Chemistry:Thiocyanic acid
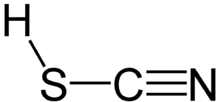
| |
 | |
| Names | |
|---|---|
| IUPAC name
Thiocyanic acid[2]
| |
| Other names | |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 25178 | |
| KEGG | |
| MeSH | thiocyanic+acid |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| HSCN | |
| Molar mass | 59.09 g·mol−1 |
| Appearance | Colorless oily liquid, white or colourless gas, white solid[2] |
| Odor | Pungent |
| Density | 2.04 g/cm3 |
| Melting point | 5 °C (41 °F; 278 K) [2] |
| Miscible | |
| Solubility | Soluble in ethanol, diethyl ether |
| log P | 0.429 |
| Vapor pressure | 4.73 mmHg (631 Pa)[2] |
| Acidity (pKa) | 0.926 |
| Basicity (pKb) | 13.071 |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H302, H312, H332, H412 | |
| P261, P264, P270, P271, P273, P280, P301+312, P302+352, P304+312, P304+340, P312, P322, P330, P363, P501 | |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Thiocyanic acid is a chemical compound with the formula HSCN and structure H–S–C≡N, which exists as a tautomer with isothiocyanic acid (H–N=C=S).[4] The isothiocyanic acid tautomer tends to dominate with the compound being about 95% isothiocyanic acid in the vapor phase.[5]
It is a moderately strong acid,[6] with a pKa of 1.1 at 20 °C and extrapolated to zero ionic strength.[7]
One of the thiocyanic acid tautomers, HSCN, is predicted to have a triple bond between carbon and nitrogen. Thiocyanic acid has been observed spectroscopically.[8]
The salts and esters of thiocyanic acid are known as thiocyanates. The salts are composed of the thiocyanate ion ([SCN]−
) and a suitable cation (e.g., potassium thiocyanate, KSCN). The esters of thiocyanic acid have the general structure R–S–C≡N, where R stands for an organyl group.
Isothiocyanic acid, HNCS, is a Lewis acid whose free energy, enthalpy and entropy changes for its 1:1 association with a variety of Lewis bases in carbon tetrachloride solution at 25 °C have been reported.[9] HNCS acceptor properties are discussed in the ECW model. The salts are composed of the thiocyanate ion ([SCN]−
) and a suitable cation (e.g., ammonium thiocyanate, [NH
4]+
[SCN]−
). Isothiocyanic acid forms isothiocyanates R–N=C=S, where R stands for an organyl group.
References
- ↑ Merck Index, 11th Edition, 9257.
- ↑ 2.0 2.1 2.2 2.3 2.4 https://pubchem.ncbi.nlm.nih.gov/compound/Thiocyanic-acid
- ↑ Richter, Victor von; Spielmann, Percy E., trans. (1922). Organic Chemistry or Chemistry of the Carbon Compounds. 1. Philadelphia, Pennsylvania, U.S.A.: P. Blakiston's Son & Co.. p. 466. https://books.google.com/books?id=WV0vAQAAMAAJ&pg=PA466.
- ↑ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
- ↑ Beard, C. I.; Dailey, B. P. (1950). "The Structure and Dipole Moment of Isothiocyanic Acid". The Journal of Chemical Physics 18 (11): 1437. doi:10.1063/1.1747507. Bibcode: 1950JChPh..18.1437B. https://dspace.mit.edu/bitstream/1721.1/4934/1/RLE-TR-112-04711196.pdf.
- ↑ Munegumi, Toratane (23 January 2013). "Where is the Border Line between Strong Acids and Weak Acids?". World Journal of Chemical Education 1 (1): 12–16. http://pubs.sciepub.com/wjce/1/1/4/index.html.
- ↑ Martell, A. E.; Smith, R. M.; Motelaitis, R. J. (2001). NIST Database 46. Gaithersburg, MD: National Institute of Standards and Technology.
- ↑ Wierzejewska, M.; Mielke, Z. (2001). "Photolysis of Isothiocyanic Acid HNCS in Low-Temperature Matrices. Infrared Detection of HSCN and HSNC Isomers". Chemical Physics Letters 349 (3–4): 227–234. doi:10.1016/S0009-2614(01)01180-0. Bibcode: 2001CPL...349..227W.
- ↑ . Barakat, T. M.; Nelson, J.; Nelson, S. M.; Pullin, A. D. E. (1969.) “Spectra and hydrogen-bonding of characteristics of thiocyanic acid. Part 4.—Association with weak proton acceptors”. Trans. Faraday Soc., 1969,65, 41-51
 |



