Biology:Macrophage
Macrophages (/ˈmækroʊfeɪdʒ/; abbreviated Mφ, MΦ or MP) are a type of white blood cell of the innate immune system that engulf and digest pathogens, such as cancer cells, microbes, cellular debris and foreign substances, which do not have proteins that are specific to healthy body cells on their surface.[1][2] This self-protection method can be contrasted with that employed by Natural Killer cells. This process of engulfment and digestion is called phagocytosis; it acts to defend the host against infection and injury.[3]
Macrophages are found in essentially all tissues,[4] where they patrol for potential pathogens by amoeboid movement. They take various forms (with various names) throughout the body (e.g., histiocytes, Kupffer cells, alveolar macrophages, microglia, and others), but all are part of the mononuclear phagocyte system. Besides phagocytosis, they play a critical role in nonspecific defense (innate immunity) and also help initiate specific defense mechanisms (adaptive immunity) by recruiting other immune cells such as lymphocytes. For example, they are important as antigen presenters to T cells. In humans, dysfunctional macrophages cause severe diseases such as chronic granulomatous disease that result in frequent infections.
Beyond increasing inflammation and stimulating the immune system, macrophages also play an important anti-inflammatory role and can decrease immune reactions through the release of cytokines. Macrophages that encourage inflammation are called M1 macrophages, whereas those that decrease inflammation and encourage tissue repair are called M2 macrophages.[5] This difference is reflected in their metabolism; M1 macrophages have the unique ability to metabolize arginine to the "killer" molecule nitric oxide, whereas M2 macrophages have the unique ability to metabolize arginine to the "repair" molecule ornithine.[6] However, this dichotomy has been recently questioned as further complexity has been discovered.[7] Macrophages are widely thought of as highly plastic and fluid cells, with a fluctuating phenotype.
Human macrophages are about 21 micrometres (0.00083 in) in diameter[8] and are produced by the differentiation of monocytes in tissues. They can be identified using flow cytometry or immunohistochemical staining by their specific expression of proteins such as CD14, CD40, CD11b, CD64, F4/80 (mice)/EMR1 (human), lysozyme M, MAC-1/MAC-3 and CD68.[9]
Macrophages were first discovered and named by Élie Metchnikoff, a Russian Empire zoologist, in 1884.[10][11]
Structure
Types
A majority of macrophages are stationed at strategic points where microbial invasion or accumulation of foreign particles is likely to occur. These cells together as a group are known as the mononuclear phagocyte system and were previously known as the reticuloendothelial system. Each type of macrophage, determined by its location, has a specific name:
| Cell Name | Anatomical Location |
| Adipose tissue macrophages | Adipose tissue (fat) |
| Monocytes | Bone marrow / blood |
| Kupffer cells | Liver |
| Sinus histiocytes | Lymph nodes |
| Alveolar macrophages (dust cells) | Pulmonary alveoli |
| Tissue macrophages (histiocytes) leading to giant cells | Connective tissue |
| Microglia | Central nervous system |
| Hofbauer cells | Placenta |
| Intraglomerular mesangial cells[12] | Kidney |
| Osteoclasts [13] | Bone |
| Dermal macrophages and Langerhans cells | Skin |
| Epithelioid cells | Granulomas |
| Red pulp macrophages (sinusoidal lining cells) | Red pulp of spleen |
| Peritoneal macrophages | Peritoneal cavity |
| Perivascular macrophages[14] | Closely associated with blood vessels |
Investigations concerning Kupffer cells are hampered because in humans, Kupffer cells are only accessible for immunohistochemical analysis from biopsies or autopsies. From rats and mice, they are difficult to isolate, and after purification, only approximately 5 million cells can be obtained from one mouse.
Macrophages can express paracrine functions within organs that are specific to the function of that organ. In the testis, for example, macrophages have been shown to be able to interact with Leydig cells by secreting 25-hydroxycholesterol, an oxysterol that can be converted to testosterone by neighbouring Leydig cells.[15] Also, testicular macrophages may participate in creating an immune privileged environment in the testis, and in mediating infertility during inflammation of the testis.
Cardiac resident macrophages participate in electrical conduction via gap junction communication with cardiac myocytes.[16]
Macrophages can be classified on basis of the fundamental function and activation. According to this grouping, there are classically activated (M1) macrophages, wound-healing macrophages (also known as alternatively-activated (M2) macrophages), and regulatory macrophages (Mregs).[17]
Development
Macrophages that reside in adult healthy tissues either derive from circulating monocytes or are established before birth and then maintained during adult life independently of monocytes.[18][19] By contrast, most of the macrophages that accumulate at diseased sites typically derive from circulating monocytes.[20] Leukocyte extravasation describes monocyte entry into damaged tissue through the endothelium of blood vessels as they become macrophages. Monocytes are attracted to a damaged site by chemical substances through chemotaxis, triggered by a range of stimuli including damaged cells, pathogens and cytokines released by macrophages already at the site. At some sites such as the testis, macrophages have been shown to populate the organ through proliferation.[21] Unlike short-lived neutrophils, macrophages survive longer in the body, up to several months.
