Chemistry:Letts nitrile synthesis
| Letts nitrile synthesis | |
|---|---|
| Named after | Edmund A. Letts |
| Reaction type | Substitution reaction |
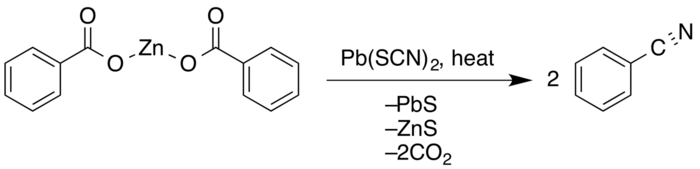
The Letts nitrile synthesis is a chemical reaction of aromatic carboxylic acids with metal thiocyanates to form nitriles. The reaction includes the loss of carbon dioxide and potassium hydrosulfide. The polar basic substitution reaction was discovered in 1872 by Edmund A. Letts.[1][2]

History
In 1857 Hugo Schiff observed that the reaction between benzoyl chloride with potassium cyanide produced the desired benzonitrile.[3] Work done later by British chemist Edmund A. Letts delved much deeper into the synthesis of nitriles. Attempting first to add cyano-groups to acetic acid, he obtained a mixture of acetamide and carbonyl sulfide. However, in 1872 he showed that treating a 2:1 molecular ratio of benzoic acid and potassium thiocyanate with heat for several hours also produced nitriles with only a small amount of amide with about 40% yield.[4]
G. Krüss expanded on Letts' work in 1884, producing better yields by utilizing lead(II) thiocyanate.[5] In 1916, E.E. Reid found that showed that dry distillation of the zinc(II) salt of the acid with a 20% excess of lead(II) thiocyanate gave an 86% conversion and 91% yield, almost double of that produced by Letts.[6]
Mechanism
Kekulé proposed the reaction mechanism in 1873.[7]
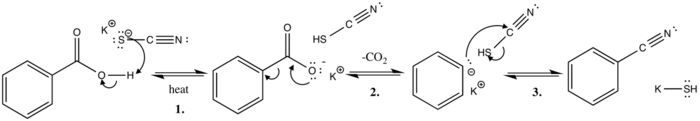
In this polar basic substitution reaction mechanism, thiocyanate ion extracts the acidic proton from benzoic acid while heated. This yields the conjugate base (stabilized by resonance structures) and thiocyanic acid.
The next step involves the evolution of carbon dioxide, where a lone pair of electrons moves from the negatively charged oxygen to form a double bond with the carboxylic carbon. The sigma bond between the ring and carboxyl group is then severed, the electron pair moving to the ring and delocalized through resonance structures.
The final step of the mechanism involves the attack of the phenyl anion attacking the cyano-carbon, pushing the electron pair over to the sulfur, which readily diffuses the negative charge and is further stabilized by the potassium ion, resulting in the final benzonitrile product and potassium hydrosulfide.
Applications
Aromatic nitriles have a few applications, including polyrecombination to form polymers,[8] are sometimes studied as biologically active molecules[9] and undergoing Ritter reactions to form amides.[10]
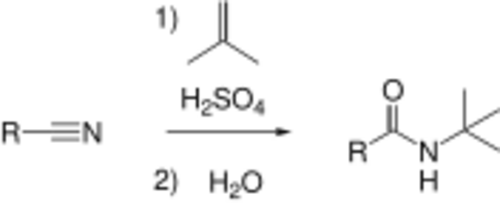
Benzonitrile, the original product of Letts, has multiple uses as a versatile reagent and as a solvent. Substituted benzonitriles are important in many fields including pharmaceuticals. Benzonitrile is a precursor in the synthesis of Fadrozole, an aromatase inhibitor used in the treatment of breast cancer.[11] 4-(trifluoromethyl)benzonitrile, produced by the Nickel catalyzed cyanation of 4-chlorobenzotrifluoride is a precursor for the antidepressant Fluvoxamine.[12]
Benzonitrile can also act a ligand in asymmetric catalysis, coordinating to transition metals and forming Lewis acids.[13][14]
See also
For synthesis of nitriles:
- Kolbe nitrile synthesis
- Rosenmund-von Braun reaction
For reactions of nitriles:
References
- ↑ Letts, E. A. (1872). "Neue Bildungsweisen der Amide und Nitrile". Chemische Berichte 5 (2): 669–674. doi:10.1002/cber.18720050228. https://zenodo.org/record/1425036.
