Chemistry:Palladium(II) nitrate

| |
| |
| Names | |
|---|---|
| IUPAC name
Palladium(II) nitrate
| |
| Other names
Palladium nitrate
Palladous nitrate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| Pd(NO3)2 | |
| Molar mass | 230.43 g/mol |
| Appearance | yellow solid |
| Density | 3.546 g/cm3[1] |
| Melting point | Decomposes >100 °C |
| Soluble | |
| Hazards | |
| Main hazards | Irritant, possibility of allergic reaction |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Palladium(II) chloride |
Other cations
|
Nickel(II) nitrate |
Related compounds
|
Silver nitrate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
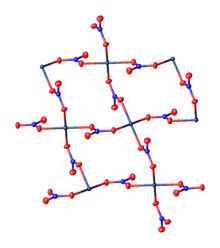
Palladium(II) nitrate is the inorganic compound with the formula Pd(NO3)2.(H2O)x where x = 0 or 2. The anhydrous and dihydrate are deliquescent solids. According to X-ray crystallography, both compounds feature square planar Pd(II) with unidentate nitrate ligands. The anhydrous compound, which is a coordination polymer, is yellow.[1][2]
As a solution in nitric acid, Pd(NO3)2 catalyzes the conversion of alkenes to dinitrate esters. Its pyrolysis affords palladium oxide.[3]
Preparation
Hydrated palladium nitrate may be prepared by dissolving palladium oxide hydrate in dilute nitric acid followed by crystallization. The nitrate crystallizes as yellow-brown deliquescent prisms. The anhydrous material is obtained by treating palladium metal with fuming nitric acid.[1]
References
- ↑ 1.0 1.1 1.2 Bruns, Jörn; Klüner, Thorsten; Wickleder, Mathias S. (2015). "Oxoanionic Noble Metal Compounds from Fuming Nitric Acid: The Palladium Examples Pd(NO3)2 and Pd(CH3SO3)2". Chemistry - A European Journal 21 (3): 1294–1301. doi:10.1002/chem.201405355. PMID 25431333.
- ↑ Laligant, Y.; Ferey, G.; Le Bail, A. (1991). "Crystal Structure of Pd(NO3)2(H2O)2". Materials Research Bulletin 26 (4): 269–275. doi:10.1016/0025-5408(91)90021-D.
- ↑ Timothy T. Wenzel "Palladium(II) Nitrate" Encyclopedia of Reagents for Organic Synthesis 2001, John Wiley & Sons. doi: 10.1002/047084289X.rp013
| HNO3 | He | ||||||||||||||||
| LiNO3 | Be(NO3)2 | B(NO3)−4 | C | NO−3, NH4NO3 |
O | FNO3 | Ne | ||||||||||
| NaNO3 | Mg(NO3)2 | Al(NO3)3 | Si | P | S | ClONO2 | Ar | ||||||||||
| KNO3 | Ca(NO3)2 | Sc(NO3)3 | Ti(NO3)4 | VO(NO3)3 | Cr(NO3)3 | Mn(NO3)2 | Fe(NO3)3, Fe(NO3)2 |
Co(NO3)2, Co(NO3)3 |
Ni(NO3)2 | Cu(NO3)2 | Zn(NO3)2 | Ga(NO3)3 | Ge | As | Se | Br | Kr |
| RbNO3 | Sr(NO3)2 | Y(NO3)3 | Zr(NO3)4 | Nb | Mo | Tc | Ru | Rh | Pd(NO3)2 | AgNO3 | Cd(NO3)2 | In | Sn | Sb(NO3)3 | Te | I | Xe(NO3)2 |
| CsNO3 | Ba(NO3)2 | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg2(NO3)2, Hg(NO3)2 |
Tl(NO3)3, TlNO3 |
Pb(NO3)2 | Bi(NO3)3 BiO(NO3) |
Po | At | Rn | |
| FrNO3 | Ra(NO3)2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La(NO3)3 | Ce(NO3)3, Ce(NO3)4 |
Pr | Nd(NO3)3 | Pm | Sm | Eu(NO3)3 | Gd(NO3)3 | Tb(NO3)3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac(NO3)3 | Th(NO3)4 | Pa | UO2(NO3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
 |
