Chemistry:Sunscreen
| Sunscreen | |
|---|---|
 Sunscreen on back under normal and UV light | |
| Other names | Sun screen, sunblock, sunburn cream, sun cream, block out[1] |
Sunscreen, also known as sunblock[lower-alpha 1] or sun cream, is a photoprotective topical product for the skin that helps protect against sunburn and most importantly prevent skin cancer. Sunscreens come as lotions, sprays, gels, foams (such as an expanded foam lotion or whipped lotion[4]), sticks, powders and other topical products. Sunscreens are common supplements to clothing, particularly sunglasses, sunhats and special sun protective clothing, and other forms of photoprotection (such as umbrellas).
Sunscreens might be classified according to the type of active ingredient(s) present in the formulation (inorganic compounds or organic molecules) as:
- Mineral sunscreens (also referred to as physical), which use only inorganic compounds (zinc oxide and/or titanium dioxide) as active ingredients. These ingredients primarily work by absorbing UV rays but also through reflection and refraction.[5][6]
- Chemical sunscreens, which use organic molecules as active ingredients. These products are sometimes referred to as petrochemical sunscreens since the active organic molecules are synthesized starting from building blocks typically derived from petroleum.[7] Chemical sunscreen ingredients also mainly work by absorbing the UV rays.[8] Over the years, some organic UV absorbers have been heavily scrutinised to assess their toxicity[9] and a few of them have been banned in places such as Hawaii[10] and Thailand[11] for their impact on aquatic life and the environment.
- Hybrid sunscreens, which contain a combination of organic and inorganic UV filters.
Medical organizations such as the American Cancer Society recommend the use of sunscreen because it aids in the prevention of squamous cell carcinomas.[12] The routine use of sunscreens may also reduce the risk of melanoma.[13] To effectively protect against all the potential damages of UV light, the use of broad-spectrum sunscreens (covering both UVA and UVB radiation) has been recommended.[3]
As of 2021, only zinc oxide and titanium dioxide are generally recognized as safe and effective (GRASE) by the U.S. Food and Drug Administration (FDA),[14] as currently there is insufficient data to support recognizing petrochemical UV filters as safe.
History


Early civilizations used a variety of plant products to help protect the skin from sun damage. For example, ancient Greeks used olive oil for this purpose, and ancient Egyptians used extracts of rice, jasmine, and lupine plants whose products are still used in skin care today.[15] Zinc oxide paste has also been popular for skin protection for thousands of years.[16] Among the nomadic sea-going Sama-Bajau people of the Philippines , Malaysia, and Indonesia, a common type of sun protection is a paste called borak or burak, which was made from water weeds, rice and spices. It is used most commonly by women to protect the face and exposed skin areas from the harsh tropical sun at sea.[17] In Myanmar, thanaka, a yellow-white cosmetic paste made of ground bark, is traditionally used for sun protection. In Madagascar , a ground wood paste called masonjoany has been worn for sun protection, as well as decoration and insect repellent, since the 18th century, and is ubiquitous in the Northwest coastal regions of the island to this day.[18][19]
The first ultraviolet B filters were produced in 1928.[20] Followed by the first sunscreen, invented in Australia by chemist H.A. Milton Blake, in 1932[21] formulating with the UV filter 'salol' (Phenyl salicylate) at a concentration of 10%.[22] Its protection was verified by the University of Adelaide.[23][24] In 1936, L'Oreal released its first sunscreen product, formulated by French chemist Eugène Schueller.[21]
Early adopters of sunscreen were the US military. In 1944, as the hazards of sun overexposure became apparent to soldiers stationed in the Pacific tropics at the height of World War II,[25][21][26][27] Benjamin Green, an airman and later a pharmacist produced Red Vet Pet (for red veterinary petrolatum) for the US military. Sales boomed when Coppertone improved and commercialized the substance under the Coppertone girl and Bain de Soleil branding in the early 1950s. In 1946, Austrian chemist Franz Greiter introduced a product, called Gletscher Crème (Glacier Cream), subsequently became the basis for the company Piz Buin, named in honor of the mountain where Greiter allegedly received the sunburn.[28][29][30]
In 1974, Greiter adapted earlier calculations from Friedrich Ellinger and Rudolf Schulze and introduced the "sun protection factor" (SPF), which has become the global standard for measuring UVB protection.[25][31] It has been estimated that Gletscher Crème had an SPF of 2.
Water-resistant sunscreens were introduced in 1977,[21] and recent development efforts have focused on overcoming later concerns by making sunscreen protection both longer-lasting and broader-spectrum (protection from both UVA & UVB rays), more environmentally friendly,[32] more appealing to use[25] and addressing the safety concerns of petrochemical sunscreens, i.e. FDA studies showing their systematic absorption into the bloodstream.[33]
Health effects
Benefits
Sunscreen use can help prevent melanoma[34][35][36] and squamous cell carcinoma, two types of skin cancer.[37] There is little evidence that it is effective in preventing basal cell carcinoma.[38]
A 2013 study concluded that the diligent, everyday application of sunscreen could slow or temporarily prevent the development of wrinkles and sagging skin.[39] The study involved 900 white people in Australia and required some of them to apply a broad-spectrum sunscreen every day for four and a half years. It found that people who did so had noticeably more resilient and smoother skin than those assigned to continue their usual practices.[39] A study on 32 subjects showed that daily use of sunscreen (SPF 30) reversed photoaging of the skin within 12 weeks and the amelioration continued until the end of the investigation period of one year.[40] Sunscreen is inherently anti-ageing as the sun is the number one cause of premature ageing, it therefore may slow or temporarily prevent the development of wrinkles, dark spots and sagging skin.

Minimizing UV damage is especially important for children and fair-skinned individuals and those who have sun sensitivity for medical reasons.[41]
Risks
In February 2019, the US Food and Drug Administration (FDA) started classifying already approved UV filter molecules into three categories: those which are generally recognized as safe and effective (GRASE), those which are non-GRASE due to safety issues, and those requiring further evaluation.[42] As of 2021, only zinc oxide and titanium dioxide are recognized as GRASE.[43] Two previously approved UV filters, para-aminobenzoic acid (PABA) and trolamine salicylate, were banned in 2021 due to safety concerns. The remaining FDA approved active ingredients were put in the third category as their manufacturers have yet to produce sufficient safety data — despite the fact that some of the chemicals have sold in sunscreen products for more than 40 years.[7] Some researchers argue that the risk of sun-induced skin cancer outweighs concerns about toxicity and mutagenicity,[44][45] although environmentalists say this ignores "ample safer alternatives available on the market containing the active ingredient minerals zinc oxide or titanium dioxide" which are also safer for the environment.[46]
Regulators can investigate and ban UV filters over safety concerns (such as PABA), which can result in withdrawal of products from the consumer market.[25][47] Regulators, such as the TGA and the FDA, have also been concerned with recent reports of contamination in sunscreen products with known possible human carcinogens such as benzene and benzophenone.[48] Independent laboratory testing carried out by Valisure found benzene contamination 27% of the sunscreens they tested, with some batches having up to triple the FDA's conditionally restricted limit of 2 parts per million (ppm).[49] This resulted in a voluntary recall by some major sunscreen brands that were implicated in the testing, as such, regulators also help publicise and coordinate these voluntary recalls.[50] VOC's (Volatile Organic Compounds) such as benzene, are particularly harmful in sunscreen formulations as many active and inactive ingredients can increase permeation across the skin.[51] Butane, which is used as a propellant in spray sunscreens, has been found to have benzene impurities from the refinement process.[52]
Recent research by the FDA on six common petrochemical UV filters (avobenzone, oxybenzone, octocrylene, homosalate, octisalate, and octinoxate) found that they could be detected on the skin, in blood, in breast milk and in urine samples weeks after a single use.[53][54]
There is a risk of an allergic reaction to sunscreen for some individuals, as "Typical allergic contact dermatitis may occur in individuals allergic to any of the ingredients that are found in sunscreen products or cosmetic preparations that have a sunscreen component. The rash can occur anywhere on the body where the substance has been applied and sometimes may spread to unexpected sites."[55]
Vitamin D production
There are some concerns about potential vitamin D deficiency arising from prolonged use of sunscreen.[56][57] The typical use of sunscreen does not usually result in vitamin D deficiency; however, extensive usage may.[58] Sunscreen prevents ultraviolet light from reaching the skin, and even moderate protection can substantially reduce vitamin D synthesis.[59][60] However, adequate amounts of vitamin D can be obtained via diet or supplements.[61] Vitamin D overdose is impossible from UV exposure due to an equilibrium the skin reaches in which vitamin D degrades as quickly as it is created.[62][63][64]
Studies have shown that sunscreen with a high UVA protection factor enabled significantly higher vitamin D synthesis than a low UVA protection factor sunscreen, likely because it allows more UVB transmission.[65][66]
Measurements of protection
Sun protection factor and labeling

The sun protection factor (SPF rating, introduced in 1974) is a measure of the fraction of sunburn-producing UV rays that reach the skin. For example, "SPF 15" means that 1⁄15 of the burning radiation will reach the skin, assuming sunscreen is applied evenly at a thick dosage of 2 milligrams per square centimeter[67] (mg/cm2). It is important to note that sunscreens with higher SPF do not last or remain effective on the skin any longer than lower SPF and must be continually reapplied as directed, usually every two hours.[68]
The SPF is an imperfect measure of skin damage because invisible damage and skin malignant melanomas are also caused by ultraviolet A (UVA, wavelengths 315–400 or 320–400 nm), which does not primarily cause reddening or pain. Conventional sunscreen blocks very little UVA radiation relative to the nominal SPF; broad-spectrum sunscreens are designed to protect against both UVB and UVA.[69][70][71] According to a 2004 study, UVA also causes DNA damage to cells deep within the skin, increasing the risk of malignant melanomas.[72] Even some products labeled "broad-spectrum UVA/UVB protection" have not always provided good protection against UVA rays.[73] Titanium dioxide probably gives good protection, but does not completely cover the UVA spectrum, as early 2000s research suggests that zinc oxide is superior to titanium dioxide at wavelengths 340–380 nm.[74]
Owing to consumer confusion over the real degree and duration of protection offered, labelling restrictions are enforced in several countries. In the EU, sunscreen labels can only go up to SPF 50+ (initially listed as 30 but soon revised to 50).[75] Australia 's Therapeutic Goods Administration increased the upper limit to 50+ in 2012.[76][77] In its 2007 and 2011 draft rules, the US Food and Drug Administration (FDA) proposed a maximum SPF label of 50, to limit unrealistic claims.[78][3][79] (As of February 2017, the FDA has not adopted the SPF 50 limit.[80]) Others have proposed restricting the active ingredients to an SPF of no more than 50, due to lack of evidence that higher dosages provide more meaningful protection.[81] Different sunscreen ingredients have different effectiveness against UVA and UVB.[82]
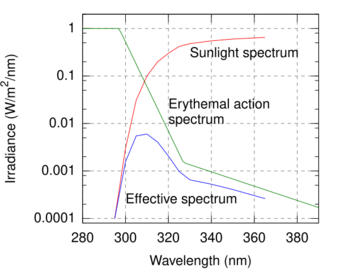
The SPF can be measured by applying sunscreen to the skin of a volunteer and measuring how long it takes before sunburn occurs when exposed to an artificial sunlight source. In the US, such an in vivo test is required by the FDA. It can also be measured in vitro with the help of a specially designed spectrometer. In this case, the actual transmittance of the sunscreen is measured, along with the degradation of the product due to being exposed to sunlight. In this case, the transmittance of the sunscreen must be measured over all wavelengths in sunlight's UVB–UVA range (290–400 nm), along with a table of how effective various wavelengths are in causing sunburn (the erythemal action spectrum) and the standard intensity spectrum of sunlight (see the figure). Such in vitro measurements agree very well with in vivo measurements.[attribution needed]
Numerous methods have been devised for evaluation of UVA and UVB protection. The most-reliable spectrophotochemical methods eliminate the subjective nature of grading erythema.[83]
The ultraviolet protection factor (UPF) is a similar scale developed for rating fabrics for sun protective clothing. According to recent testing by Consumer Reports, UPF ~30+ is typical for protective fabrics, while UPF ~20 is typical for standard summer fabrics.[84]
Mathematically, the SPF (or the UPF) is calculated from measured data as:[citation needed]
where is the solar irradiance spectrum, the erythemal action spectrum, and the monochromatic protection factor, all functions of the wavelength . The MPF is roughly the inverse of the transmittance at a given wavelength.[citation needed]
The above means that the SPF is not simply the inverse of the transmittance in the UVB region. If that were true, then applying two layers of SPF 5 sunscreen would always be equivalent to SPF 25 (5 times 5). The actual combined SPF may be lower than the square of the single-layer SPF.[85]
UVA protection
Persistent pigment darkening
The persistent pigment darkening (PPD) method is a method of measuring UVA protection, similar to the SPF method of measuring sunburn protection. Originally developed in Japan, it is the preferred method used by manufacturers such as L'Oréal.
