Medicine:Aneurysm
| Aneurysm | |
|---|---|
| Other names | Aneurism |
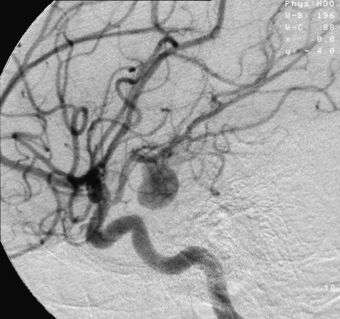 | |
| Angiography of an aneurysm in a brain artery. The aneurysm is the large bulge in the center of the image. | |
| Specialty | Vascular surgery |
An aneurysm is an outward bulging, likened to a bubble or balloon, caused by a localized, abnormal, weak spot on a blood vessel wall.[1] Aneurysms may be a result of a hereditary condition or an acquired disease. Aneurysms can also be a nidus (starting point) for clot formation (thrombosis) and embolization. As an aneurysm increases in size, the risk of rupture, which leads to uncontrolled bleeding, increases.[2] Although they may occur in any blood vessel, particularly lethal examples include aneurysms of the circle of Willis in the brain, aortic aneurysms affecting the thoracic aorta, and abdominal aortic aneurysms. Aneurysms can arise in the heart itself following a heart attack, including both ventricular and atrial septal aneurysms. There are congenital atrial septal aneurysms, a rare heart defect.
Etymology
The word is from Greek: ἀνεύρυσμα, aneurysma, "dilation", from ἀνευρύνειν, aneurynein, "to dilate".
Classification
Aneurysms are classified by type, morphology, or location.
True and false aneurysms
A true aneurysm is one that involves all three layers of the wall of an artery (intima, media and adventitia). True aneurysms include atherosclerotic, syphilitic, and congenital aneurysms, as well as ventricular aneurysms that follow transmural myocardial infarctions (aneurysms that involve all layers of the attenuated wall of the heart are also considered true aneurysms).[3]
A false aneurysm, or pseudoaneurysm, is a collection of blood leaking completely out of an artery or vein but confined next to the vessel by the surrounding tissue. This blood-filled cavity will eventually either thrombose (clot) enough to seal the leak or rupture out of the surrounding tissue.[3]: 357
Pseudoaneurysms can be caused by trauma that punctures the artery, such as knife and bullet wounds,[4] as a result of percutaneous surgical procedures such as coronary angiography or arterial grafting,[5] or use of an artery for injection.[6]
Morphology
Aneurysms can also be classified by their macroscopic shapes and sizes and are described as either saccular or fusiform. The shape of an aneurysm is not specific for a specific disease.[3]: 357 The size of the base or neck is useful in determining the chance of for example endovascular coiling.[7]
Saccular aneurysms, or "berry" aneurysms, are spherical in shape and involve only a portion of the vessel wall; they usually range from 5 to 20 cm (2.0 to 7.9 in) in diameter, and are often filled, either partially or fully, by a thrombus.[3]: 357 Saccular aneurysms have a "neck" that connects the aneurysm to its main ("parent") artery, a larger, rounded area, called the dome.[citation needed]
Fusiform aneurysms ("spindle-shaped" aneurysms) are variable in both their diameter and length; their diameters can extend up to 20 cm (7.9 in). They often involve large portions of the ascending and transverse aortic arch, the abdominal aorta, or, less frequently, the iliac arteries.[3]: 357
Location
Aneurysms can also be classified by their location:

- Arterial and venous, with arterial being more common.[8]
- The heart, including coronary artery aneurysms, ventricular aneurysms, aneurysm of sinus of Valsalva, and aneurysms following cardiac surgery.
- The aorta, namely aortic aneurysms including thoracic aortic aneurysms and abdominal aortic aneurysms.[9]
- The brain, including cerebral aneurysms, berry aneurysms, and Charcot–Bouchard aneurysms.
- The legs, including the popliteal arteries.[10]
- The kidney, including renal artery aneurysms and intraparenchymal aneurysms.[11]
- Capillaries, specifically capillary aneurysms.
