Biology:Paneth cell
This article needs more medical references for verification or relies too heavily on primary sources. (April 2020) |
| Paneth cell | |
|---|---|
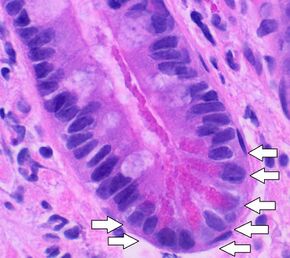 Paneth cells, located at the base of the crypts of the small intestinal mucosa, and displaying bright red cytoplasmic granules. H&E stain. | |
| Details | |
| Location | Small intestine epithelium |
| Identifiers | |
| Latin | cellula panethensis |
| Anatomical terms of microanatomy | |
Paneth cells are cells in the small intestine epithelium, alongside goblet cells, enterocytes, and enteroendocrine cells.[1] Some can also be found in the cecum and appendix. They are located below the intestinal stem cells in the intestinal glands (also called crypts of Lieberkühn) and the large eosinophilic refractile granules that occupy most of their cytoplasm.
When exposed to bacteria or bacterial antigens, Paneth cells secrete several anti-microbial compounds (notably defensins and lysozyme) that are known to be important in immunity and host-defense into the lumen of the intestinal gland, thereby contributing to maintenance of the gastrointestinal barrier by controlling the enteric bacteria. Therefore, Paneth cells play a role in the innate immune system.
Paneth cells are named after 19th-century pathologist Joseph Paneth.
Structure

Paneth cells are found throughout the small intestine and the appendix at the base of the intestinal glands.[2] There is an increase in Paneth cell numbers towards the end of the small intestine.[3] Like the other epithelial cell lineages in the small intestine, Paneth cells originate at the stem cell region near the bottom of the gland.[4] There are on average 5–12 Paneth cells in each small intestinal crypt.[5]
Unlike the other epithelial cell types, Paneth cells migrate downward from the stem cell region and settle just adjacent to it.[4] This close relationship to the stem cell region suggests that Paneth cells are important in defending the gland stem cells from microbial damage,[4] although their function is not entirely known.[2] Furthermore, among the four aforementioned intestinal cell lineages, Paneth cells live the longest (approximately 57 days).[6]
Function
Paneth cells secrete antimicrobial peptides and proteins, which are "key mediators of host-microbe interactions, including homeostatic balance with colonizing microbiota and innate immune protection from enteric pathogens."[7]
Small intestinal crypts house stem cells that serve to constantly replenish epithelial cells that die and are lost from the villi.[7] Paneth cells support the physical barrier of the epithelium by providing essential niche signals to their neighboring intestinal stem cells. Protection and stimulation of these stem cells is essential for long-term maintenance of the intestinal epithelium, in which Paneth cells play a critical role.[8]
Paneth cells display merocrine secretion, that is, secretion via exocytosis.[9]
Sensing microbiota
Paneth cells are stimulated to secrete defensins when exposed to bacteria (both Gram positive and Gram-negative types), or such bacterial products as lipopolysaccharide, lipoteichoic acid, muramyl dipeptide and lipid A.[10] They are also stimulated by cholinergic signaling normally preceding the arrival of food which potentially may contain a new bacterial load.[10]
Paneth cells sense bacteria via MyD88-dependent toll-like receptor (TLR) activation which then triggers antimicrobial action.[11] For example, research showed that in the secretory granules, murine and human Paneth cells express high levels of TLR9. TLR9 react to CpG-ODN and unmethylated oligonucleotides, pathogen-associated molecular patterns (PAMPs) typical for bacterial DNA. Internalizing these PAMPs and activating TLR9 leads to degranulation and release of antimicrobial peptides and other secretions.[12] Surprisingly, murine Paneth cells do not express mRNA transcripts for TLR4.[5]
Antimicrobial secretions
The principal defense molecules secreted by Paneth cells are alpha-defensins, which are known as cryptdins in mice.[13] These peptides have hydrophobic and positively charged domains that can interact with phospholipids in cell membranes. This structure allows defensins to insert into membranes, where they interact with one another to form pores that disrupt membrane function, leading to cell lysis. Due to the higher concentration of negatively charged phospholipids in bacterial than vertebrate cell membranes, defensins preferentially bind to and disrupt bacterial cells, sparing the cells they are functioning to protect.[14]