Instead of measuring erythema, the PPD method uses UVA radiation to cause a persistent darkening or tanning of the skin. Theoretically, a sunscreen with a PPD rating of 10 should allow a person 10 times as much UVA exposure as would be without protection. The PPD method is an in vivo test like SPF. In addition, the European Cosmetic and Perfumery Association (Colipa) has introduced a method that, it is claimed, can measure this in vitro and provide parity with the PPD method.[86]
SPF equivalence


As part of revised guidelines for sunscreens in the EU, there is a requirement to provide the consumer with a minimum level of UVA protection in relation to the SPF. This should be a UVA protection factor of at least 1/3 of the SPF to carry the UVA seal.[87] The 1/3 threshold derives from the European Commission recommendation 2006/647/EC.[88] This Commission recommendation specifies that the UVA protection factor should be measured using the PPD method as modified by the French health agency AFSSAPS (now ANSM) "or an equivalent degree of protection obtained with any in vitro method".[89]
A set of final US FDA rules effective from summer 2012 defines the phrase "broad spectrum" as providing UVA protection proportional to the UVB protection, using a standardized testing method.[3]
Star rating system
In the UK and Ireland, the Boots star rating system is a proprietary in vitro method used to describe the ratio of UVA to UVB protection offered by sunscreen creams and sprays. Based on original work by Brian Diffey at Newcastle University, the Boots Company in Nottingham, UK, developed a method that has been widely adopted by companies marketing these products in the UK.
One-star products provide the lowest ratio of UVA protection, five-star products the highest. The method was revised in light of the Colipa UVA PF test and the revised EU recommendations regarding UVA PF. The method still uses a spectrophotometer to measure absorption of UVA versus UVB; the difference stems from a requirement to pre-irradiate samples (where this was not previously required) to give a better indication of UVA protection and photostability when the product is used. With the current methodology, the lowest rating is three stars, the highest being five stars.
In August 2007, the FDA put out for consultation the proposal that a version of this protocol be used to inform users of American product of the protection that it gives against UVA;[78] but this was not adopted, for fear it would be too confusing.[81]
PA system
Asian brands, particularly Japanese ones, tend to use The Protection Grade of UVA (PA) system to measure the UVA protection that a sunscreen provides. The PA system is based on the PPD reaction and is now widely adopted on the labels of sunscreens. According to the Japan Cosmetic Industry Association, PA+ corresponds to a UVA protection factor between two and four, PA++ between four and eight, and PA+++ more than eight. This system was revised in 2013 to include PA++++ which corresponds to a PPD rating of sixteen or above.
Expiration Date
Some sunscreens include an expiration date—a date indicating when they may become less effective.[90]
Active ingredients
Sunscreen formulations contain UV absorbing compounds (the active ingredients) dissolved or dispersed in a mixture of other ingredients, such as water, oils, moisturizers, and antioxidants. The UV filters can be either:
- Organic compounds which absorb ultraviolet light.[91] Some organic compounds (bisoctrizole and phenylene bis-diphenyltriazine) also partially reflect incident light.[92] These are also referred to as "chemical" UV filters.
- Inorganic compounds (zinc oxide and titanium dioxide) which reflect, scatter, and absorb UV light.[93] These are also referred to as "mineral" filters.
The organic compounds used as UV filter are often aromatic molecules conjugated with carbonyl groups. This general structure allows the molecule to absorb high-energy ultraviolet rays and release the energy as lower-energy rays, thereby preventing the skin-damaging ultraviolet rays from reaching the skin. So, upon exposure to UV light, most of the ingredients (with the notable exception of avobenzone) do not undergo significant chemical change, allowing these ingredients to retain the UV-absorbing potency without significant photodegradation.[94] A chemical stabilizer is included in some sunscreens containing avobenzone to slow its breakdown. The stability of avobenzone can also be improved by bemotrizinol,[95] octocrylene[96] and various other photostabilisers. Most organic compounds in sunscreens slowly degrade and become less effective over the course of several years even if stored properly, resulting in the expiration dates calculated for the product.[97]
Sunscreening agents are used in some hair care products such as shampoos, conditioners and styling agents to protect against protein degradation and color loss. Currently, benzophenone-4 and ethylhexyl methoxycinnamate are the two sunscreens most commonly used in hair products. The common sunscreens used on skin are rarely used for hair products due to their texture and weight effects.
UV filters need usually to be approved by local agencies (such as the FDA in the United States) to be used in sunscreen formulations. As of 2023, 29 compounds are approved in the European Union and 17 in the USA.[92] No UV filters have been approved by the FDA for use in cosmetics since 1999.
The following are the FDA allowable active ingredients in sunscreens:
| UV-filter | Other names | Maximum concentration | Known permitting jurisdictions | Results of safety testing | UVA | UVB |
|---|---|---|---|---|---|---|
| p-Aminobenzoic acid | PABA | 15% (USA), (EU: banned from sale to consumers from 8 October 2009) | USA, AUS | Protects against skin tumors in mice.[98][99][100] Shown to increase DNA defects, and not generally recognised as safe and effective according to the FDA[101] | X | |
| Padimate O | OD-PABA, octyldimethyl-PABA, σ-PABA | 8% (USA, AUS) 10% (JP)
(Not currently supported in EU and may be delisted) |
EU, USA, AUS, JP | X | ||
| Phenylbenzimidazole sulfonic acid | Ensulizole, PBSA | 4% (USA, AUS) 8% (EU) 3% (JP) | EU, USA, AUS, JP | Genotoxic in bacteria[102] | X | |
| Cinoxate | 2-Ethoxyethyl p-methoxycinnamate | 3% (USA) 6% (AUS) | USA, AUS | X | X | |
| Dioxybenzone | Benzophenone-8 | 3% (USA) | USA, AUS | X | X | |
| Oxybenzone | Benzophenone-3 | 6% (USA), 2.2% (body) / 6% (face) EU,[103] 10% AUS, | EU, USA, AUS | Banned in Hawaii since 2018[10] - "harmful to coral reefs, fish, and other ocean life"[104] | X | X |
| Homosalate | Homomethyl salicylate | 7.34% (EU) 15% (USA, AUS) | EU, USA, AUS | X | ||
| Menthyl anthranilate | Meradimate | 5% (USA) | USA, AUS | X | ||
| Octocrylene | Eusolex OCR, Parsol 340, 2-Cyano-3,3-diphenyl acrylic acid, 2-ethylhexylester | 10% (USA) | EU, USA, AUS | Increases reactive oxygen species (ROS)[105] | X | X |
| Octinoxate | Octyl-methoxycinnamate, Ethylhexyl methoxycinnamate, 2-Ethylhexyl-paramethoxycinnamate | 7.5% (USA) 10% (EU, AUS) 20% (JP) | EU, USA, AUS, JP | Banned in Hawaii since 2021 - harmful to coral[106] | X | |
| Octyl salicylate | Octisalate, 2-Ethylhexyl salicylate | 5% (EU, USA, AUS) 10% (JP) | EU, USA, AUS, JP | X | ||
| Sulisobenzone | 2-Hydroxy-4-Methoxybenzophenone-5-sulfonic acid, 3-Benzoyl-4-hydroxy-6-methoxybenzenesulfonic acid, Benzophenone-4 | 5% (EU) 10% (USA, AUS, JP) | EU, USA, AUS, JP | X | X | |
| Avobenzone | 1-(4-methoxyphenyl)-3-(4-tert-butyl phenyl)propane-1,3-dione, Butyl methoxy dibenzoylmethane, |
3% (USA) 5% (EU, AUS) | EU, USA, AUS | X | ||
| Ecamsule | Terephthalylidene Dicamphor Sulfonic Acid | 10% | EU, AUS (US: approved in certain formulations up to 3% via New Drug Application (NDA) Route) | Protects against skin tumors in mice[107][108][109] | X | |
| Titanium dioxide | CI77891, TiO₂ | 25% (US) No limit (JP) | EU, USA, AUS, JP | Generally recognized as safe and effective by the FDA[101] | X | |
| Zinc oxide | CI77947, ZnO | 25% (US) No limit (AUS, JP) | EU, USA, AUS, JP | Generally recognized as safe and effective by the FDA.[101] Protects against skin tumors in mice[107] | X | X |
Zinc oxide was approved as a UV filter by the EU in 2016.[110]
Other ingredients approved within the EU[111] and other parts of the world,[112] that have not been included in the current FDA Monograph:
| UV-filter | Other names | Maximum concentration | Permitted in | Results of safety testing | UVA | UVB |
|---|---|---|---|---|---|---|
| 4-Methylbenzylidene camphor | Enzacamene, MBC | 4%* | EU, AUS | |||
| Bisoctrizole | Methylene Bis-Benzotriazolyl Tetramethylbutylphenol, MBBT | 10%* | EU, AUS, JP | |||
| Bemotrizinol | Bis-ethylhexyloxyphenol methoxyphenol triazine, BEMT, anisotriazine | 10% (EU, AUS) 3% (JP)* | EU, AUS, JP | |||
| Tris-biphenyl triazine | 10% | EU | ||||
| Trolamine salicylate | Triethanolamine salicylate | 12% | AUS | Not generally recognised as safe and effective according to the FDA.[101] Removed from the market in the USA.[113] | X | |
| Drometrizole trisiloxane | 15% | EU, AUS | ||||
| Benzophenone-9 | CAS 3121-60-6, Sodium Dihydroxy Dimethoxy Disulfobenzophenone [114] | 10% | JP | |||
| Ethylhexyl triazone | octyl triazone, EHT | 5% (EU, AUS) 3% (JP)* | EU, AUS | |||
| Diethylamino hydroxybenzoyl hexyl benzoate | 10% (EU, JP) | EU, JP | ||||
| Iscotrizinol | diethylhexyl butamido triazone, DBT | 10% (EU) 5% (JP)* | EU, JP | |||
| Polysilicone-15 | Dimethico-diethylbenzalmalonate | 10% | EU, AUS, JP | |||
| Amiloxate | Isopentyl-4-methoxycinnamate, Isoamyl p-Methoxycinnamate, IMC | 10%* | EU, AUS |
* Time and Extent Application (TEA), Proposed Rule on FDA approval originally expected 2009, now expected 2015.[needs update]