- The large vessels such as external and internal jugular veins[12]
Cerebral aneurysms, also known as intracranial or brain aneurysms, occur most commonly in the anterior cerebral artery, which is part of the circle of Willis. This can cause severe strokes leading to death. The next most common sites of cerebral aneurysm occurrence are in the internal carotid artery.[13]
Size
| Ectatic or mild dilatation |
>2.0 cm and <3.0 cm[14] |
| Moderate | 3.0–5.0 cm[14] |
| Large or severe | >5.0[14] or 5.5[15] cm |
Abdominal aortic aneurysms are commonly divided according to their size and symptomatology. An aneurysm is usually defined as an outer aortic diameter over 3 cm (normal diameter of the aorta is around 2 cm),[16] or more than 50% of normal diameter that of a healthy individual of the same sex and age.[9][17] If the outer diameter exceeds 5.5 cm, the aneurysm is considered to be large.[15]
The common iliac artery is classified as:[18]
| Normal | Diameter ≤12 mm |
| Ectatic | Diameter 12 to 18 mm |
| Aneurysm | Diameter ≥18 mm |
Signs and symptoms
Aneurysm presentation may range from life-threatening complications of hypovolemic shock to being found incidentally on X-ray.[19] Symptoms will differ by the site of the aneurysm and can include:
Cerebral aneurysm
Symptoms can occur when the aneurysm pushes on a structure in the brain. Symptoms will depend on whether an aneurysm has ruptured or not. There may be no symptoms present at all until the aneurysm ruptures.[20] For an aneurysm that has not ruptured the following symptoms can occur:[citation needed]
- Fatigue
- Loss of perception
- Loss of balance
- Speech problems
- Double vision
For a ruptured aneurysm, symptoms of a subarachnoid hemorrhage may present:
- Severe headaches
- Loss of vision
- Double vision
- Neck pain or stiffness
- Pain above or behind the eyes
Abdominal aneurysm
File:Aortic Aneurism 76F 3D SR Nevit Dilmen.stl Abdominal aortic aneurysm involves a regional dilation of the aorta and is diagnosed using ultrasonography, computed tomography, or magnetic resonance imaging. A segment of the aorta that is found to be greater than 50% larger than that of a healthy individual of the same sex and age is considered aneurysmal.[9] Abdominal aneurysms are usually asymptomatic but in rare cases can cause lower back pain or lower limb ischemia.
Renal (kidney) aneurysm
- Flank pain and tenderness
- Hypertension
- Haematuria
- Signs of hypovolemic shock
Risk factors
Risk factors for an aneurysm include diabetes, obesity, hypertension, tobacco use, alcoholism, high cholesterol, copper deficiency, increasing age, and tertiary syphilis infection.[19]: 602 Connective tissue disorders such as Loeys-Dietz syndrome, Marfan syndrome, and certain forms of Ehlers-Danlos syndrome are also associated with aneurysms. Aneurysms, dissections, and ruptures in individuals under 40 years of age are a major diagnostic criteria of the vascular form of Ehlers-Danlos syndrome (vEDS).[21]
Specific infective causes associated with aneurysm include:
- Advanced syphilis infection resulting in syphilitic aortitis and an aortic aneurysm
- Tuberculosis, causing Rasmussen's aneurysms
- Brain infections, causing infectious intracranial aneurysms
A minority of aneurysms are associated with genetic factors. Examples include:
- Berry aneurysms of the anterior communicating artery of the circle of Willis, associated with autosomal dominant polycystic kidney disease[22]
- Familial thoracic aortic aneurysms
- Cirsoid aneurysms, secondary to congenital arteriovenous malformations
Pathophysiology
Aneurysms form for a variety of interacting reasons. Multiple factors, including factors affecting a blood vessel wall and the blood through the vessel, contribute.
The pressure of blood within the expanding aneurysm may also injure the blood vessels supplying the artery itself, further weakening the vessel wall. Without treatment, these aneurysms will ultimately progress and rupture.[23]
Infection. A mycotic aneurysm is an aneurysm that results from an infectious process that involves the arterial wall.[24] A person with a mycotic aneurysm has a bacterial infection in the wall of an artery, resulting in the formation of an aneurysm. One of the causes of mycotic aneurysms is infective endocarditis.[25] The most common locations include arteries in the abdomen, thigh, neck, and arm. A mycotic aneurysm can result in sepsis, or life-threatening bleeding if the aneurysm ruptures. Less than 3% of abdominal aortic aneurysms are mycotic aneurysms.[26]
Syphilis. The third stage of syphilis also manifests as aneurysm of the aorta, which is due to loss of the vasa vasorum in the tunica adventitia.[27]
Copper deficiency. A minority of aneurysms are caused by copper deficiency, which results in a decreased activity of the lysyl oxidase enzyme, affecting elastin, a key component in vessel walls.[28][29][30] Copper deficiency results in vessel wall thinning,[31] and thus has been noted as a cause of death in copper-deficient humans,[32] chickens, and turkeys.[33]
Mechanics
Aneurysmal blood vessels are prone to rupture under normal blood pressure and flow due to the special mechanical properties that make them weaker. To better understand this phenomenon, we can first look at healthy arterial vessels which exhibit a J-shaped stress-strain curve with high strength and high toughness[clarify] (for a biomaterial in vivo).[34] Unlike crystalline materials whose linear elastic region follows Hooke's Law under uniaxial loading, many biomaterials exhibit a J-shaped stress-strain curve which is non-linear and concave up.[34] The blood vessel can be under large strain, or the amount of stretch the blood vessel can undergo, for a range of low applied stress before fracture, as shown by the lower part of the curve. The area under the curve up to a given strain is much lower than that for the equivalent Hookean curve, which is correlated to toughness. Toughness is defined as the amount of energy per unit volume material can absorb before rupturing. Because the amount of energy released is proportional to the amount of crack propagation, the blood vessel wall can withstand pressure and is "tough." Thus, healthy blood vessels with the mechanical properties of the J-shaped stress-strain curve have greater stability against aneurysms than materials with linear elasticity.[citation needed]
