Chemistry:Caesium hydride
 Caesium cation, Cs+ Hydrogen anion, H− | |
| Names | |
|---|---|
| IUPAC name
Caesium hydride
| |
| Other names
Cesium hydride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| CsH | |
| Molar mass | 133.91339 g/mol |
| Appearance | White or colorless crystals or powder[1] |
| Density | 3.42 g/cm3[1] |
| Melting point | ~170 °C (decomposes)[1] |
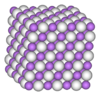
| Structure | |
| Face centered cubic | |
| Octahedral | |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Caesium hydride or cesium hydride is an inorganic compound of caesium and hydrogen with the chemical formula CsH. It is an alkali metal hydride. It was the first substance to be created by light-induced particle formation in metal vapor,[2] and showed promise in early studies of an ion propulsion system using caesium.[3] It is the most reactive stable alkaline metal hydride of all. It is a powerful superbase and reacts with water extremely vigorously.
The caesium nuclei in CsH can be hyperpolarized through interactions with an optically pumped caesium vapor in a process known as spin-exchange optical pumping (SEOP). SEOP can increase the nuclear magnetic resonance (NMR) signal of caesium nuclei by an order of magnitude.[4]
It is very difficult to make caesium hydride in a pure form. Caesium hydride can be produced by heating caesium carbonate and metallic magnesium in hydrogen at 580 to 620 °C.[5]
Crystal structure
At room temperature and atmospheric pressure, CsH has the same structure as NaCl.
References
- ↑ 1.0 1.1 1.2 Lide, D. R., ed (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. p. 4.57. ISBN 0-8493-0486-5.
- ↑ Tam, A.; Moe, G.; Happer, W. (1975). "Particle Formation by Resonant Laser Light in Alkali-Metal Vapor". Phys. Rev. Lett. 35 (24): 1630–33. doi:10.1103/PhysRevLett.35.1630. Bibcode: 1975PhRvL..35.1630T.
- ↑ Burkhart, J. A.; Smith, F. J. (November 1963). "Application of dynamic programming to optimizing the orbital control process of a 24-hour communications satellite". https://ntrs.nasa.gov/search.jsp?R=543868&id=2&qs=N%3D4294965816.
- ↑ Ishikawa, K.; Patton, B.; Jau, Y.-Y.; Happer, W. (2007). "Spin Transfer from an Optically Pumped Alkali Vapor to a Solid". Phys. Rev. Lett. 98 (18): 183004. doi:10.1103/PhysRevLett.98.183004. PMID 17501572. Bibcode: 2007PhRvL..98r3004I. https://zenodo.org/record/1059117.
- ↑ A. Jamieson Walker (1924). A Text Book Of Inorganic Chemistry Volume I The Alkali Metals And Their Congeners. https://archive.org/details/in.ernet.dli.2015.153684.
 |