Many of the ingredients awaiting approval by the FDA are relatively new, and developed to absorb UVA.[115] The 2014 Sunscreen Innovation Act was passed to accelerate the FDA approval process.[116][117]
Inactive ingredients
It is known that SPF is affected by not only the choice of active ingredients and the percentage of active ingredients but also the formulation of the vehicle/base. Final SPF is also impacted by the distribution of active ingredients in the sunscreen, how evenly the sunscreen applies on the skin, how well it dries down on the skin and the pH value of the product among other factors. Changing any inactive ingredient may potentially alter a sunscreen's SPF.[118][119]
When combined with UV filters, added antioxidants can work synergistically to affect the overall SPF value positively. Furthermore, adding antioxidants to sunscreen can amplify its ability to reduce markers of extrinsic photoaging, grant better protection from UV induced pigment formation, mitigate skin lipid peroxidation, improve the photostability of the active ingredients, neutralize reactive oxygen species formed by irradiated photocatalysts (e.g., uncoated TiO₂) and aid in DNA repair post-UVB damage, thus enhancing the efficiency and safety of sunscreens.[120][121][122][123] Compared with sunscreen alone, it has been shown that the addition of antioxidants has the potential to suppress ROS formation by an additional 1.7-fold for SPF 4 sunscreens and 2.4-fold for SPF 15-to-SPF 50 sunscreens, but the efficacy depends on how well the sunscreen in question has been formulated.[124] Sometimes osmolytes are also incorporated into commercially available sunscreens in addition to antioxidants since they also aid in protecting the skin from the detrimental effects of UVR.[125] Examples include the osmolyte taurine, which has demonstrated the ability to protects against UVB-radiation induced immunosuppression[126] and the osmolyte ectoine, which aids in counteracting cellular accelerated aging & UVA-radiation induced premature photoaging.[127]
Other inactive ingredients can also assist in photostabilizing unstable UV filters. Cyclodextrins have demonstrated the ability to reduce photodecomposition, protect antioxidants and limit skin penetration past the uppermost skin layers, allowing them to longer maintain the protection factor of sunscreens with UV filters that are highly unstable and/or easily permeate to the lower layers of the skin.[128][129][121] Similarly, film-forming polymers like polyester-8 and polycryleneS1 have the ability to protect the efficacy of older petrochemical UV filters by preventing them from destabilizing due to extended light exposure. These kinds of ingredients also increase the water resistance of sunscreen formulations.[130][131]

In the 2010s & 2020s, there has been increasing interest in sunscreens that protect the wearer from the sun's high-energy visible light & iinfrared light as well as ultraviolet light. This is due to newer research revealing blue & violet visible light and certain wavelengths of infrared light (e.g., NIR, IR-A) work synergistically with UV light in contributing to oxidative stress, free radical generation, dermal cellular damage, suppressed skin healing, decreased immunity, erythema, inflammation, dryness, and several aesthetic concerns, such as: wrinkle formation, loss of skin elasticity and dyspigmentation.[132][133][134][135][136][137][138] Increasingly, a number of commercial sunscreens are being produced that have manufacturer claims regarding skin protection from blue light, infrared light and even air pollution.[138] However, as of 2021 there are no regulatory guidelines or mandatory testing protocols that govern these claims.[124] Historically, the American FDA has only recognized protection from sunburn (via UVB protection) and protection from skin cancer (via SPF 15+ with some UVA protection) as drug/medicinal sunscreen claims, so they do not have regulatory authority over sunscreen claims regarding protecting the skin from damage from these other environmental stressors.[139] Since sunscreen claims not related to protection from ultraviolet light are treated as cosmeceutical claims rather than drug/medicinal claims, the innovative technologies and additive ingredients used to allegedly reduce the damage from these other environmental stressors may vary widely from brand to brand.
Some studies show that mineral sunscreens primarily made with substantially large particles (i.e., neither nano nor micronized) may help protect from visible and infrared light to some degree,[138][124][140] but these sunscreens are often unacceptable to consumers due to leaving an obligatory opaque white cast on the skin. Further research has shown that sunscreens with added iron oxide pigments and/or pigmentary titanium dioxide can provide the wearer with a substantial amount of HEVL protection.[124][141][142][143] Cosmetic chemists have found that other cosmetic-grade pigments can be functional filler ingredients. Mica was discovered to have significant synergistic effects with UVR filters when formulated in sunscreens, in that it can notably increase the formula's ability to protect the wearer from HEVL.[136]
There is a growing amount of research demonstrating that adding various vitamer antioxidants (eg; retinol, alpha tocopherol, gamma tocopherol, tocopheryl acetate, ascorbic acid, ascorbyl tetraisopalmitate, ascorbyl palmitate, sodium ascorbyl phosphate, ubiquinone) and/or a mixture of certain botanical antioxidants (eg; epigallocatechin-3-gallate, b-carotene, vitis vinifera, silymarin, spirulina extract, chamomile extract and possibly others) to sunscreens efficaciously aids in reducing damage from the free radicals produced by exposure to solar ultraviolet radiation, visible light, near infrared radiation and infrared-a radiation.[120][144][134][124][145][122][125] Since sunscreen's active ingredients work preventatively by creating a shielding film on the skin that absorbs, scatters, and reflects light before it can reach the skin, UV filters have been deemed an ideal “first line of defense” against sun damage when exposure can't be avoided. Antioxidants have been deemed a good “second line of defense” since they work responsively by decreasing the overall burden of free radicals that do reach the skin.[136] The degree of the free radical protection from the entire solar spectral range that a sunscreen can offer has been termed the "radical protection factor" (RPF) by some researchers.
Application
SPF 30 or above must be used to effectively prevent UV rays from damaging skin cells. This is the amount that is recommended to prevent against skin cancer. Sunscreen must also be applied thoroughly and re-applied during the day, especially after being in the water. Special attention should be paid to areas like the ears and nose, which are common spots of skin cancer. Dermatologists may be able to advise about what sunscreen is best to use for specific skin types.[146]
The dose used in FDA sunscreen testing is 2 mg/cm2 of exposed skin.[94] If one assumes an "average" adult build of height 5 ft 4 in (163 cm) and weight 150 lb (68 kg) with a 32-inch (82-cm) waist, that adult wearing a bathing suit covering the groin area should apply approximately 30 g (or 30 ml, approximately 1 oz) evenly to the uncovered body area. This can be more easily thought of as a "golf ball" size amount of product per body, or at least six teaspoonfuls. Larger or smaller individuals should scale these quantities accordingly.[147] Considering only the face, this translates to about 1/4 to 1/3 of a teaspoon for the average adult face.
Some studies have shown that people commonly apply only 1/4 to 1/2 of the amount recommended for achieving the rated sun protection factor (SPF), and in consequence the effective SPF should be downgraded to a 4th root or a square root of the advertised value, respectively.[85] A later study found a significant exponential relation between SPF and the amount of sunscreen applied, and the results are closer to linearity than expected by theory.[148]
Claims that substances in pill form can act as sunscreen are false and disallowed in the United States.[149]
Regulation
- Palau
On 1 January 2020, Palau banned the manufacturing and selling of sun cream products containing any of the following ingredients: benzophenone-3, octyl methoxycinnamate, octocrylene, 4-methyl-benzylidene camphor, triclosan, methylparaben, ethylparaben, butylparaben, benzyl paraben, and phenoxyethanol.[150] The decision was taken to protect the local coral reef and sea life.[151] Those compounds are known or suspected to be harmful to coral or other sea life.[151]
- United States
Sunscreen labeling standards have been evolving in the United States since the FDA first adopted the SPF calculation in 1978.[152] The FDA issued a comprehensive set of rules in June 2011, taking effect in 2012–2013, designed to help consumers identify and select suitable sunscreen products offering protection from sunburn, early skin aging, and skin cancer.[153][154][155] However, unlike other countries, the United States classifies sunscreen as an over-the-counter drug rather than a cosmetic product. As FDA approval of a new drug is typically far slower than for a cosmetic, the result is fewer ingredients available for sunscreen formulations in the US compared with many other countries.[156][157]
In 2019, the FDA proposed tighter regulations on sun protection and general safety, including the requirement that sunscreen products with SPF greater than 15 must be broad spectrum, and imposing a prohibition on products with SPF greater than 60.[158]
- To be classified as "broad spectrum", sunscreen products must provide protection against both UVA and UVB, with specific tests required for both.