Blood vessels with aneurysms, on the other hand, are under the influence of an S-shaped stress-strain curve. As a visual aid, aneurysms can be understood as a long, cylindrical balloon. Because it's a tight balloon under pressure, it can pop at any time stress beyond a certain force threshold is applied. In the same vein, an unhealthy blood vessel has elastic instabilities that lead to rupture.[34] Initially, for a given radius and pressure, stiffness of the material increases linearly. At a certain point, the stiffness of the arterial wall starts to decrease with increasing load. At higher strain values, the area under the curve increases, thus increasing the impact on the material that would promote crack propagation. The differences in the mechanical properties of the aneurysmal blood vessels and the healthy blood vessels stem from the compositional differences of the vessels. Compared to normal aortas, aneurysmal aortas have a much higher volume fraction of collagen and ground substance (54.8% vs. 95.6%) and a much lower volume fraction of elastin (22.7% vs. 2.4%) and smooth muscles (22.6% vs. 2.2%), which contribute to higher initial stiffness.[35] It was also found that the ultimate tensile strength, or the strength to withstand rupture, of aneurysmal vessel wall is 50% lower than that of normal aortas.[36] The wall strength of ruptured aneurysmal aortic wall was also found to be 54.2 N/cm2, which is much lower than that of a repaired aorta wall, 82.3 N/cm2.[36] Due to the change in composition of the arterial wall, aneurysms overall have much lower strength to resist rupture. Predicting the risk of rupture is difficult due to the regional anisotropy the hardened blood vessels exhibit, meaning that the stress and strength values vary depending on the region and the direction of the vessel they are measured along.[37]
Diagnosis

Diagnosis of a ruptured cerebral aneurysm is commonly made by finding signs of subarachnoid hemorrhage on a computed tomography (CT) scan. If the CT scan is negative but a ruptured aneurysm is still suspected based on clinical findings, a lumbar puncture can be performed to detect blood in the cerebrospinal fluid. Computed tomography angiography (CTA) is an alternative to traditional angiography and can be performed without the need for arterial catheterization. This test combines a regular CT scan with a contrast dye injected into a vein. Once the dye is injected into a vein, it travels to the cerebral arteries, and images are created using a CT scan. These images show exactly how blood flows into the brain arteries.[38]
Treatment
Historically, the treatment of arterial aneurysms has been limited to either surgical intervention or watchful waiting in combination with control of blood pressure. At least, in the case of abdominal aortic aneurysm (AAA), the decision does not come without significant risk and cost, hence, there is a great interest in identifying more advanced decision-making approaches that are not solely based on the AAA diameter, but involve other geometrical and mechanical nuances such as local thickness and wall stress.[9] In recent years,[when?] endovascular or minimally invasive techniques have been developed for many types of aneurysms. Aneurysm clips are used for surgical procedure i.e. clipping of aneurysms.[39]
Intracranial
There are currently two treatment options for brain aneurysms: surgical clipping or endovascular coiling. There is currently debate in the medical literature about which treatment is most appropriate given particular situations.[40]
Surgical clipping was introduced by Walter Dandy of the Johns Hopkins Hospital in 1937. It consists of a craniotomy to expose the aneurysm and closing the base or neck of the aneurysm with a clip. The surgical technique has been modified and improved over the years.[citation needed]
Endovascular coiling was introduced by Italian neurosurgeon Guido Guglielmi at UCLA in 1989. It consists of passing a catheter into the femoral artery in the groin, through the aorta, into the brain arteries, and finally into the aneurysm itself. Platinum coils initiate a clotting reaction within the aneurysm that, if successful, fills the aneurysm dome and prevents its rupture.[41] A flow diverter can be used, but risks complications.[42]
Aortic and peripheral
For aneurysms in the aorta, arms, legs, or head, the weakened section of the vessel may be replaced by a bypass graft that is sutured at the vascular stumps. Instead of sewing, the graft tube ends, made rigid and expandable by nitinol wireframe, can be easily inserted in its reduced diameter into the vascular stumps and then expanded up to the most appropriate diameter and permanently fixed there by external ligature.[43][44] New devices were recently developed to substitute the external ligature by expandable ring allowing use in acute ascending aorta dissection, providing airtight (i.e. not dependent on the coagulation integrity), easy and quick anastomosis extended to the arch concavity[45][46][47] Less invasive endovascular techniques allow covered metallic stent grafts to be inserted through the arteries of the leg and deployed across the aneurysm.