Human Paneth cells produce two α-defensins known as human α-defensin HD-5 (DEFA5) and HD-6 (DEFA6).[15] HD-5 has a wide spectrum of killing activity against both Gram positive and Gram negative bacteria as well as fungi (Listeria monocytogenes, Escherichia coli, Salmonella typhimurium, and Candida albicans).[5] The antimicrobial activity of HD-6 consists of self-assembling into extracellular nets that entrap bacteria in the intestine and thereby preventing their translocation across the epithelial barrier.[16]
Human Paneth cells also produce other AMPs including lysozyme, secretory phospholipase A2, and regenerating islet-derived protein IIIA.[17] Lysozyme is an antimicrobial enzyme that dissolves the cell walls of many bacteria, and phospholipase A2 is an enzyme specialized in the lysis of bacterial phospholipids .[10] This battery of secretory molecules gives Paneth cells a potent arsenal against a broad spectrum of agents, including bacteria, fungi and even some enveloped viruses.[18]
Secretory autophagy
During conventional protein secretion, proteins are transported through the ER-Golgi complex packaged in secretory granules and released to the extracellular space. Should invasive pathogens disrupt the Golgi apparatus, causing an impairment in the Paneth cell secretion of antimicrobial proteins, an alternative secretion pathway exists: it has been shown that lysozyme can be rerouted through secretory autophagy. In secretory autophagy, cargo is transported in an LC3+ vesicle and discharged at the plasma membrane, thus bypassing the ER-Golgi complex. Not all bacteria prompts secretory autophagy: commensal bacteria, for example, does not cause Golgi breakdown and therefore does not trigger the secretory autophagy of lysozyme. A dysfunction in secretory autophagy is thought to be a possible contributing factor to Crohn's disease.[19]
Phagocytic function
Paneth cells maintain the health of the intestine by acting as macrophages; it has been shown that Paneth cells clear dying cells via apoptotic cell uptake. The phagocytic function of Paneth cells was discovered using a series of experiments, one of which made use of mice that were radiated with a low dose Cesium-137 (137Cs), mimicking chemotherapy undergone by cancer patients.[20] These findings may be significant for addressing the side effects suffered by cancer patient whose intestinal health is damaged by chemotherapy: approximately 40% of all cancer therapy patients experience gastrointestinal (GI) mucositis during their treatment, with the number jumping to 80% in patients receiving abdominal or pelvic irradiation.[21]
Epithelium maintenance
Paneth cells participate in the Wnt signaling pathway and Notch signalling pathway, which regulate proliferation of intestinal stem cells and enterocytes necessary for epithelium cell renewal. They express the canonical Wnt ligands: Wnt3a, Wnt9b, and Wnt11, which bind to Frizzled receptors on intestinal stem cells to drive β-catenin/Tcf signaling. Paneth cells are also a major source of Notch ligands DLL1 and DLL4, binding to Notch receptors Notch1 and Notch2 on intestinal stem cells and enterocyte progenitors.[8]
Recently, however, it has been discovered that the regenerative potential of intestinal epithelial cells declines over time as a result of aged Paneth cells secreting the protein Notum, which is an extracellular inhibitor of Wnt signaling. If Notum secretion is inhibited, the regenerative potential of the intestinal epithelium could increase.[22]
Zinc
It has been established that zinc is essential for the function of Paneth cells. A defect in the Zn transporter (ZnT)2 impairs Paneth cell function by causing uncoordinated granule secretion. Mice lacking the (ZnT)2 transporter not only exhibit impaired granule secretion, they also suffer from increased inflammatory response to lipopolysaccharide and are less capable of bactericidal activity.[23] Normally, zinc is stored in the secretory granules and, upon degranulation, is released in the lumen. It has been speculated that the storage of heavy metals contributes to direct antimicrobial toxicity, as Zn is released upon cholinergic PC stimulation.[24]
Zinc deficiency is also implicated in alcohol‐induced Paneth cell α‐defensin dysfunction, which contributes to alcohol-related steatohepatitis. Zinc can stabilize human α‐defensin 5 (HD5), which is responsible for microbiome homeostasis. In line with this, the administration of HD5 can effectively alter the microbiome (especially by increasing Akkermansia muciniphila), and reverse the damage inflicted on the microbiome by excessive alcohol consumption. Dietary zinc deficiency on the other hand exacerbates the deleterious effect of alcohol on the bactericidal activity of Paneth cells.[25]
Clinical significance