- Claims of products being "waterproof" or "sweatproof" are prohibited, while the terms "sunblock" and "instant protection" and "protection for more than 2 hours" are all prohibited without specific FDA approval.
- "Water resistance" claims on the front label must indicate how long the sunscreen remains effective and specify whether this applies to swimming or sweating, based on standard testing.
- Sunscreens must include standardized "Drug Facts" information on the container. However, there is no regulation that deems it necessary to mention whether the contents contain nanoparticles of mineral ingredients. Furthermore, US products do not require the expiration date of products to be displayed on the label.[159]
In 2021, the FDA introduced an additional administrative order regarding the safety classification of cosmetic UV filters, to categorize a given ingredient as either:
- Generally recognized as safe and effective (GRASE)
- Not GRASE due to safety issues
- Not GRASE because additional safety data are needed.[92][14]
To be considered a GRASE active ingredient, the FDA requires it to have undergone both non-clinical animal studies as well as human clinical studies. The animal studies evaluate the potential for inducing carcinogenesis, genetic or reproductive harm, and any toxic effects of the ingredient once absorbed and distributed in the body. The human trials expand upon the animal trials, providing additional information on safety in the pediatric population, protection against UVA and UVB, and the potential for skin reactions after application. Two previously approved UV filters, para-aminobenzoic acid (PABA) and trolamine salicylate, were reclassified as not GRASE due to safety concerns and have consequently been removed from the market.[92]
Europe
In Europe, sunscreens are considered a cosmetic product rather than an over-the-counter drug. These products are regulated by the Cosmetic Regulation (EC) No 1223/2009, which was created in July 2013.[159] The recommendations for formulating sunscreen products are guided by the Scientific Community on Consumer Safety (SCCS).[160] The regulation of cosmetic products in Europe requires the producer to follow six domains when formulating their product:
I. Cosmetic safety report must be conducted by a qualified personnel
II. The product must not contain substances banned for cosmetic products
III. The product must not contain substances restricted for cosmetic products
IV. The product must adhere to the approved list of colourants for cosmetic products.
V. The product must adhere to the approved list of preservatives for cosmetic products.
VI. The product must contain UV filters approved in Europe.[160]
According to the EC, sunscreens at a minimum must exhibit:
- A SPF of 6
- UVA/UVB ratio ≥ 1/3
- The critical wavelength is at least 370 nanometers (indicating that it is "broad-spectrum").
- Instructions for using and precautions.
- Evidence the sunscreen meets UVA and SPF requirements.[160]
- Labels of European sunscreens must disclose the use of nanoparticles in addition to the shelf life of the product.[159]
Canada
Regulation of sunscreen is dependent on the ingredient used; It is then classified and follows the regulations for either natural health products or drug product. Companies must complete a product licensing application prior to introducing their sunscreen on the market.[160]
ASEAN (Brunei, Cambodia, Indonesia, Laos, Malaysia, Myanmar, the Philippines, Singapore, Thailand, Vietnam)
The regulation of sunscreen for ASEAN countries closely follows European regulations. However, products are regulated by the ASEAN scientific community rather than the SCCS. Additionally, there are minor differences in the allowed phrasing printed on sunscreen packages.[160]
Japan
Sunscreen is considered a cosmetic product, and is regulated under the Japan Cosmetic Industry Association (JCIA). Products are regulated mostly for the type of UV filter and SPF. SPF may range from 2 to 50.[160]
China
Sunscreen is regulated as cosmetic product under the State Food and Drug Administration (SFDA). The list of approved filters is the same as it is in Europe. However, sunscreen in China requires safety testing in animal studies prior to approval.[160]
Australia
Sunscreens are divided into therapeutic and cosmetic sunscreens. Therapeutic sunscreens are classified into primary sunscreens (SPF ≥ 4) and secondary sunscreens (SPF < 4). Therapeutic sunscreens are regulated by the Therapeutic Goods Administration (TGA). Cosmetic sunscreens are products that contain a sunscreen ingredient, but do not protect from the sun. These products are regulated by the National Industrial Chemicals Notification and Assessment Scheme (NICNAS).[160]
New Zealand
Sunscreen is classified as a cosmetic product, and closely follows EU regulations. However, New Zealand has a more extensive list of approved UV filters than Europe.[160]
Mercosur
Mercosur is an international group consisting of Argentina, Brazil, Paraguay, and Uruguay. Regulation of sunscreen as a cosmetic product began in 2012, and is similar in structure to the European regulations. Sunscreens must meet specific standards including water resistance, sun protection factor, and a UVA/UVB ratio of 1/3. The list of approved sunscreen ingredients is greater than in Europe or the US.[160]
Environmental effects
Some sunscreen active ingredients have been shown to cause toxicity towards marine life and coral, resulting in bans in different states, countries and ecological areas.[161][162] Coral reefs, comprising organisms in delicate ecological balances, are vulnerable to even minor environmental disturbances. Factors like temperature changes, invasive species, pollution, and detrimental fishing practices have previously been highlighted as threats to coral health.[163][164]
In 2018, Hawaii passed a legislation that prohibits the sale of sunscreens containing oxybenzone and octinoxate. These chemicals, based on different researches, have been found to negatively impact coral reefs. In sufficient concentrations, these compounds can damage coral DNA, induce deformities in juvenile corals,[162] heighten the risk of viral infections, and make corals more vulnerable to bleaching. Such threats are even more concerning given that coral ecosystems are already compromised by climate change, pollution, and other environmental stressors. While there is ongoing debate regarding the real-world concentrations of these chemicals versus laboratory settings,[165][166][167][168] an assesment in Kahaluu Bay in Hawaii showed oxybenzone concentrations to be 262 times higher than what the U.S. Environmental Protection Agency designates as high-risk. Another study in Hanauma Bay found levels of the chemical ranging from 30 ng/L to 27,880 ng/L, noting that concentrations beyond 63 ng/L could induce toxicity in corals.[169]
Echoing Hawaii's initiative, other regions including Key West, Florida, the U.S. Virgin Islands, Bonaire, and Palau[170] have also instituted bans on these harmful sunscreen chemicals.
The environmental implications of sunscreen usage on marine ecosystems are multi-faceted and vary in severity. In a 2015 study, titanium dioxide, when introduced to water and subjected to ultraviolet light, was shown to amplify the production of hydrogen peroxide, a compound known to damage phytoplankton.[171] In 2002, research indicated that sunscreens might escalate virus abundance in seawater, compromising the marine environment in a manner akin to other pollutants.[172] Further probing the matter, a 2008 investigation examining a variety of sunscreen brands, protective factors, and concentrations revealed unanimous bleaching effects on hard corals. Alarmingly, the degree of bleaching magnified with increasing sunscreen quantities. When assessing individual compounds prevalent in sunscreens, substances such as butylparaben, ethylhexylmethoxycinnamate, benzophenone-3, and 4-methylbenzylidene camphor induced complete coral bleaching at even minimal concentrations.[173]
A 2020 study from the journal Current Dermatology Report summarised the situation as the US FDA currently approves only zinc oxide (ZnO) and titanium dioxide (TiO2) as safe ultraviolet filters and for those concerned with coral bleaching they should use non-nano ZnO or TiO2, as they have the most consistent safety data.[174]
Research and Development
New products are in development such as sunscreens based on bioadhesive nanoparticles. These function by encapsulating commercially used UV filters, while being not only adherent to the skin but also non-penetrant. This strategy inhibits primary UV-induced damage as well as secondary free radicals.[175] UV filters based on sinapate esters are also under study.[176] Sunscreens with natural and sustainable connotations are increasingly being developed, as a result of increased environmental concern.[177]
Note
References
- ↑ "Preventing melanoma". Cancer Research UK. http://www.cancerhelp.org.uk/help/default.asp?page=3007.
- ↑ EU SPF Regulations: Labelling and claims
- ↑ 3.0 3.1 3.2 3.3 "Questions and Answers: FDA announces new requirements for over-the-counter (OTC) sunscreen products marketed in the U.S.". 2011-06-23. https://www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/UnderstandingOver-the-CounterMedicines/ucm258468.htm.
- ↑ "WHIPPED FORMULATIONS WITH DESIRED SENSORY IMPACT". July 8, 2022. https://www.ic.gc.ca/opic-cipo/cpd/eng/patent/3023702/summary.html?query=whipped%2C+carpenter&type=basic_search.
- ↑ Cole, Curtis; Shyr, Thomas; Ou‐Yang, Hao (2015-10-02). "Metal oxide sunscreens protect skin by absorption, not by reflection or scattering" (in en). Photodermatology, Photoimmunology & Photomedicine 32 (1): 5–10. doi:10.1111/phpp.12214. ISSN 0905-4383. PMID 26431814. https://onlinelibrary.wiley.com/doi/10.1111/phpp.12214.
- ↑ Schneider, Samantha L.; Lim, Henry W. (2018-11-16). "A review of inorganic UV filters zinc oxide and titanium dioxide" (in en). Photodermatology, Photoimmunology & Photomedicine 35 (6): 442–446. doi:10.1111/phpp.12439. ISSN 0905-4383. PMID 30444533. https://onlinelibrary.wiley.com/doi/10.1111/phpp.12439.
- ↑ 7.0 7.1 "Failure to Protect: Do Sunscreens Prevent Skin Cancer in Humans?". April 2021. https://www.researchgate.net/publication/352061297.
- ↑ ""Physical" vs. "chemical" sunscreens and other sunscreen myths". September 2016. https://kindofstephen.com/physical-vs-chemical-sunscreens-myths/.
- ↑ "The toxicological impact of the sunscreen active ingredient octinoxate on the photosynthesis activity of Chlorella sp". Marine Environmental Research 171: 105469. October 2021. doi:10.1016/j.marenvres.2021.105469. PMID 34500299. Bibcode: 2021MarER.17105469T.