Renal
Renal aneurysms are very rare consisting of only 0.1–0.09%[48] while rupture is even more rare.[48][49] Conservative treatment with control of concomitant hypertension being the primary option with aneurysms smaller than 3 cm. If symptoms occur, or enlargement of the aneurysm, then endovascular or open repair should be considered.[50] Pregnant women (due to high rupture risk of up to 80%) should be treated surgically.[51]
Epidemiology
Incidence rates of cranial aneurysms are estimated at between 0.4% and 3.6%. Those without risk factors have expected prevalence of 2–3%.[13]: 181 In adults, females are more likely to have aneurysms. They are most prevalent in people ages 35 – 60 but can occur in children as well. Aneurysms are rare in children with a reported prevalence of .5% to 4.6%. The most common incidence is among 50-year-olds, and there are typically no warning signs. Most aneurysms develop after the age of 40. [citation needed]
Pediatric aneurysms
Pediatric aneurysms have different incidences and features than adult aneurysms.[52] Intracranial aneurysms are rare in childhood, with over 95% of all aneurysms occurring in adults.[13]: 235
Risk factors
Incidence rates are two to three times higher in males, while there are more large and giant aneurysms and fewer multiple aneurysms.[13]: 235 Intracranial hemorrhages are 1.6 times more likely to be due to aneurysms than cerebral arteriovenous malformations in whites, but four times less in certain Asian populations.[13]: 235
Most patients, particularly infants, present with subarachnoid hemorrhage and corresponding headaches or neurological deficits. The mortality rate for pediatric aneurysms is lower than in adults.[13]: 235
Modeling

Modeling of aneurysms consists of creating a 3D model that mimics a particular aneurysm. Using patient data for the blood velocity, and blood pressure, along with the geometry of the aneurysm, researchers can apply computational fluid dynamics (CFD) to predict whether an aneurysm is benign or if it is at risk of complication. One risk is rupture. Analyzing the velocity and pressure profiles of the blood flow leads to obtaining the resulting wall shear stress on the vessel and aneurysm wall. The neck of the aneurysm is the most at risk due to the combination of a small wall thickness and high wall shear stress. When the wall shear stress reaches its limit, the aneurysm ruptures, leading to intracranial hemorrhage. Conversely, another risk of aneurysms is the creation of clots. Aneurysms create a pocket which diverts blood flow. This diverted blood flow creates a vortex inside of the aneurysm. This vortex can lead to areas inside of the aneurysm where the blood flow is stagnant, which promotes formations of clots. Blood clots can dislodge from the aneurysm, which can then lead to an embolism when the clot gets stuck and disrupts blood flow. Model analysis allows these risky aneurysms to be identified and treated.[53][54][55][56]
In the past, aneurysms were modeled as rigid spheres with linear inlets and outlets. As technology advances, the ability to detect and analyze aneurysms becomes easier. Researchers are able to CT scan a patient's body to create a 3D computer model that possesses the correct geometry. Aneurysms can now be modeled with their distinctive "balloon" shape. Nowadays researchers are optimizing the parameters required to accurately model a patient's aneurysm that will lead to a successful intervention. Current modeling is not able to take into account all variables though. For example, blood is considered to be a non-Newtonian fluid. Some researchers treat blood as a Newtonian fluid instead, as it sometimes has negligible effects to the analysis in large vessels. When analyzing small vessels though, such as those present in intracranial aneurysms. Similarly, sometimes it is difficult to model the varying wall thickness in small vessels, so researchers treat wall thickness as constant. Researchers make these assumptions to reduce computational time. Nonetheless, making erroneous assumptions could lead to a misdiagnosis that could put a patient's life at risk.[53][57][58][59]
Notable cases
- U.S. Senator Joe Biden, who later became President, had two brain aneurysms in 1988. He recovered after successful surgeries to correct them.
- Lucille Ball died from an aortic rupture in the abdominal area days after having undergone apparently successful heart surgery for a dissecting aortic aneurysm.[60][61][62]
- Laura Branigan died of a cerebral aneurysm.
- David Cone had an aneurysm and missed most of the 1996 baseball season.
- Davie Cooper died in 1995 following a subarachnoid hemorrhage whilst filming a football television series.
- John Olerud had an aneurysm in 1989 and has worn a batting helmet on the field all of his career since then.
- Albert Einstein died from a repaired aortic aneurysm.[63]
- Thomas Mikal Ford died from a ruptured aneurysm in his abdomen at age 52.