Abnormal Paneth cells with reduced expression or secretion of defensins HD-5 and HD-6 (in human) and antimicrobial peptides are associated with inflammatory bowel disease.[26][17] In addition to that, several of the Crohn's disease-risk alleles are associated with Paneth cell dysfunction are involved in processes such as autophagy, the unfolded protein response, and the regulation of mitochondrial function.[17]
It is believed that the dysfunction of Paneth cells compromises antimicrobial peptides leading to a microbiota composition shift, and even dysbiosis.[27] Crohn's disease patients with a higher percentage of abnormal Paneth cells showed significantly reduced bacterial diversity compared with patients with a lower percentage of abnormal Paneth cells, reflecting a reduced abundance of anti-inflammatory microbes.[28] Collectively, these findings support the theory that Paneth cell dysfunction may lead to a dysbiotic microbiota that, in turn, could predispose an individual to the development of Crohn's disease.[17] However, it is yet to be established whether Paneth cell dysfunction is the cause of dysbiosis, or its concomitant effect.[27]
Necrotizing enterocolitis
Paneth cells develop gradually during gestation and therefore preterm babies might not have them in sufficient numbers. This leaves preterm babies vulnerable to necrotizing enterocolitis. About mid-way though the development of the small intestine, cathelicidin secretion is replaced by α-defensin secretion.[29] The small intestine of the premature baby is at this transition stage when the baby is born, making preterm babies susceptible to intestinal injury and, subsequently, to necrotizing enterocolitis.[18] It should furthermore be noted that early Paneth cells do not possess fully functional, mature granules.[30]
The mechanism that links Paneth cells to necrotizing enterocolitis remains unclear, but it has been theorized that a bloom of Proteobacteria and, more specifically, Enterobacteriaceae species precedes the development of the condition.[31] When an inflammation then subsequently occurs, nitrates can be fermented by Enterobacteriaceae sp. but not by obligate anaerobes, which cannot use nitrates as a growth substrate. Thus, Proteobacteria are able to use this selective pressure to out-compete the obligate anaerobic Firmicutes and Bacteroidetes, resulting in their overgrowth and consequent dysbiosis.[18]
The process is thought to begin when the premature infant is exposed to foreign antigens via formula feeding. Inflammatory cytokines are subsequently released, creating a more aerobic state leading to a competitive advantage for Proteobacteria. As the microbiome becomes more dysbiotic, anti-inflammatory mechanisms weaken, which contributes to a cycle of increasing intestinal inflammation. The inflammation leads to a further loss in Paneth cells density and function, resulting in the impairment of AMP secretion and the destruction of the stem cell niche.[18]
Non-alcoholic fatty liver disease
Whereas the role of Paneth cells in irritable bowel syndrome and Crohn's disease has received ample attention,[32][17] relatively little is known about the effect Panth cell impairment has on the pathogenesis of non-alcoholic steato-hepatitis or non-alcoholic fatty liver disease.
Murine models indicate that obesity may decrease the secretion of α-defensin from Paneth cells, leading to dysbiosis.[33] and at least one murine model suggests that when α-defensin levels in the intestinal lumen are restored by intravenous administration of R-Spondin1 to induce Paneth cell regeneration, liver fibrosis is ameliorated as a result of the dysbiosis resolving. It is hypothesized that selective microbicidal activities, as well as increasing Muribaculaceae and decreasing Harryflintia, contribute to amelioration in fibrogenesis.[34]
One study described the injection of dithizone, which can disrupt cell granulates, into mice that were fed a high-fat diet in order to identify Paneth-cell-oriented microbial alterations. The application of dithizone improved high-fat diet glucose intolerance and insulin resistance and was associated with an alleviation in the severity of liver steatosis in HFD mice, possibly through gut microbiome modulation involving the increase in Bacteroides. It has therefore been suggested that microbiome-targeted therapies may have a role in the treatment of non-alcoholic fatty liver disease.[35]
Further research is needed to elucidate the connection between Paneth cells and the gut-liver-axis.
See also
- List of human cell types derived from the germ layers
- List of distinct cell types in the adult human body
References
- ↑ "Go to Cells-Talk.com". http://www.copewithcytokines.org/cope.cgi?key=Paneth%20cells.
- ↑ 2.0 2.1 "Paneth's cell | anatomy". http://www.britannica.com/EBchecked/topic/441200/Paneths-cell.