- ↑ 10.0 10.1 "The banned sunscreen ingredients and their impact on human health: a systematic review". International Journal of Dermatology 59 (9): 1033–1042. September 2020. doi:10.1111/ijd.14824. PMID 32108942.
- ↑ "Environmental impacts due to the use of sunscreen products: a mini-review". Ecotoxicology 31 (9): 1331–1345. November 2022. doi:10.1007/s10646-022-02592-w. PMID 36173495.
- ↑ "Skin Cancer - Skin Cancer Facts - Common Skin Cancer Types". http://www.cancer.org/docroot/PED/content/ped_7_1_What_You_Need_To_Know_About_Skin_Cancer.asp.
- ↑ Sunscreens and Photoprotection at eMedicine
- ↑ 14.0 14.1 Research, Center for Drug Evaluation and (2021-11-16). "Questions and Answers: FDA posts deemed final order and proposed order for over-the-counter sunscreen" (in en). FDA. https://www.fda.gov/drugs/understanding-over-counter-medicines/questions-and-answers-fda-posts-deemed-final-order-and-proposed-order-over-counter-sunscreen.
- ↑ "Sunscreen Evolution". Sunscreens : regulations and commercial development (3rd ed.). Boca Raton, Fl.: Taylor & Francis. 2005. ISBN 978-0824757946.
- ↑ 2000 Years of Zinc and Brass. British Museum. 1998. p. 27. ISBN 978-0-86159-124-4.
- ↑ "In Pictures: Nomads of the sea". Al Jazeera. 20 March 2014. http://www.aljazeera.com/indepth/inpictures/2014/03/pictures-nomads-sea-2014312760932684.html.
- ↑ "Ethnobotany of Madagascar", The New Natural History of Madagascar (Princeton University Press): pp. 237, 2022-11-15, doi:10.2307/j.ctv2ks6tbb.34, ISBN 978-0-691-22940-9
- ↑ Miora (2020-09-06). "Le masque au Santal" (in fr-FR). https://www.madavoyages.com/le-masque-au-santal.
- ↑ "History of sunscreen: An updated view". Journal of Cosmetic Dermatology 20 (4): 1044–1049. April 2021. doi:10.1111/jocd.14004. PMID 33583116.
- ↑ 21.0 21.1 21.2 21.3 "Photoprotection" (in en). Photoaging. CRC Press. 2004-01-30. pp. 73–74. ISBN 978-0-8247-5209-5. https://books.google.com/books?id=RvCINU2hOHAC&q=milton+blake+sunscreen&pg=PA74.
- ↑ (in en) Photoaging. CRC Press. 2004-01-30. ISBN 978-0-8247-5209-5. https://books.google.com/books?id=RvCINU2hOHAC.
- ↑ "7 Wonders of South Australia winners: Innovations - ABC (none) - Australian Broadcasting Corporation". https://www.abc.net.au/local/stories/2014/07/23/4051703.htm.
- ↑ "History Of Hamilton" (in en-US). https://www.hamiltonsunandskin.com.au/history-of-hamilton.
- ↑ 25.0 25.1 25.2 25.3 "Quantum Leaps: New, Improved Sunscreens Have Arrived". The Skin Cancer Foundation. http://www.skincancer.org/prevention/sun-protection/sunscreen/Quantum-Leaps-New-Improved-Sunscreens-Have-Arrived.
- ↑ "Challenges in Making an Effective Sunscreen". The Skin Cancer Foundation. http://www.skincancer.org/publications/the-melanoma-letter/summer-2012-vol-30-no-2/effective-sunscreen.
- ↑ "A Practical Sunscreen -- "RED VET PET"". Archives of Dermatology 89 (1): 147–150. January 1964. doi:10.1001/archderm.1964.01590250153027. PMID 14070829.
- ↑ Sunscreens: Regulations and Commercial Development, Third Edition. Taylor & Francis Group. 2005.
- ↑ "Sunscreen: A History". The New York Times. June 23, 2010. https://www.nytimes.com/2010/06/24/fashion/24skinside.html.
- ↑ "Gletscher Crème". 2010-04-22. Piz Buin. http://www.pizbuin.com/v1/en/brand_story.html.
- ↑ Photodermatology. CRC Press. 2007. p. 6. ISBN 9781420019964. https://books.google.com/books?id=g-YCKEPYMpYC&pg=PA6. Retrieved July 24, 2014.
- ↑ "Environmental impacts of the ultraviolet filter oxybenzone". The Science of the Total Environment 863: 160966. March 2023. doi:10.1016/j.scitotenv.2022.160966. PMID 36535482. Bibcode: 2023ScTEn.863p0966S.
- ↑ Research, Center for Drug Evaluation and (2022-12-16). "An update on sunscreen requirements: The deemed final order and the proposed order" (in en). FDA. https://www.fda.gov/drugs/news-events-human-drugs/update-sunscreen-requirements-deemed-final-order-and-proposed-order.
- ↑ "Ultraviolet radiation and melanoma". Seminars in Cutaneous Medicine and Surgery 30 (4): 222–228. December 2011. doi:10.1016/j.sder.2011.08.003. PMID 22123420.
- ↑ World Cancer Report 2014.. World Health Organization. 2014. pp. Chapter 5.14. ISBN 978-9283204299.
- ↑ "Epidemiology, risk factors, prevention, and early detection of melanoma". The Surgical Clinics of North America 94 (5): 945–62, vii. October 2014. doi:10.1016/j.suc.2014.07.013. PMID 25245960.
- ↑ "Current sunscreen controversies: a critical review". Photodermatology, Photoimmunology & Photomedicine 27 (2): 58–67. April 2011. doi:10.1111/j.1600-0781.2011.00557.x. PMID 21392107.
- ↑ "UV-induced skin cancer at workplace and evidence-based prevention". International Archives of Occupational and Environmental Health 83 (8): 843–854. December 2010. doi:10.1007/s00420-010-0532-4. PMID 20414668.
- ↑ 39.0 39.1 "Sunscreen and prevention of skin aging: a randomized trial". Annals of Internal Medicine 158 (11): 781–790. June 2013. doi:10.7326/0003-4819-158-11-201306040-00002. PMID 23732711. http://annals.org/article.aspx?articleid=1691732. Retrieved June 6, 2013.
- ↑ "Daily Use of a Facial Broad Spectrum Sunscreen Over One-Year Significantly Improves Clinical Evaluation of Photoaging". Dermatologic Surgery 42 (12): 1354–1361. December 2016. doi:10.1097/DSS.0000000000000879. PMID 27749441.
- ↑ "Ultraviolet Radiation". Ohioline Fact Sheet Series. Ohio State University Extension. 2008. http://ohioline.osu.edu/cd-fact/pdf/0199.pdf.
- ↑ Sabzevari, Nina; Qiblawi, Sultan; Norton, Scott A.; Fivenson, David (2020-05-26). "Sunscreens: UV filters to protect us: Part 1: Changing regulations and choices for optimal sun protection" (in en). International Journal of Women's Dermatology 7 (1): 28–44. doi:10.1016/j.ijwd.2020.05.017. PMID 33537394.
- ↑ Center for Drug Evaluation and Research (2021-11-16). "Questions and Answers: FDA posts deemed final order and proposed order for over-the-counter sunscreen" (in en). FDA. https://www.fda.gov/drugs/understanding-over-counter-medicines/questions-and-answers-fda-posts-deemed-final-order-and-proposed-order-over-counter-sunscreen.
- ↑ "Skincare Products as Sources of Mutagenic Exposure to Infants: An Imperative Study Using a Battery of Microbial Bioassays". Archives of Environmental Contamination and Toxicology 80 (2): 499–506. February 2021. doi:10.1007/s00244-021-00814-6. PMID 33523258.
- ↑ "Sunscreen: What science says about ingredient safety" (in en). https://www.bbc.com/future/article/20190722-sunscreen-safe-or-toxic.
- ↑ "State of Hawaii, Department of Health, Testimony Commenting on hb1519, Relating to Sunscreen, House Committee on Energy & Environmental Protection". 8 February 2022. https://www.capitol.hawaii.gov/sessions/Session2022/Testimony/HB1519_TESTIMONY_EEP_02-08-22_.PDF.
- ↑ "Food and Drug Administration's proposed sunscreen final administrative order: How does it affect sunscreens in the United States?". Journal of the American Academy of Dermatology 86 (2): e83–e84. February 2022. doi:10.1016/j.jaad.2021.09.052. PMID 34606770.
- ↑ "Sunscreens - ensuring products are effective and safe for the 2021-22 summer". June 21, 2022. https://www.tga.gov.au/news/news/sunscreens-ensuring-products-are-effective-and-safe-2021-22-summer.
- ↑ "Valisure" (in en). https://www.valisure.com/valisure-newsroom/valisure-detects-benzene-in-sunscreen.
- ↑ Center for Drug Evaluation and Research (2023-01-30). "Edgewell Personal Care Issues Voluntary Nationwide Recall of Banana Boat Hair & Scalp Sunscreen Due to the Presence of Benzene" (in en). https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/edgewell-personal-care-issues-voluntary-nationwide-recall-banana-boat-hair-scalp-sunscreen-due.
- ↑ "Occurrence of and dermal exposure to benzene, toluene and styrene in sunscreen products marketed in the United States". The Science of the Total Environment 888: 164196. August 2023. doi:10.1016/j.scitotenv.2023.164196. PMID 37201845. Bibcode: 2023ScTEn.888p4196P.
- ↑ "What is benzene, and why does it keep causing beauty product recalls?". The Washington Post. 1 February 2023. https://www.washingtonpost.com/wellness/2023/02/01/benzene-aerosol-recalls-sunscreen-shampoo/.
- ↑ "Effect of Sunscreen Application on Plasma Concentration of Sunscreen Active Ingredients: A Randomized Clinical Trial". JAMA 323 (3): 256–267. January 2020. doi:10.1001/jama.2019.20747. PMID 31961417.
- ↑ "Fundamental questions to sun protection: A continuous education symposium on vitamin D, immune system and sun protection at the University of Zürich". Dermato-Endocrinology 2 (1): 19–25. January 2010. doi:10.4161/derm.2.1.12016. PMID 21547144.
- ↑ "Sunscreen allergy". https://www.dermnetnz.org/topics/sunscreen-allergy/.