- ↑ Letts, E. A. (1872). "New Method for Producing Amides and Nitriles". Proceedings of the Royal Society of London 21 (139–147): 61–66. doi:10.1098/rspl.1872.0012. ISSN 0370-1662. Bibcode: 1872RSPS...21...61L.
- ↑ Schiff, Hugo (1857). "Ueber einige Derivate des Naphtylamins". Annalen der Chemie und Pharmacie 101 (1): 90–93. doi:10.1002/jlac.18571010113. https://zenodo.org/record/1427088.
- ↑ Reid, E.E. (1910). "Studies in the preparation of Nitriles.". Am. Chem. J. 43: 162–181.
- ↑ Krüss, G. (1884). "Ueber eine neue Darstellungsmethode für Nitrile". Ber. 17 (2): 1766–1768. doi:10.1002/cber.18840170245. https://zenodo.org/record/1425371.
- ↑ Mowry, D.T. The Preparation of Nitriles. (1948). "The preparation of nitriles". Chem. Rev. 42 (2): 189–283. doi:10.1021/cr60132a001. PMID 18914000.
- ↑ Kekulé, A. (1872). "Neue Bildungsweisen der Amide und Nitrile". Ber. 5 (2): 669–674. doi:10.1002/cber.18720050228. https://zenodo.org/record/1425036.
- ↑ Vasnev, V.A.; Sosin, S.L.; Korshak, V.V. (1964). "Synthesis of polymers by polyrecombination of the nitrile of aromatic and aliphatic acids". Polymer Science U.S.S.R 6 (5): 928. doi:10.1016/0032-3950(64)90510-6.
- ↑ Zhao, Aimin; Li, Whenzong; Yang, Huifung (1993). "Microbial Conversion of aromatic nitriles". Weis. Tong. 20: 169–171.
- ↑ Sadeghi, Bahareh; Farahzadi, Ebrahim; Hassanabadi, Alireza (2012-01-01). "KAl(SO4)2.12H2O as an eco-friendly and reusable catalyst for the synthesis of amides by the Ritter reaction". Journal of Chemical Research 36 (9): 539–540. doi:10.3184/174751912x13418518739562.
- ↑ Raats, J. I.; Falkson, G.; Falkson, H. C. (1992). "A study of fadrozole, a new aromatase inhibitor, in postmenopausal women with advanced metastatic breast cancer". Journal of Clinical Oncology 10 (1): 111–116. doi:10.1200/jco.1992.10.1.111. ISSN 0732-183X. PMID 1530798.
- ↑ Schareina, Thomas; Zapf, Alexander; Beller, Matthias (2004-01-01). "Improving palladium-catalyzed cyanation of aryl halides: development of a state-of-the-art methodology using potassium hexacyanoferrate(II) as cyanating agent". Journal of Organometallic Chemistry 689 (24): 4576–4583. doi:10.1016/j.jorganchem.2004.08.020.
- ↑ Becker, Jennifer J.; Orden, Lori J. Van; White, Peter S.; Gagné, Michel R. (2002-01-01). "Electron-Poor Benzonitriles as Labile, Stabilizing Ligands in Asymmetric Catalysis". Organic Letters 4 (5): 727–730. doi:10.1021/ol017218q. PMID 11869112.
- ↑ Anderson, Gordon K.; Lin, Minren (1990). "Bis(Benzonitrile)Dichloro Complexes of Palladium and Platinum". Inorganic Syntheses. 28. 60–63. doi:10.1002/9780470132593.ch13. ISBN 9780470132593.
 |
