Chemistry:Cyclopentadienyl allyl palladium
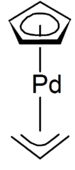
| |
| |
| Names | |
|---|---|
| Other names
(Cp)Pd(allyl)
| |
| Identifiers | |
| ChemSpider | |
PubChem CID
|
|
| Properties | |
| C8H10Pd | |
| Molar mass | 212.59 g·mol−1 |
| Appearance | Reddish needle-shaped solid |
| Melting point | 60 to 62 °C (140 to 144 °F; 333 to 335 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cyclopentadienyl allyl palladium is an organopalladium compound with formula (C5H5)Pd(C3H5). This reddish solid is volatile with an unpleasant odor. It is soluble in common organic solvents. The molecule consists of a Pd centre sandwiched between a Cp and allyl ligands.[1]
Preparation
This complex is produced by the reaction of allylpalladium chloride dimer with sodium cyclopentadienide:[1][2]
- 2 C5H5Na + (C3H5)2Pd2Cl2 → 2 (C5H4)Pd(C3H5) + 2 NaCl
Structure and reactions
The 18-electron complex adopts a half-sandwich structure with Cs symmetry, i.e., the molecule has a plane of symmetry. The complex can be decomposed readily by reductive elimination.
- C3H5PdC3H5 → Pd(0) + C5H5C3H5
The compound readily reacts with alkyl isocyanides to produce clusters with the approximate formula [Pd(CNR)2]n. It reacts with bulky alkyl phosphines to produce two-coordinated palladium(0) complexes:[3]
- CpPd(allyl) + 2 PR3 → Pd(PR3)2 + C5H5C3H5
The compound has been used to deposit thin film chemical vapor deposition of metallic palladium.[4][5]
References
- ↑ 1.0 1.1 Y. Tatsuno, T. Yoshida, S. Otsuka (1979). Shriver, Duward F.. ed. Inorganic Syntheses. 19. 220. doi:10.1002/9780470132500. ISBN 978-0-470-13250-0.
- ↑ R. B. King (1963). "Organometallic Chemistry of the Transition Metals. Organometallic Chemistry of the Transition Metals. III. Reactions between Sodium Cyclopentadienide and Certain Complex Transition Metal Halides". Inorg. Chem. 2 (3): 528. doi:10.1021/ic50007a026.
- ↑ Otsuka, S.; Yoshida, T.; Matsumoto, M.; Nakatsu, K. J. (1976). "Cyclohexyneplatinum(0) Complexes Containing Di-t-butylphenylphosphine, t-butyldiphenylphosphine or Trimethylphosphine". Australian Journal of Chemistry 45 (1): 135–142. doi:10.1071/CH9920135.
- ↑ Y. G Kim; S. Bialy; R.W. Miller; J. T. Spencer; Dowben A. Peter; Datta S. (1989). "Selective area deposition of conducting palladium films on polyimide resins". Mater. Res. Soc. Symp. Proc. 158: 103–7. doi:10.1557/PROC-158-103. http://digitalcommons.unl.edu/physicsdowben/150.
- ↑ K. Rajalingam; T. Strunskus; A. Terfort; R. A. Fischer; C. Wo (2008). "Metallization of a thiol-terminated organic surface using chemical vapor deposition". Langmuir 24 (15): 7886–7994. doi:10.1021/la8008927. PMID 18590295.
 |