- ↑ "Vitamin D Deficiency, Its Role in Health and Disease, and Current Supplementation Recommendations". The Journal of the American Osteopathic Association 117 (5): 301–305. May 2017. doi:10.7556/jaoa.2017.055. PMID 28459478.
- ↑ "Sunscreen may cause vitamin D deficiency, says study". 2017-05-03. https://www.medicalnewstoday.com/articles/317278.
- ↑ "Does chronic sunscreen use reduce vitamin D production to insufficient levels?". The British Journal of Dermatology 161 (4): 732–736. October 2009. doi:10.1111/j.1365-2133.2009.09332.x. PMID 19663879.
- ↑ "Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease". The American Journal of Clinical Nutrition 80 (6 Suppl): 1678S–1688S. December 2004. doi:10.1093/ajcn/80.6.1678S. PMID 15585788.
- ↑ "Darkness at noon: sunscreens and vitamin D3". Photochemistry and Photobiology 83 (2): 459–463. 2007. doi:10.1562/2006-06-29-RC-956. PMID 17115796.
- ↑ "Vitamin D" (in en). 2017-10-23. https://www.nhs.uk/conditions/vitamins-and-minerals/vitamin-d/.
- ↑ "Vitamin D: the underappreciated D-lightful hormone that is important for skeletal and cellular health". Current Opinion in Endocrinology, Diabetes and Obesity 9 (1): 87–98. February 2002. doi:10.1097/00060793-200202000-00011.
- ↑ "Sunlight and vitamin D: both good for cardiovascular health". Journal of General Internal Medicine 17 (9): 733–735. September 2002. doi:10.1046/j.1525-1497.2002.20731.x. PMID 12220371.
- ↑ "Vitamin D deficiency". The New England Journal of Medicine 357 (3): 266–281. July 2007. doi:10.1056/NEJMra070553. PMID 17634462.
- ↑ "Are Vitamin D Levels Jeopardized by Sunscreen?". 2019-05-10. https://www.genengnews.com/news/are-vitamin-d-levels-jeopardized-by-sunscreen/.
- ↑ "Optimal sunscreen use, during a sun holiday with a very high ultraviolet index, allows vitamin D synthesis without sunburn". The British Journal of Dermatology 181 (5): 1052–1062. November 2019. doi:10.1111/bjd.17888. PMID 31069787.
- ↑ "Sunscreen: The comprehensive guide to sunscreen in Australia" (in en). Surf Nation. https://www.surfnation.com.au/blogs/news/sunscreen-the-comprehensive-guide-to-sunscreen-in-australia.
- ↑ "Sunscreen FAQs". American Academy of Dermatology. http://www.aad.org/media-resources/stats-and-facts/prevention-and-care/sunscreens.
- ↑ "Sunscreens with high SPF values are not equivalent in protection from UVA induced polymorphous light eruption". European Journal of Dermatology 12 (4): IV-VI. 2002. PMID 12118426.
- ↑ "Sunscreens inadequately protect against ultraviolet-A-induced free radicals in skin: implications for skin aging and melanoma?". The Journal of Investigative Dermatology 121 (4): 862–868. October 2003. doi:10.1046/j.1523-1747.2003.12498.x. PMID 14632206.
- ↑ "Broad-spectrum sunscreens provide better protection from solar ultraviolet-simulated radiation and natural sunlight-induced immunosuppression in human beings". Journal of the American Academy of Dermatology 58 (5 Suppl 2): S149–S154. May 2008. doi:10.1016/j.jaad.2007.04.035. PMID 18410801.
- ↑ "Induction of the photoaging-associated mitochondrial common deletion in vivo in normal human skin". The Journal of Investigative Dermatology 122 (5): 1277–1283. May 2004. doi:10.1111/j.0022-202X.2004.22502.x. PMID 15140232.
- ↑ "Sunscreen makers sued for misleading claims". Associated Press. April 24, 2006. http://www.nbcnews.com/id/12081374/.
- ↑ "Microfine zinc oxide is a superior sunscreen ingredient to microfine titanium dioxide". Dermatologic Surgery 26 (4): 309–314. April 2000. doi:10.1046/j.1524-4725.2000.99237.x. PMID 10759815.
- ↑ "Commission Recommendation of 22 September 2006 on the efficacy of sunscreen products and the claims made relating thereto". Official Journal of the European Union. 2006-09-22. http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:32006H0647:EN:NOT. Retrieved 2009-09-25.
- ↑ "UV Resource Guide - Sunscreens". Arpansa. 2008-12-20. http://www.arpansa.gov.au/uvrg/rginfo_p13.cfm.
- ↑ "SPF50+ Sunscreen". 2013-02-01. http://www.cancer.org.au/preventing-cancer/sun-protection/preventing-skin-cancer/spf50sunscreen.html.
- ↑ 78.0 78.1 "Questions and Answers on the 2007 Sunscreen Proposed Rule". https://www.fda.gov/cder/drug/infopage/sunscreen/qa.htm.
- ↑ Department of Health and Human Services: Food and Drug Administration (June 17, 2011). "Revised Effectiveness Determination; Sunscreen Drug Products for Over-the-Counter Human Use". Federal Register 76 (117): 35672–35678. https://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/DevelopmentResources/Over-the-CounterOTCDrugs/StatusofOTCRulemakings/UCM313542.pdf. Retrieved November 21, 2013.
- ↑ Center for Drug Evaluation and Research (April 23, 2019). "Status of OTC Rulemakings - Rulemaking History for OTC Sunscreen Drug Products". FDA. https://www.fda.gov/Drugs/DevelopmentApprovalProcess/DevelopmentResources/Over-the-CounterOTCDrugs/StatusofOTCRulemakings/ucm072134.htm. Retrieved April 11, 2017.
- ↑ 81.0 81.1 "Sunscreen Takes Some Heat: New Dangers, New Rules". 2011-06-16. http://onpoint.wbur.org/2011/06/16/sunscreen.
- ↑ "The Burning Facts". 2006. https://www.epa.gov/sites/production/files/documents/sunscreen.pdf.
- ↑ "How to measure UVA protection afforded by sunscreen products.". Expert Review of Dermatology 3 (3): 307–13. June 2008. doi:10.1586/17469872.3.3.307. http://www.medscape.com/viewarticle/576849. Retrieved August 17, 2016.
- ↑ "What to Know About Sunscreen Before Buying It". Consumer Reports. May 2014. http://www.consumerreports.org/cro/magazine/2014/07/what-to-know-about-sunscreen-before-buying-it/. Retrieved December 20, 2014.
- ↑ 85.0 85.1 "The relation between sun protection factor and amount of suncreen applied in vivo". The British Journal of Dermatology 156 (4): 716–719. April 2007. doi:10.1111/j.1365-2133.2006.07684.x. PMID 17493070.
- ↑ "Method for the in vitro determination of uva protection provided by sunscreen products, 2007a". June 9, 2008. http://www.colipa.com/site/index.cfm?SID=15588&OBJ=28546&back=1.
- ↑ "www.cosmeticseurope.eu". https://www.cosmeticseurope.eu/using-cosmetics-colipa-the-european-cosmetic-cosmetics-association/sun-products/the-european-commission-recommendation-on-the-efficacy-of-sunscreen-products.html.
- ↑ Cosmetics Europe (February 2009). "N° 23 IMPORTANT USAGE AND LABELLING INSTRUCTIONS FOR SUN PROTECTION PRODUCTS". https://www.cosmeticseurope.eu/files/9814/6408/4022/CR-23-Sunscreens_Labelling.pdf.
- ↑ "Commission Recommendation of 22 September 2006 on the efficacy of sunscreen products and the claims made relating thereto (notified under document number C(2006) 4089) (Text with EEA relevance)" (in en). 2006-09-26. http://data.europa.eu/eli/reco/2006/647/oj/eng.
- ↑ "Is sunscreen from last year still good? When does sunscreen expire?". Mayo Clinic. https://www.mayoclinic.org/healthy-lifestyle/adult-health/expert-answers/sunscreen-expire/faq-20057957.
- ↑ "Metal oxide sunscreens protect skin by absorption, not by reflection or scattering". Photodermatology, Photoimmunology & Photomedicine 32 (1): 5–10. January 2016. doi:10.1111/phpp.12214. PMID 26431814.
- ↑ 92.0 92.1 92.2 92.3 "Ultraviolet filters in the United States and European Union: A review of safety and implications for the future of US sunscreens". Journal of the American Academy of Dermatology 88 (3): 632–646. March 2023. doi:10.1016/j.jaad.2022.11.039. PMID 36442641.
- ↑ "Nanotechnology Information Center: Properties, Applications, Research, and Safety Guidelines". American Elements. http://www.americanelements.com/nanotech.htm.
- ↑ 94.0 94.1 "Re: Tentative Final Monograph for OTC Sunscreen". Cosmetic, Toiletry, and Fragrance Association. 1998-09-11. https://www.fda.gov/ohrms/dockets/dailys/00/Sep00/090600/c000573_10_Attachment_F.pdf.
- ↑ "Photostabilization of butyl methoxydibenzoylmethane (Avobenzone) and ethylhexyl methoxycinnamate by bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S), a new UV broadband filter". Photochemistry and Photobiology 74 (3): 401–406. September 2001. doi:10.1562/0031-8655(2001)074<0401:POBMAA>2.0.CO;2. PMID 11594052.
- ↑ "Parsol 340 – Octocrylene". DSM. http://www.dsm.com/en_US/html/dnpus/pe_parsol_340.htm.
- ↑ "Does sunscreen become ineffective with age?". The Skin Cancer Foundation. http://www.skincancer.org/skin-cancer-information/ask-the-experts/does-sunscreen-become-ineffective-with-age.
- ↑ "The inhibiting effect of PABA on photocarcinogenesis". Archives of Dermatological Research 282 (1): 38–41. 1990. doi:10.1007/BF00505643. PMID 2317082.
- ↑ "The effect of short-term application of PABA on photocarcinogenesis". Acta Dermato-Venereologica 70 (1): 72–75. 1990. doi:10.2340/00015555707275. PMID 1967881.
- ↑ "The sensitization of near-ultraviolet radiation killing of mammalian cells by the sunscreen agent para-aminobenzoic acid". The Journal of Investigative Dermatology 79 (6): 354–357. December 1982. doi:10.1111/1523-1747.ep12529409. PMID 6982950.
