato) cobaltate(II) tetrahydrate | Cs 2[Co(C 2O 4) 2] · 4H2O |
[6] | |
| caesium bis(oxalato)nickelate(III) tetrahydrate | Cs[Ni(C 2O 4) 2] · 4H2O |
[7] | |
| caesium tris(oxalato)germanate(IV) | Cs 2[Ge(C 2O 4) 3] |
[8] | |
| caesium yttrium(III) oxalate monohydrate | CsY(C 2O 4) 2 · H2O |
monoclinic a = 8.979, b = 6.2299, c = 8.103 Å, β = 90.05° V = 453.3 Å3, space group P2/n | [9] |
| caesium (diaquo)bis(oxalato)oxoniobate(V) dihydrate | Cs[NbO(C 2O 4) 2(H 2O) 2] · 2H2O |
[10] | |
| Cs 2[NH 4] 2[Mo 3O 8(C 2O 4) 3] |
[11] | ||
| tetracaesium dilanthanum(III) oxalate octahydrate | Cs 4La 2(C 2O 4) 5 · 8H2O |
[12] | |
| tetracaesium dipraseodymium(III) oxalate octahydrate | Cs 4Pr 2(C 2O 4) 5 · 8H2O |
[12] | |
| caesium neodymium(III) oxalate hexahydrate | CsNd(C 2O 4) 2 · 6H2O |
[12] | |
| caesium samarium(III) oxalate hexahydrate | CsSm(C 2O 4) 2 · 6H2O |
[12] | |
| caesium dysprosium(III) oxalate hydrate | CsDy(C 2O 4) 2 · ?H 2O |
[12] | |
| caesium gadolinium(III) oxalate sesquihydrate | CsGd(C 2O 4) 2 · 1.5H2O |
[12] | |
| caesium terbium(III) oxalate sesquihydrate | CsTb(C 2O 4) 2 · 1.5H2O |
[12] | |
| caesium dysprosium(III) oxalate sesquihydrate | CsDy(C 2O 4) 2 · 1.5H2O |
[12] | |
| caesium holmium(III) oxalate sesquihydrate | CsHo(C 2O 4) 2 · 1.5H2O |
[12] | |
| caesium ytterbium(III) oxalate sesquihydrate | CsYb(C 2O 4) 2 · 1.5H2O |
[12] | |
| caesium lutetium(III) oxalate sesquihydrate | CsLu(C 2O 4) 2 · 1.5H2O |
[12] | |
| dicaesium dioxotungsten(VI) oxalate | Cs 2[WO 2](C 2O 4) 2 |
[13] | |
| dicaesium dioxotungsten(VI) difluoride monooxalate | Cs 2[WO 2]F 2(C 2O 4) |
[13] | |
| caesium tris(oxalato)rhenate(III) | Cs 3[Re(C 2O 4) 3] |
[14] | |
| dicaesium uranyl monooxalate monosulfate dihydrate | Cs 2[UO 2](C 2O 4)(SO 4) · 2H2O |
[15] | |
| ammonium caesium uranyl monooxalate monosulfate dihydrate | [NH 4]Cs[UO 2](C 2O 4)(SO 4) · 2H2O |
[15] | |
| caesium dioxoneptunium(VI) oxalate hydrate | Cs[NpO 2]C 2O 4 · nH 2O |
[16] |
Mixed anion compounds containing caesium, oxalate and another anion also exist, such as the uranyl sulfate above, and caesium bis(oxalato)borate (CsBOB) (Cs[B(C
2O
4)
2]).[17]
References
- ↑ 1.0 1.1 Kudo, Kiyoshi; Ikoma, Futoshi; Mori, Sadayuki; Komatsu, Koichi; Sugita, Nobuyuki (1997). "Synthesis of oxalate from carbon monoxide and carbon dioxide in the presence of caesium carbonate". Journal of the Chemical Society, Perkin Transactions 2 (4): 679–682. doi:10.1039/A607856G.
- ↑ Dinnebier, Robert E.; Vensky, Sascha; Panthöfer, Martin; Jansen, Martin (2003-03-01). "Crystal and Molecular Structures of Alkali Oxalates: First Proof of a Staggered Oxalate Anion in the Solid State". Inorganic Chemistry 42 (5): 1499–1507. doi:10.1021/ic0205536. ISSN 0020-1669. PMID 12611516. https://doi.org/10.1021/ic0205536.
- ↑ Fester, A.; Bensch, W.; Trömel, M. (March 1992). "Crystal structure of cesium-bis(oxalato)oxo-titanate(IV) hydrate". Inorganica Chimica Acta 193 (1): 99–103. doi:10.1016/S0020-1693(00)83801-3.
- ↑ Bhaumik, B. B.; Chattopadhyay, R. K. (April 1981). "Oxalatooxovanadates (IV)". Indian J. Chem. 20A: 417–419. http://nopr.niscair.res.in/bitstream/123456789/50080/1/IJCA_20A%284%29_417-419.pdf.