- Charles de Gaulle died from a ruptured aneurysm within his neck.[64]
- Richard Holbrooke died from a thoracic aortic aneurysm.[65]
- Édith Piaf died from an aneurysm due to liver failure.
- Stuart Sutcliffe died from an aneurysm in his brain's right hemisphere.[66]
- Raymond F. Boyce died in 1974 as a result of an aneurysm.[67]
- John Ritter died in 2003 of a misdiagnosed thoracic aortic dissection (aortic aneurysm).[68][69]
- Isabel Granada died of a cerebral aneurysm.[70][71]
- Geoffrey Thompson died of a brain aneurysm at his daughter's wedding, hosted at his theme park, Blackpool Pleasure Beach.
- Edwin Rosario died of an aneurysm in 1997.
- Joni Mitchell had a brain aneurysm in 2015, but survived.
- Grant Imahara died from a brain aneurysm in July 2020.
- Dr. Dre had a brain aneurysm in January 2021.
- Jovit Baldivino died from a brain aneurysm in December 2022.
- Tom Sizemore died from a brain aneurysm in March 2023.
References
- ↑ "Aneurysms". https://www.nhlbi.nih.gov/health-topics/aneurysm.
- ↑ "Actuarial analysis of variables associated with rupture of small abdominal aortic aneurysms". Surgery 98 (3): 472–83. September 1985. PMID 3898453.
- ↑ 3.0 3.1 3.2 3.3 3.4 Kumar, Vinay, ed (2007). Robbins basic pathology (8th ed.). Philadelphia: Saunders/Elsevier.
- ↑ "The False Aneurysm". Canadian Medical Association Journal 91 (6): 281–84. August 1964. PMID 14180533.
- ↑ "The changing face of femoral artery false aneurysms". European Journal of Vascular and Endovascular Surgery 27 (4): 385–88. April 2004. doi:10.1016/j.ejvs.2004.01.001. PMID 15015188.
- ↑ "Management of femoral artery pseudoaneurysm due to addictive drug injection". Chinese Journal of Traumatology = Zhonghua Chuang Shang Za Zhi 7 (4): 244–46. August 2004. PMID 15294105.
- ↑ "Endovascular treatment of intracranial aneurysms: review of current practice". Postgraduate Medical Journal 87 (1023): 41–50. January 2011. doi:10.1136/pgmj.2010.105387. PMID 20937736.
- ↑ Perrin, Michel (17 February 2010). "Venous aneurysms" (in en-US). https://www.phlebolymphology.org/venous-aneurysms/.
- ↑ 9.0 9.1 9.2 9.3 "Mechanical and geometrical determinants of wall stress in abdominal aortic aneurysms: A computational study". PLOS ONE 13 (2): e0192032. February 2018. doi:10.1371/journal.pone.0192032. PMID 29401512. Bibcode: 2018PLoSO..1392032A.
- ↑ "Abdominal Aortic Aneurysms". 16 October 2020. https://www.lecturio.com/concepts/abdominal-aortic-aneurysms/.
- ↑ "A Cough Deteriorating Gross Hematuria: A Clinical Sign of a Forthcoming Life-Threatening Rupture of an Intraparenchymal Aneurysm of Renal Artery (Wunderlich's Syndrome)". Case Reports in Vascular Medicine 2013: 452317. 2013. doi:10.1155/2013/452317. PMID 23864981.
- ↑ Avinash P Mural International Journal of Medical Case Reports Vol 3 Iss. 4, pp. 1–4 http://ijomcr.net/saccular-aneurysm-of-external-jugularvein/
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 Christianto B. Lumenta, ed (2010). Neurosurgery. Heidelberg: Springer. p. 181. ISBN 978-3-540-79564-3. https://archive.org/details/neurosurgery00lume_223.
- ↑ 14.0 14.1 14.2 Lumb, Philip (2014). Critical Care Ultrasound E-Book. Elsevier Health Sciences. p. 56. ISBN 978-0323278171. https://books.google.com/books?id=zbydAgAAQBAJ&pg=PA56. Retrieved 23 August 2017.
- ↑ 15.0 15.1 "Screening for abdominal aortic aneurysms: single centre randomised controlled trial". BMJ 330 (7494): 750. April 2005. doi:10.1136/bmj.38369.620162.82. PMID 15757960.
- ↑ "ACC/AHA Guidelines for the Management of Patients with Peripheral Arterial Disease". Journal of Vascular and Interventional Radiology 17 (9): 1383–97; quiz 1398. September 2006. doi:10.1097/01.RVI.0000240426.53079.46. PMID 16990459.