- ↑ 101.0 101.1 101.2 101.3 Center for Drug Evaluation and Research (2022-12-16). "Questions and Answers: FDA posts deemed final order and proposed order for over-the-counter sunscreen" (in en). FDA. https://www.fda.gov/drugs/understanding-over-counter-medicines/questions-and-answers-fda-posts-deemed-final-order-and-proposed-order-over-counter-sunscreen.
- ↑ "Light-induced cytotoxicity and genotoxicity of a sunscreen agent, 2-phenylbenzimidazole in Salmonella typhimurium TA 102 and HaCaT keratinocytes". International Journal of Environmental Research and Public Health 4 (2): 126–131. June 2007. doi:10.3390/ijerph2007040006. PMID 17617675.
- ↑ "Scientific Committee on Consumer Safety (SCCS) - OPINION on Benzophenone-3". https://health.ec.europa.eu/system/files/2022-08/sccs_o_247.pdf.
- ↑ "Sunscreen Use in Hawaii - AN ASSESSMENT OF BEACH-GOER USE OF SUNSCREEN PRIOR TO IMPLEMENTATION OF THE 2021 CHEMICAL BAN". September 2019. https://www.kohalacenter.org/docs/reports/Sunscreen_Report_Sept2019.pdf.
- ↑ "Sunscreen enhancement of UV-induced reactive oxygen species in the skin". Free Radical Biology & Medicine 41 (8): 1205–1212. October 2006. doi:10.1016/j.freeradbiomed.2006.06.011. PMID 17015167. http://www.escholarship.org/uc/item/9f14s2dd. Retrieved July 25, 2019.
- ↑ "Hawaii is about to ban your favorite sunscreen to protect its coral reefs". The Washington Post. https://www.washingtonpost.com/news/energy-environment/wp/2018/07/02/hawaii-is-about-to-ban-your-favorite-sunscreen-to-protect-its-coral-reefs/?noredirect=on.
- ↑ 107.0 107.1 "Photoprotection". Lancet 370 (9586): 528–537. August 2007. doi:10.1016/S0140-6736(07)60638-2. PMID 17693182.
- ↑ "Percutaneous absorption of Mexoryl SX in human volunteers: comparison with in vitro data". Skin Pharmacology and Applied Skin Physiology 16 (6): 343–355. Nov–Dec 2003. doi:10.1159/000072929. PMID 14528058.
- ↑ "Mexoryl SX protects against solar-simulated UVR-induced photocarcinogenesis in mice". Photochemistry and Photobiology 64 (4): 688–693. October 1996. doi:10.1111/j.1751-1097.1996.tb03125.x. PMID 8863475.
- ↑ "Amending Annex VI to Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products". 2016-04-21. http://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32016R0621&from=EN.
- ↑ "Regulation No. 1223/2009 on cosmetic products". Official Journal of the European Union. December 22, 2009. http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32009R1223&qid=1406148728060&from=EN. Retrieved May 26, 2015.
- ↑ Australian Government: Therapeutic Goods Administration (November 2012). "Australian Regulatory Guidelines for Sunscreens". http://www.tga.gov.au/publication/australian-regulatory-guidelines-sunscreens-args.
- ↑ Research, Center for Drug Evaluation and (2022-12-16). "Questions and Answers: FDA posts deemed final order and proposed order for over-the-counter sunscreen" (in en). FDA. https://www.fda.gov/drugs/understanding-over-counter-medicines/questions-and-answers-fda-posts-deemed-final-order-and-proposed-order-over-counter-sunscreen.
- ↑ "Uvinul Grades". http://www.basf-korea.co.kr/02_products/04_finechemicals/document/cosmetic/tech/uvabsorber/down.asp?file=uvinulgrades.pdf.
- ↑ Kapes B (July 2005). "Docs rally for better sun protection — Advances still unavailable in United States". Dermatology Times 26 (7): 100. http://www.modernmedicine.com/modernmedicine/article/articleDetail.jsp?id=169626. Retrieved July 23, 2014.
- ↑ "Sunscreen Innovation Act". United States Congress. November 26, 2014. https://www.congress.gov/bill/113th-congress/senate-bill/2141/all-actions.
- ↑ "We're One Step Closer to Better Sunscreen". Time. July 16, 2014. http://time.com/2992200/were-one-step-closer-to-better-sunscreen/. Retrieved August 1, 2014.
- ↑ Australian Government Department of Health Therapeutic Goods Administration (2019-08-30). "Australian Regulatory Guidelines for Sunscreens (ARGS)" (in en). https://www.tga.gov.au/resource/australian-regulatory-guidelines-sunscreens-args.
- ↑ "Sunscreen Formulas With Multilayer Lamella Structure". Cosmetics & Toiletries. Croda Inc , North American Technical Center. June 24, 2009. https://www.cosmeticsandtoiletries.com/formulating/function/viscositymod/2696511.html.
- ↑ 120.0 120.1 "Antioxidants add protection to a broad-spectrum sunscreen". Clinical and Experimental Dermatology 36 (2): 178–187. March 2011. doi:10.1111/j.1365-2230.2010.03916.x. PMID 20804506.
- ↑ 121.0 121.1 "Sunscreens Containing Cyclodextrin Inclusion Complexes for Enhanced Efficiency: A Strategy for Skin Cancer Prevention". Molecules 26 (6): 1698. March 2021. doi:10.3390/molecules26061698. PMID 33803643.
- ↑ 122.0 122.1 "Effectiveness of antioxidants (vitamin C and E) with and without sunscreens as topical photoprotectants". Acta Dermato-Venereologica 76 (4): 264–268. July 1996. doi:10.2340/0001555576264268. PMID 8869680.
- ↑ "Silymarin: An interesting modality in dermatological therapeutics". Indian Journal of Dermatology, Venereology and Leprology 84 (2): 238–243. 2018. doi:10.4103/ijdvl.IJDVL_746_16. PMID 29350205. https://ijdvl.com/?view-pdf=1&embedded=true&article=6d28812c6c2103e3ad1d17bc15c089c1haeLWQ%3D%3D.
- ↑ 124.0 124.1 124.2 124.3 124.4 "Current challenges in photoprotection". Journal of the American Academy of Dermatology 76 (3S1): S91–S99. March 2017. doi:10.1016/j.jaad.2016.09.040. PMID 28038886.
- ↑ 125.0 125.1 "Update on photoprotection". Indian Journal of Dermatology 57 (5): 335–342. September 2012. doi:10.4103/0019-5154.100472. PMID 23112351.
- ↑ "The osmolyte taurine protects against ultraviolet B radiation-induced immunosuppression". Journal of Immunology 179 (6): 3604–3612. September 2007. doi:10.4049/jimmunol.179.6.3604. PMID 17785795.
- ↑ "Ectoin: an effective natural substance to prevent UVA-induced premature photoaging". Skin Pharmacology and Physiology 17 (5): 232–237. September 2004. doi:10.1159/000080216. PMID 15452409.
- ↑ "Influence of hydroxypropyl-beta-cyclodextrin on transdermal penetration and photostability of avobenzone". European Journal of Pharmaceutics and Biopharmaceutics 69 (2): 605–612. June 2008. doi:10.1016/j.ejpb.2007.12.015. PMID 18226883.
- ↑ "The effect of Beta-cyclodextrin on percutaneous absorption of commonly used Eusolex® sunscreens". Drug Research 63 (11): 591–596. November 2013. doi:10.1055/s-0033-1349089. PMID 23842944.
- ↑ "Polycrylene for Photostabilization and Water Resistance". July 3, 2012. https://www.cosmeticsandtoiletries.com/formulating/category/suncare/161093645.html.
- ↑ "Hallstar develops photostabilizer for sun care products" (in en-GB). https://www.cosmeticsdesign.com/Article/2012/07/02/Hallstar-develops-photostabilizer-for-sun-care-products.
- ↑ "[New aspects in the development of sunscreening agents]" (in de). Der Hautarzt; Zeitschrift Fur Dermatologie, Venerologie, und Verwandte Gebiete 68 (5): 349–353. May 2017. doi:10.1007/s00105-017-3965-9. PMID 28280909.
- ↑ "[Sun-damaged skin (photoaging): what is new?]" (in de). Der Hautarzt; Zeitschrift Fur Dermatologie, Venerologie, und Verwandte Gebiete 72 (1): 2–5. January 2021. doi:10.1007/s00105-020-04747-4. PMID 33346860.
- ↑ 134.0 134.1 "Radical-Scavenging Activity of a Sunscreen Enriched by Antioxidants Providing Protection in the Whole Solar Spectral Range". Skin Pharmacology and Physiology 30 (2): 81–89. 2017. doi:10.1159/000458158. PMID 28319939.
- ↑ "What You Didn't Know About the Sun: Infrared Radiation and Its Role in Photoaging". Plastic Surgical Nursing 40 (3): 166–168. July 2020. doi:10.1097/PSN.0000000000000334. PMID 32852443.
- ↑ 136.0 136.1 136.2 "Synergy of mica and inorganic UV filters maximizes Blue Light Protection as first defense line". Germany: Merck. 2 May 2018. https://ifscc.org/wp-content/uploads/2019/05/Emanuele-Piras-_Synergy-of-mica-and-inorganic-UV-filters-for-Blue-Light-Protection.pdf.
- ↑ "[UV, visible and infrared light. Which wavelengths produce oxidative stress in human skin?]" (in de). Der Hautarzt; Zeitschrift Fur Dermatologie, Venerologie, und Verwandte Gebiete 60 (4): 310–317. April 2009. doi:10.1007/s00105-008-1628-6. PMID 19319493.
- ↑ 138.0 138.1 138.2 "Advanced Sun protection with Titanium Dioxides and Functional Fillers". Merck. June 2017. https://www.crq4.org.br/sms/files/file/eventos/seminario_prot_inoutdoor_2018/palestra_advanced_alexander_kielbassa.pdf.
- ↑ "Labeling and Effectiveness Testing: Sunscreen Drug Products for Over-The-Counter Human Use — Small Entity Compliance Guide" (in en). Center for Drug Evaluation and Research. 22 March 2018. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/labeling-and-effectiveness-testing-sunscreen-drug-products-over-counter-human-use-small-entity.
- ↑ "A novel in vivo test method for evaluating the infrared radiation protection provided by sunscreen products". Skin Research and Technology 25 (6): 890–895. November 2019. doi:10.1111/srt.12754. PMID 31338921.