- ↑ "Paneth cells as the cornerstones of intestinal and organismal health: a primer". EMBO Molecular Medicine 15 (2). December 2022. doi:10.15252/emmm.202216427. PMID 36573340.
- ↑ 4.0 4.1 4.2 Nutrition in Pediatrics: Basic Science, Clinical Applications. B.C. Decker. 2008. p. 244. ISBN 978-1-55009-361-2. https://books.google.com/books?id=wSTISCdSIosC&q=Paneth+cell&pg=PA244. Retrieved 2016-09-17.
- ↑ 5.0 5.1 5.2 "Paneth cells: their role in innate immunity and inflammatory disease". Gut 54 (12): 1802–1809. December 2005. doi:10.1136/gut.2005.068601. PMID 16284290.
- ↑ "Cellular inheritance of a Cre-activated reporter gene to determine Paneth cell longevity in the murine small intestine". Developmental Dynamics 233 (4): 1332–1336. August 2005. doi:10.1002/dvdy.20446. PMID 15937933.
- ↑ 7.0 7.1 "Paneth cells: maestros of the small intestinal crypts". Annual Review of Physiology 75: 289–311. 2013. doi:10.1146/annurev-physiol-030212-183744. PMID 23398152.
- ↑ 8.0 8.1 "Secretory Sorcery: Paneth Cell Control of Intestinal Repair and Homeostasis". Cellular and Molecular Gastroenterology and Hepatology 12 (4): 1239–1250. 2021. doi:10.1016/j.jcmgh.2021.06.006. PMID 34153524.
- ↑ Matsubara F (1977). "Morphological study of the Paneth cell. Paneth cells in intestinal metaplasia of the stomach and duodenum of man.". Acta Pathol Jpn 27 (5): 677–95. doi:10.1111/j.1440-1827.1977.tb00185.x. PMID 930588. https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=930588.
- ↑ 10.0 10.1 10.2 "Paneth cells--guardians of the gut cell hatchery". Nature Immunology 1 (2): 99–100. August 2000. doi:10.1038/77884. PMID 11248797.
- ↑ "Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface". Proceedings of the National Academy of Sciences of the United States of America 105 (52): 20858–20863. December 2008. doi:10.1073/pnas.0808723105. PMID 19075245. Bibcode: 2008PNAS..10520858V.
- ↑ "Degranulation of paneth cells via toll-like receptor 9". The American Journal of Pathology 165 (2): 373–381. August 2004. doi:10.1016/S0002-9440(10)63304-4. PMID 15277213.
- ↑ "Regulation of intestinal alpha-defensin activation by the metalloproteinase matrilysin in innate host defense". Science 286 (5437): 113–117. October 1999. doi:10.1126/science.286.5437.113. PMID 10506557.
- ↑ "Secretion of microbicidal alpha-defensins by intestinal Paneth cells in response to bacteria". Nature Immunology 1 (2): 113–118. August 2000. doi:10.1038/77783. PMID 11248802.
- ↑ "Paneth cell α-defensins HD-5 and HD-6 display differential degradation into active antimicrobial fragments". Proceedings of the National Academy of Sciences of the United States of America 116 (9): 3746–3751. February 2019. doi:10.1073/pnas.1817376116. PMID 30808760. Bibcode: 2019PNAS..116.3746E.
- ↑ "Paneth cell α-defensin 6 (HD-6) is an antimicrobial peptide". Mucosal Immunology 8 (3): 661–671. May 2015. doi:10.1038/mi.2014.100. PMID 25354318.
- ↑ 17.0 17.1 17.2 17.3 17.4 "Implications of Paneth cell dysfunction on gastrointestinal health and disease". Current Opinion in Gastroenterology 38 (6): 535–540. November 2022. doi:10.1097/MOG.0000000000000887. PMID 36165037.
- ↑ 18.0 18.1 18.2 18.3 "The Paneth Cell: The Curator and Defender of the Immature Small Intestine". Frontiers in Immunology 11. 2020. doi:10.3389/fimmu.2020.00587. PMID 32308658.
- ↑ "Paneth cells secrete lysozyme via secretory autophagy during bacterial infection of the intestine". Science 357 (6355): 1047–1052. September 2017. doi:10.1126/science.aal4677. PMID 28751470. Bibcode: 2017Sci...357.1047B.
- ↑ "Efferocytosis by Paneth cells within the intestine". Current Biology 31 (11): 2469–2476.e5. June 2021. doi:10.1016/j.cub.2021.03.055. PMID 33852873. Bibcode: 2021CBio...31E2469S.