- ↑ "Clinical practice. Abdominal aortic aneurysms". The New England Journal of Medicine 371 (22): 2101–08. November 2014. doi:10.1056/NEJMcp1401430. PMID 25427112.
- ↑ Melissa L Kirkwood. "Iliac artery aneurysm". https://www.uptodate.com/contents/iliac-artery-aneurysm. Last updated: 27 March 2017.
- ↑ 19.0 19.1 Walker, Brian R.; Colledge, Nicki R; Ralston, Stuart H. (2010). Davidson's principles and practice of medicine (21st ed.). Edinburgh: Churchill Livingstone/Elsevier. p. 604. ISBN 978-0-7020-3085-7. https://archive.org/details/davidsonsprincip00frcp.
- ↑ Manasco, Hunter. "The Aphasias". Introduction to Neurogenic Communication Disorders. p. 93.
- ↑ Byers PH. Vascular Ehlers-Danlos Syndrome. 1999 Sep 2 [Updated 2019 Feb 21]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2020. Available from: https://www.ncbi.nlm.nih.gov/sites/books/NBK1494/
- ↑ Schueler, Stephen J.; Beckett, John H.; Gettings, D. Scott (18 August 2010). "Berry Aneurysm in the Brain". freemd. http://www.freemd.com/berry-aneurysm-in-the-brain/overview.htm.
- ↑ "Natural history of unruptured intracranial aneurysms: probability of and risk factors for aneurysm rupture". Journal of Neurosurgery 108 (5): 1052–60. May 2008. doi:10.3171/JNS/2008/108/5/1052. PMID 18447733.
- ↑ emedicine – Cerebral Aneurysm Author: Jonathan L Brisman. Coauthors: Emad Soliman, Abraham Kader, Norvin Perez. Updated: 23 September 2010
- ↑ https://www.uptodate.com/contents/overview-of-infected-mycotic-arterial-aneurysm (Retrieved 8 September 2021)
- ↑ Schueler, Stephen J.; Beckett, John H.; Gettings, Scott (13 November 2011). "Mycotic Aneurysm". http://www.freemd.com/mycotic-aneurysm/overview.htm.
- ↑ "Syphilitic aneurysm of the ascending aorta". Interactive Cardiovascular and Thoracic Surgery 14 (2): 223–25. February 2012. doi:10.1093/icvts/ivr067. PMID 22159251.
- ↑ Mäki, Joni (2002). Lysyl oxidases : cloning and characterization of the fourth and the fifth human lysyl oxidase isoenzymes, and the consequences of a targeted inactivaton of the first described lysyl oxidase isoenzyme in mice. Oulu: Oulun yliopisto. ISBN 951-42-6739-7. http://herkules.oulu.fi/isbn9514267397/isbn9514267397.pdf.
- ↑ "Copper, lysyl oxidase, and extracellular matrix protein cross-linking". The American Journal of Clinical Nutrition 67 (5 Suppl): 996S–1002S. May 1998. doi:10.1093/ajcn/67.5.996S. PMID 9587142.
- ↑ "Lysyl oxidase: properties, regulation and multiple functions in biology". Matrix Biology 16 (7): 387–98. February 1998. doi:10.1016/s0945-053x(98)90012-9. PMID 9524359.
- ↑ "Is tissue copper deficiency associated with aortic aneurysms?". The British Journal of Surgery 72 (5): 352–53. May 1985. doi:10.1002/bjs.1800720507. PMID 3995240.
- ↑ "Decreased hepatic copper levels. A possible chemical marker for the pathogenesis of aortic aneurysms in man". Archives of Surgery 117 (9): 1212–13. September 1982. doi:10.1001/archsurg.1982.01380330070017. PMID 7202350.
- ↑ "Copper salts for growth stimulation and alleviation of aortic rupture losses in turkeys". Poultry Science 57 (5): 1313–24. September 1978. doi:10.3382/ps.0571313. PMID 724600.
- ↑ 34.0 34.1 34.2 "DoITPoMS – TLP Library Elasticity in Biological Materials". https://www.doitpoms.ac.uk/tlplib/bioelasticity/printall.php.
- ↑ He, Chang M.; Roach, Margot R. (July 1994). "The composition and mechanical properties of abdominal aortic aneurysms". Journal of Vascular Surgery 20 (1): 6–13. doi:10.1016/0741-5214(94)90169-4. PMID 8028090.
- ↑ 36.0 36.1 Vorp, David A.; Geest, Jonathan P. Vande (August 2005). "Biomechanical Determinants of Abdominal Aortic Aneurysm Rupture". Arteriosclerosis, Thrombosis, and Vascular Biology 25 (8): 1558–1566. doi:10.1161/01.ATV.0000174129.77391.55. ISSN 1079-5642. PMID 16055757.