- ↑ Randhawa, B. S. (September 1995). "Mössbauer study on thermal decomposition of cesium tris(oxalato) ferrate(III) dihydrate". Journal of Radioanalytical and Nuclear Chemistry 201 (1): 57–63. doi:10.1007/bf02204772.
- ↑ Schwendtner, Karolina; Kolitsch, Uwe (2004-05-15). "Cs 2 Co II (C 2 O 4 ) 2 ·4H 2 O". Acta Crystallographica Section E 60 (5): m659–m661. doi:10.1107/S1600536804009626. ISSN 1600-5368. Bibcode: 2004AcCrE..60M.659S. http://scripts.iucr.org/cgi-bin/paper?S1600536804009626.
- ↑ Modebadze, M. E. (1987). "Study of oxalate compounds of nickel" (in Russian). Investigations in Chemistry of Complexes and Compounds of Certain Transition and Rare Metals. No. 4. http://inis.iaea.org/Search/search.aspx?orig_q=RN:20028788.
- ↑ Lopez, Jordan R.; Akutsu, Hiroki; Martin, Lee (November 2015). "Radical-cation salt with novel BEDT-TTF packing motif containing tris(oxalato)germanate(IV)". Synthetic Metals 209: 188–191. doi:10.1016/j.synthmet.2015.07.019. http://irep.ntu.ac.uk/id/eprint/28068/1/PubSub5587_Martin.pdf.
- ↑ Bataille, Thierry; Auffrédic, Jean-Paul; Louër, Daniel (2000). "Crystal structure and thermal behaviour of the new layered oxalate Y(H2O)Cs(C2O4)2 studied by powder X-ray diffraction". Journal of Materials Chemistry 10 (7): 1707–1711. doi:10.1039/b001201g. http://xlink.rsc.org/?DOI=b001201g.
- ↑ Brničević, N.; Djordjević, C. (February 1976). "Co-ordination complexes of niobium and tantalum XVI. salts of oxy-bis-oxalato niobate (V)" (in en). Journal of the Less Common Metals 45 (1): 45–52. doi:10.1016/0022-5088(76)90195-8. https://linkinghub.elsevier.com/retrieve/pii/0022508876901958.
- ↑ Goel, S. P.; Verma, G. R.; Kumar, Sanjive; Sharma, M. P. (February 1991). "Preparation of cesium trimolybdate by the thermal decomposition of a new oxomolybdenum(VI) oxalato complex" (in en). Journal of Thermal Analysis 37 (2): 427–432. doi:10.1007/BF02055943. ISSN 0368-4466. http://link.springer.com/10.1007/BF02055943.
- ↑ 12.00 12.01 12.02 12.03 12.04 12.05 12.06 12.07 12.08 12.09 12.10 Genčova, O.; Šiftar, J. (May 1995). "Synthesis and thermal characteristics of caesium oxalato-metallates of some rare earths" (in en). Journal of Thermal Analysis 44 (5): 1171–1176. doi:10.1007/BF02547547. ISSN 0368-4466. http://link.springer.com/10.1007/BF02547547.
- ↑ 13.0 13.1 Sengupta, A. K.; Bhaumik, B. B.; Nath, S. K. (June 1983). "Oxalatofluorotungstates (VI) & Oxalatotungstates (VI) of Some Alkali Metals & Complex Cations". Indian Journal of Chemistry 22A: 535–536. http://nopr.niscair.res.in/bitstream/123456789/49275/1/IJCA_22A%286%29_535-536.pdf.
- ↑ Hadadzadeh, Hassan; Rezvani, Ali Reza; Salehi Rad, Ali Reza; Khozeymeh, Elahe (August 2008). "A Novel Method for Preparation of Alumina-Supported Rhenium-Cesium Catalyst, Re-Cs/g-Al2O3". Iranian Journal of Chemistry and Chemical Engineering 27 (3). doi:10.30492/ijcce.2008.6965. https://doi.org/10.30492/ijcce.2008.6965.
- ↑ 15.0 15.1 Chernyaev, I. I.; Golovnya, V. A.; Shchelokov, R. N. (1960-07-01). "Aquo-Oxalato-Sulfate Compounds of Uranium" (in Russian). Zhur. Neorg. Khim. 5. https://www.osti.gov/biblio/4166316.
- ↑ Charushnikova, I. A.; Krot, N. N.; Polyakova, I. N. (May 2006). "Synthesis and crystal structure of double Np(V) cesium oxalate CsNpO2C2O4 · nH2O". Radiochemistry 48 (3): 223–226. doi:10.1134/S1066362206030039.
- ↑ Kazdobin, K. A.; Diamant, V. A.; Trachevskii, V. V.. MICROWAVE – ASSISTED SYNTHESIS OF BIS (OXALATO) ALKALI METAL BORATES. https://www.researchgate.net/publication/279929586.
 |