- ↑ "Radiation enteropathy--pathogenesis, treatment and prevention". Nature Reviews. Gastroenterology & Hepatology 11 (8): 470–479. August 2014. doi:10.1038/nrgastro.2014.46. PMID 24686268.
- ↑ "Notum produced by Paneth cells attenuates regeneration of aged intestinal epithelium". Nature 571 (7765): 398–402. July 2019. doi:10.1038/s41586-019-1383-0. PMID 31292548.
- ↑ "ZnT2-Mediated Zinc Import Into Paneth Cell Granules Is Necessary for Coordinated Secretion and Paneth Cell Function in Mice". Cellular and Molecular Gastroenterology and Hepatology 2 (3): 369–383. May 2016. doi:10.1016/j.jcmgh.2015.12.006. PMID 28174721.
- ↑ "Paneth cells as the cornerstones of intestinal and organismal health: a primer". EMBO Molecular Medicine 15 (2). February 2023. doi:10.15252/emmm.202216427. PMID 36573340.
- ↑ "Paneth Cell Dysfunction Mediates Alcohol-related Steatohepatitis Through Promoting Bacterial Translocation in Mice: Role of Zinc Deficiency". Hepatology 71 (5): 1575–1591. May 2020. doi:10.1002/hep.30945. PMID 31520476.
- ↑ "Reduced Paneth cell alpha-defensins in ileal Crohn's disease". Proceedings of the National Academy of Sciences of the United States of America 102 (50): 18129–18134. December 2005. doi:10.1073/pnas.0505256102. PMID 16330776. Bibcode: 2005PNAS..10218129W.
- ↑ 27.0 27.1 "Dysbiosis--a consequence of Paneth cell dysfunction". Seminars in Immunology. Microbiota and the immune system, an amazing mutualism forged by co-evolution 25 (5): 334–341. November 2013. doi:10.1016/j.smim.2013.09.006. PMID 24239045.
- ↑ "Paneth cell defects in Crohn's disease patients promote dysbiosis". JCI Insight 1 (8). June 2016. doi:10.1172/jci.insight.86907. PMID 27699268.
- ↑ "Innate Immunity in the Small Intestine of the Preterm Infant". NeoReviews 12 (9): e517–e526. September 2011. doi:10.1542/neo.12-9-e517. PMID 22639551.
- ↑ "Examining the role of Paneth cells in the small intestine by lineage ablation in transgenic mice". The Journal of Biological Chemistry 272 (38): 23729–23740. September 1997. doi:10.1074/jbc.272.38.23729. PMID 9295317.
- ↑ "Necrotizing enterocolitis: a clinical review on diagnostic biomarkers and the role of the intestinal microbiota". Inflammatory Bowel Diseases 21 (2): 436–444. February 2015. doi:10.1097/MIB.0000000000000184. PMID 25268636. https://pure.amsterdamumc.nl/en/publications/fd89fd84-4b35-4076-acc9-736a73824069.
- ↑ "Paneth Cell Alterations in the Development and Phenotype of Crohn's Disease". Gastroenterology 152 (2): 322–326. February 2017. doi:10.1053/j.gastro.2016.10.003. PMID 27729212.
- ↑ "Bile acid toxicity in Paneth cells contributes to gut dysbiosis induced by high-fat feeding". JCI Insight 5 (20). October 2020. doi:10.1172/jci.insight.138881. PMID 33055426.
- ↑ "Decreased Paneth cell α-defensins promote fibrosis in a choline-deficient L-amino acid-defined high-fat diet-induced mouse model of nonalcoholic steatohepatitis via disrupting intestinal microbiota". Scientific Reports 13 (1). March 2023. doi:10.1038/s41598-023-30997-y. PMID 36894646. Bibcode: 2023NatSR..13.3953N.
- ↑ "Alleviation of Hepatic Steatosis: Dithizone-Related Gut Microbiome Restoration During Paneth Cell Dysfunction". Frontiers in Microbiology 13. 2022-02-25. doi:10.3389/fmicb.2022.813783. PMID 35283810.
Further reading
- "Defensins and host defense". Science 286 (5439): 420–421. October 1999. doi:10.1126/science.286.5439.420. PMID 10577203.
- "Paneth cells--guardians of the gut cell hatchery". Nature Immunology 1 (2): 99–100. August 2000. doi:10.1038/77884. PMID 11248797.
 |