- ↑ "Mechanical properties of abdominal aortic aneurysm wall". J Med Eng Technol 25 (4): 133–42. 2001. doi:10.1080/03091900110057806. ISSN 0309-1902. PMID 11601439.
- ↑ Vu, K; Kaitoukov, Y; Morin-Roy, F; Kauffmann, C; Tang, A; Giroux, C; Therasse, E; Soulez, G (2014). "Rupture signs on computed tomography, treatment, and outcome of abdominal aortic aneurysms". Insights Imaging 5 (3): 281–293. doi:10.1007/s13244-014-0327-3. PMID 24789068.
- ↑ "Aneurysm Clip". Surgical Units. 18 September 2016. http://surgicalunits.com/aneurysm-clip-311.html.
- ↑ "Microsurgical clipping and endovascular coiling of intracranial aneurysms: a critical review of the literature". Neurosurgery 62 (6): 1187–202; discussion 1202–3. June 2008. doi:10.1227/01.neu.0000333291.67362.0b. PMID 18824986.
- ↑ "History of endovascular endosaccular occlusion of brain aneurysms: 1965–1990". Interventional Neuroradiology 13 (3): 217–24. September 2007. doi:10.1177/159101990701300301. PMID 20566113.
- ↑ "Flow-diverter devices in the treatment of intracranial aneurysms: A meta-analysis and systematic review". The Neuroradiology Journal 29 (1): 66–71. February 2016. doi:10.1177/1971400915621321. PMID 26838174.
- ↑ Nazari, S. (2010). "sp.html". Interactive Cardiovascular and Thoracic Surgery (Fondazionecarrel.org) 10 (2): 161–4. doi:10.1510/icvts.2009.216291. PMID 19933306. http://www.fondazionecarrel.org/sp/sp.html. Retrieved 30 May 2014.
- ↑ "Improved device for sutureless aortic anastomosis applied in a case of cancer". Texas Heart Institute Journal 29 (1): 56–9. 2002. PMID 11995854.
- ↑ "Expandable device type III for easy and reliable approximation of dissection layers in sutureless aortic anastomosis. Ex vivo experimental study". Interactive Cardiovascular and Thoracic Surgery 10 (2): 161–4. February 2010. doi:10.1510/icvts.2009.216291. PMID 19933306.
- ↑ Stefano Nazari. "Expandable device type III for easy and reliable approximation of dissection layers in sutureless aortic anastomosis. Ex vivo experimental study". Icvts.ctsnetjournals.org. http://icvts.ctsnetjournals.org/cgi/content/full/10/2/161.
- ↑ Nazari, S. (2010). "ndicvts.html". Interactive Cardiovascular and Thoracic Surgery (Fondazionecarrel.org) 10 (2): 161–4. doi:10.1510/icvts.2009.216291. PMID 19933306. http://www.fondazionecarrel.org/ejcts/ndaod&wikien111.html. Retrieved 30 May 2014.
- ↑ 48.0 48.1 "Kidney salvage in a case of ruptured renal artery aneurysm: case report and literature review". Cardiovasc Surg. 5 (1): 134–136. 1997. doi:10.1016/s0967-2109(95)00041-0. PMID 9158136.
- ↑ "Renal artery aneurysms. Natural history and prognosis". Annals of Surgery 197 (3): 348–52. March 1983. doi:10.1097/00000658-198303000-00016. PMID 6830341.
- ↑ Uflacker R. Interventional management of visceral artery aneurysms. In: Strandness DE, ed. Vascular Diseases: Surgical & Interventional Therapy. New York, NY: Churchill Livingstone; 1994:823–844.
- ↑ "Renal artery an?eurysm: a report of 28 cases". Cardiovasc Surg. 4 (2): 185–189. 1996. doi:10.1016/0967-2109(96)82312-X. PMID 8861434.
- ↑ "Brain Aneurysm Basics | The Brain Aneurysm Foundation". Bafound.org. http://www.bafound.org/Pediatric_Aneurysms.
- ↑ 53.0 53.1 Nabong, Jennica Rica; David, Guido (October 2017). "Finite element model of size, shape and blood pressure on rupture of intracranial saccular aneurysms". Journal of Physics: Conference Series 893 (1): 012054. doi:10.1088/1742-6596/893/1/012054. ISSN 1742-6596. Bibcode: 2017JPhCS.893a2054R.
- ↑ Algabri, Y. A.; Rookkapan, S.; Chatpun, S. (September 2017). "Three-dimensional finite volume modelling of blood flow in simulated angular neck abdominal aortic aneurysm". IOP Conference Series: Materials Science and Engineering 243 (1): 012003. doi:10.1088/1757-899X/243/1/012003. ISSN 1757-899X. Bibcode: 2017MS&E..243a2003A.