- ↑ "Impact of Iron-Oxide Containing Formulations Against Visible Light-Induced Skin Pigmentation in Skin of Color Individuals". Journal of Drugs in Dermatology 19 (7): 712–717. July 2020. doi:10.36849/JDD.2020.5032. PMID 32726103.
- ↑ "Iron oxides in novel skin care formulations attenuate blue light for enhanced protection against skin damage". Journal of Cosmetic Dermatology 20 (2): 532–537. February 2021. doi:10.1111/jocd.13803. PMID 33210401.
- ↑ "Photoprotection beyond ultraviolet radiation: A review of tinted sunscreens". Journal of the American Academy of Dermatology 84 (5): 1393–1397. May 2021. doi:10.1016/j.jaad.2020.04.079. PMID 32335182.
- ↑ "Effective photoprotection of human skin against infrared A radiation by topically applied antioxidants: results from a vehicle controlled, double-blind, randomized study". Photochemistry and Photobiology 91 (1): 248–250. January 2015. doi:10.1111/php.12375. PMID 25349107.
- ↑ "Specific effects of single antioxidants in the lipid peroxidation caused by nano-titania used in sunscreen lotions". Journal of Photochemistry and Photobiology. B, Biology 96 (2): 130–135. August 2009. doi:10.1016/j.jphotobiol.2009.05.001. PMID 19527937.
- ↑ "Skin Cancer Foundation". https://www.skincancer.org/skin-cancer-prevention/.
- ↑ "How and why we use sunscreen". Cosmetic, Toiletry & Perfumery Association. http://www.thefactsabout.co.uk/how-and-why-we-use-sunscreen/content/239.
- ↑ "The influence of the amount of sunscreen applied and its sun protection factor (SPF): evaluation of two sunscreens including the same ingredients at different concentrations". Photodermatology, Photoimmunology & Photomedicine 25 (4): 175–180. August 2009. doi:10.1111/j.1600-0781.2009.00408.x. PMID 19614894.
- ↑ "Press Announcements - Statement from FDA Commissioner Scott Gottlieb, M.D., on new FDA actions to keep consumers safe from the harmful effects of sun exposure, and ensure the long-term safety and benefits of sunscreens" (in en). https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm608499.htm.
- ↑ "Palau Bans 'Reef Toxic' Sunscreens". 23 January 2020. https://www.allcleartravel.co.uk/blog/palau-bans-reef-toxic-sunscreens/.
- ↑ 151.0 151.1 "Palau is first country to ban 'reef toxic' sun cream". BBC News. 1 January 2020. https://www.bbc.com/news/world-asia-50963080.
- ↑ Department of Health and Human Services: Food and Drug Administration (August 25, 1978). "Sunscreen Drug Products for Over-the-Counter Human Use". Federal Register 43 (166): 38206–38269. https://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/DevelopmentResources/Over-the-CounterOTCDrugs/StatusofOTCRulemakings/ucm090127.pdf. Retrieved July 30, 2014.
- ↑ "Questions and Answers: FDA announces new requirements for over-the-counter (OTC) sunscreen products marketed in the U.S.". 2011-06-23. https://www.fda.gov/Drugs/ResourcesForYou/Consumers/BuyingUsingMedicineSafely/UnderstandingOver-the-CounterMedicines/ucm258468.htm.
- ↑ Department of Health and Human Services: Food and Drug Administration (June 17, 2011). "Sunscreen Drug Products for Over-the-Counter Human Use; Final Rules and Proposed Rules". Federal Register 76 (117): 35620–35665. http://www.gpo.gov/fdsys/pkg/FR-2011-06-17/pdf/2011-14766.pdf. Retrieved August 19, 2014.
- ↑ Department of Health and Human Services: Food and Drug Administration (May 11, 2012). "Sunscreen Drug Products for Over-the-Counter Human Use; Delay of Compliance Dates". Federal Register 77 (92): 27591–27593. http://www.gpo.gov/fdsys/pkg/FR-2012-05-11/pdf/2012-11390.pdf. Retrieved September 27, 2012.
- ↑ "Sunscreen: FDA regulation, and environmental and health impact". Photochemical & Photobiological Sciences 19 (1): 66–70. January 2020. doi:10.1039/c9pp00366e. PMID 31845952. https://pubmed.ncbi.nlm.nih.gov/31845952. Retrieved February 20, 2023.
- ↑ "History of sunscreen: An updated view". Journal of Cosmetic Dermatology 20 (4): 1044–1049. April 2021. doi:10.1111/jocd.14004. PMID 33583116. https://pubmed.ncbi.nlm.nih.gov/33583116. Retrieved February 20, 2023.
- ↑ "Majority of sunscreens could flunk proposed FDA standards for safety and efficacy, report to say". CNN. May 21, 2019. https://www-m.cnn.com/2019/05/15/health/sunscreen-fda-safety-standards-study/index.html?r=https%3A%2F%2Fwww.cnn.com%2Fsearch%2F%3Fq%3DSunscreen.
- ↑ 159.0 159.1 159.2 "Sunscreen products: Rationale for use, formulation development and regulatory considerations". Saudi Pharmaceutical Journal 27 (7): 1009–1018. November 2019. doi:10.1016/j.jsps.2019.08.003. PMID 31997908.
- ↑ 160.00 160.01 160.02 160.03 160.04 160.05 160.06 160.07 160.08 160.09 "Sunscreen Regulation in the World". Sunscreens in Coastal Ecosystems. The Handbook of Environmental Chemistry. 94. Cham: Springer International Publishing. 2020. pp. 15–35. doi:10.1007/698_2019_440. ISBN 978-3-030-56076-8.
- ↑ "Sunscreen bans: Coral reefs and skin cancer". Journal of Clinical Pharmacy and Therapeutics 44 (1): 134–139. February 2019. doi:10.1111/jcpt.12778. PMID 30484882.
- ↑ 162.0 162.1 "Toxicopathological Effects of the Sunscreen UV Filter, Oxybenzone (Benzophenone-3), on Coral Planulae and Cultured Primary Cells and Its Environmental Contamination in Hawaii and the U.S. Virgin Islands". Archives of Environmental Contamination and Toxicology 70 (2): 265–288. February 2016. doi:10.1007/s00244-015-0227-7. PMID 26487337. https://repository.library.noaa.gov/view/noaa/13720. Retrieved April 2, 2023.
- ↑ "Some Sunscreens May Kill Corals. Should They Be Banned?". https://www.pewtrusts.org/en/research-and-analysis/blogs/stateline/2019/03/07/some-sunscreens-may-kill-corals-should-they-be-banned.
- ↑ "What is coral bleaching?" (in EN-US). National Oceanic and Atmospheric Administration. https://oceanservice.noaa.gov/facts/coral_bleach.html.
- ↑ "Coral decline—is sunscreen a scapegoat?" (in en). https://phys.org/news/2021-02-coral-declineis-sunscreen-scapegoat.html.
- ↑ "Examine all available evidence before making decisions on sunscreen ingredient bans". The Science of the Total Environment 674: 211–212. July 2019. doi:10.1016/j.scitotenv.2019.04.137. PMID 31004897. Bibcode: 2019ScTEn.674..211S. https://linkinghub.elsevier.com/retrieve/pii/S004896971931664X. Retrieved October 27, 2021.
- ↑ "New study measures UV-filters in seawater and corals from Hawaii". University of Maryland Center for Environmental Science. 1 April 2019. https://www.umces.edu/content/new-study-measures-uv-filters-seawater-and-corals-hawaii.
- ↑ "Occurrence and distribution of UV-filters and other anthropogenic contaminants in coastal surface water, sediment, and coral tissue from Hawaii". The Science of the Total Environment 670: 398–410. June 2019. doi:10.1016/j.scitotenv.2019.03.034. PMID 30904653. Bibcode: 2019ScTEn.670..398M.
- ↑ "Sunscreen use and awareness of chemical toxicity among beach goers in Hawaii prior to a ban on the sale of sunscreens containing ingredients found to be toxic to coral reef ecosystems". Marine Policy 117: 103875. July 2020. doi:10.1016/j.marpol.2020.103875. ISSN 0308-597X. https://www.sciencedirect.com/science/article/pii/S0308597X19309029.
- ↑ "Coral: Palau to ban sunscreen products to protect reefs". 1 November 2018. https://www.bbc.com/news/science-environment-46046064.
- ↑ "Sunscreens as a source of hydrogen peroxide production in coastal waters". Environmental Science & Technology 48 (16): 9037–9042. August 2014. doi:10.1021/es5020696. PMID 25069004. Bibcode: 2014EnST...48.9037S.
- ↑ "Sunscreen products increase virus production through prophage induction in marine bacterioplankton". Microbial Ecology 45 (2): 109–118. February 2003. doi:10.1007/s00248-002-1033-0. PMID 12545312.
- ↑ "Sunscreens cause coral bleaching by promoting viral infections". Environmental Health Perspectives 116 (4): 441–447. April 2008. doi:10.1289/ehp.10966. PMID 18414624.
- ↑ "Sunscreen Safety: a Review of Recent Studies on Humans and the Environment" (in en). Current Dermatology Reports 9 (1): 1–9. 2020-03-01. doi:10.1007/s13671-020-00284-4. ISSN 2162-4933. https://doi.org/10.1007/s13671-020-00284-4.
- ↑ "A sunblock based on bioadhesive nanoparticles". Nature Materials 14 (12): 1278–1285. December 2015. doi:10.1038/nmat4422. PMID 26413985. Bibcode: 2015NatMa..14.1278D.
- ↑ "Towards symmetry driven and nature inspired UV filter design". Nature Communications 10 (1): 4748. October 2019. doi:10.1038/s41467-019-12719-z. PMID 31628301. Bibcode: 2019NatCo..10.4748H.
- ↑ Tortini, Guido; Ziosi, Paola; Cesa, Elena; Molesini, Sonia; Baldini, Erika; De Lucia, Daniela; Rossi, Caterina; Durini, Elisa et al. (June 2022). "Criticisms in the Development of High-Protection and Broad-Spectrum "Natural/Organic" Certifiable Sunscreen" (in en). Cosmetics 9 (3): 56. doi:10.3390/cosmetics9030056. ISSN 2079-9284.
External links
- Does it work, or not? – illustrated explanation of how UV light is absorbed by chemicals in sunscreen from Wired
- 56% of Americans Rarely or Never Use Sunscreen – A survey conducted about the sunscreen habits of modern Americans.
 |