- ↑ Sarrami-Foroushani, Ali; Lassila, Toni; Hejazi, Seyed Mostafa; Nagaraja, Sanjoy; Bacon, Andrew; Frangi, Alejandro F. (25 June 2019). "A computational model for prediction of clot platelet content in flow-diverted intracranial aneurysms". Journal of Biomechanics 91: 7–13. doi:10.1016/j.jbiomech.2019.04.045. ISSN 0021-9290. PMID 31104921.
- ↑ Zhong, Liang; Zhang, Jun-Mei; Su, Boyang; Tan, Ru San; Allen, John C.; Kassab, Ghassan S. (26 June 2018). "Application of Patient-Specific Computational Fluid Dynamics in Coronary and Intra-Cardiac Flow Simulations: Challenges and Opportunities". Frontiers in Physiology 9: 742. doi:10.3389/fphys.2018.00742. ISSN 1664-042X. PMID 29997520.
- ↑ Liepsch, D.; Sindeev, S.; Frolov, S. (August 2018). "An impact of non-Newtonian blood viscosity on hemodynamics in a patient-specific model of a cerebral aneurysm". Journal of Physics: Conference Series 1084 (1): 012001. doi:10.1088/1742-6596/1084/1/012001. ISSN 1742-6596. Bibcode: 2018JPhCS1084a2001L.
- ↑ Thenier-Villa, José Luis; Riveiro Rodríguez, Antonio; Martínez-Rolán, Rosa María; Gelabert-González, Miguel; González-Vargas, Pedro Miguel; Galarraga Campoverde, Raúl Alejandro; Díaz Molina, Jorge; De La Lama Zaragoza, Adolfo et al. (1 October 2018). "Hemodynamic Changes in the Treatment of Multiple Intracranial Aneurysms: A Computational Fluid Dynamics Study". World Neurosurgery 118: e631–e638. doi:10.1016/j.wneu.2018.07.009. ISSN 1878-8750. PMID 30017759.
- ↑ Sforza, Daniel M.; Putman, Christopher M.; Cebral, Juan R. (June 2012). "Computational fluid dynamics in brain aneurysms". International Journal for Numerical Methods in Biomedical Engineering 28 (6–7): 801–808. doi:10.1002/cnm.1481. ISSN 2040-7939. PMID 25364852.
- ↑ Ball, Lucille (27 April 1989). "Lucy dies". Chicago Tribune. http://articles.chicagotribune.com/1989-04-27/news/8904070889_1_love-lucy-lucy-ricardo-fred-mertz.
- ↑ "Article: Lucille Ball, Pioneer of Television Comedy, Dies at 77". http://www.highbeam.com/doc/1P2-1187461.html.
- ↑ Ball, Lucille (27 April 1989). "Ball dies of ruptured aorta". Los Angeles Times. http://articles.latimes.com/1989-04-27/news/mn-1618_1_love-lucy-aorta-emergency-heart-surgery.
- ↑ "Dr. Albert Einstein Dies in Sleep at 76; World Mourns Loss of Great Scientist". The New York Times. 19 April 1955. https://www.nytimes.com/learning/general/onthisday/bday/0314.html.
- ↑ "World Leaders to Gather in Paris to Honour General de Gaulle". The Times. 11 November 1970.
- ↑ "US Diplomat Holbrooke dies after tearing arota". 14 December 2010. http://www.nbcnews.com/id/40649624/ns/politics/t/us-diplomat-holbrooke-dies-after-tearing-aorta/.
- ↑ Wright, Jade (9 April 2012). "Stuart Sutcliffe: Legacy of the fifth Beatle 50 years after his death". Echo. http://www.liverpoolecho.co.uk/news/liverpool-news/stuart-sutcliffe-legacy-fifth-beatle-3346849.
- ↑ Chamberlin, Donald (July 21, 2009). "Chamberlin, Donald oral history" (PDF). Oral history collection (Interview). Interviewed by Paul McJones. Mountain View, California: Computer History Museum. p. 19.
- ↑ Considine, Bob (4 February 2008). "John Ritter's widow talks about wrongful death suit". today.com. http://www.today.com/id/22989512.
- ↑ "John Ritter: 1948–2003". people.com. 18 September 2003. p. 2. http://www.people.com/people/article/0,,628262_2,00.html.
- ↑ Roxas, Patricia Ann (25 October 2017). "Report: Isabel Granada in coma in Qatar hospital". Inquirer.net. http://entertainment.inquirer.net/247654/report-isabel-granada-coma-qatar-hospital.
- ↑ ALG (5 November 2017). "Isabel Granada passes away in Qatar". GMA News. http://www.gmanetwork.com/news/showbiz/chikaminute/631946/isabel-granada-passes-away-in-qatar/story/.
External links
| Classification | |
|---|---|
| External resources |
 |