Function
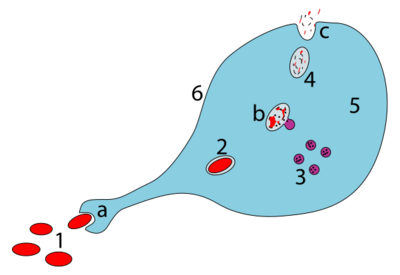
a. Ingestion through phagocytosis, a phagosome is formed
b. The fusion of lysosomes with the phagosome creates a phagolysosome; the pathogen is broken down by enzymes
c. Waste material is expelled or assimilated (the latter not pictured)
Parts:
1. Pathogens
2. Phagosome
3. Lysosomes
4. Waste material
5. Cytoplasm
6. Cell membrane
Phagocytosis
Macrophages are professional phagocytes and are highly specialized in removal of dying or dead cells and cellular debris. This role is important in chronic inflammation, as the early stages of inflammation are dominated by neutrophils, which expend themselves and are ingested by macrophages.[22] Macrophages normally present themselves at the wound site within 2 days following the injury.
The neutrophils are at first attracted to a site, where they perform their function and die, before they or their neutrophil extracellular traps are phagocytized by the macrophages.[22][23] The first wave of neutrophils acts for approximately 2 days at the site and signals to attract macrophages. These macrophages will then ingest the aged neutrophils.[22]
The removal of dying cells is, to a greater extent, handled by fixed macrophages, which will stay at strategic locations such as the lungs, liver, neural tissue, bone, spleen and connective tissue, ingesting foreign materials such as pathogens and recruiting additional macrophages if needed.[24] The phagocytosis and clearance of apoptotic remains is called efferocytosis and is also carried out by other cell types, not all of which are professional phagocytes.
When a macrophage ingests a pathogen, the pathogen becomes trapped in a phagosome, which then fuses with a lysosome. Within the phagolysosome, enzymes and toxic peroxides digest the pathogen. However, some bacteria (such as Mycobacterium tuberculosis) have become resistant to these methods of digestion. Typhoidal Salmonellae induce their own phagocytosis by host macrophages in vivo and inhibit digestion by lysosomal action, thereby using macrophages for their own replication and causing macrophage apoptosis.[25] Macrophages are capable of engulfing and digesting many bacteria during their life. They can die eventually due to factors including pathogenic cytotoxicity, oxidative stress, and phagocytosis-induced apoptosis.[26] Phagocytosis-induced apoptosis results from the powerful apoptotic stimulus of consuming bacteria and is observed in (at least) macrophages and neutrophils.
Role in innate immune response
When a pathogen invades, tissue resident macrophages are among the first cells to respond.[27] Two of the main roles of the tissue resident macrophages are to phagocytose incoming antigen and to secrete proinflammatory cytokines that induce inflammation and recruit other immune cells to the site.[28]
Phagocytosis of pathogens

Macrophages can internalize antigens through receptor-mediated phagocytosis.[29] Macrophages have a wide variety of pattern recognition receptors (PRRs) that can recognize microbe-associated molecular patterns (MAMPs) from pathogens. Many PRRs, such as toll-like receptors (TLRs), scavenger receptors (SRs), C-type lectin receptors, among others, recognize pathogens for phagocytosis.[29] Macrophages can also recognize pathogens for phagocytosis indirectly through opsonins, which are molecules that attach to pathogens and mark them for phagocytosis.[30] Opsonins can cause a stronger adhesion between the macrophage and pathogen during phagocytosis, hence opsonins tend to enhance macrophages' phagocytic activity.[31] Both complement proteins and antibodies can bind to antigens and opsonize them. Macrophages have complement receptor 1 (CR1) and 3 (CR3) that recognize pathogen-bound complement proteins C3b and iC3b, respectively, as well as fragment crystallizable γ receptors (FcγRs) that recognize the fragment crystallizable (Fc) region of antigen-bound immunoglobulin G (IgG) antibodies.[30][32] When phagocytosing and digesting pathogens, macrophages go through a respiratory burst where more oxygen is consumed to supply the energy required for producing reactive oxygen species (ROS) and other antimicrobial molecules that digest the consumed pathogens.[28][33]
Chemical secretion
Recognition of MAMPs by PRRs can activate tissue resident macrophages to secrete proinflammatory cytokines that recruit other immune cells. Among the PRRs, TLRs play a major role in signal transduction leading to cytokine production.[29] The binding of MAMPs to TLR triggers a series of downstream events that eventually activates transcription factor NF-κB and results in transcription of the genes for several proinflammatory cytokines, including IL-1β, IL-6, TNF-α, IL-12B, and type I interferons such as IFN-α and IFN-β.[34] Systemically, IL-1β, IL-6, and TNF-α induce fever and initiate the acute phase response in which the liver secretes acute phase proteins.[27][28][35] Locally, IL-1β and TNF-α cause vasodilation, where the gaps between blood vessel epithelial cells widen, and upregulation of cell surface adhesion molecules on epithelial cells to induce leukocyte extravasation.[27][28] Additionally, activated macrophages have been found to have delayed synthesis of prostaglandins (PGs) which are important mediators of inflammation and pain. Among the PGs, anti-inflammatory PGE2 and pro-inflammatory PGD2 increase the most after activation, with PGE2 increasing expression of IL-10 and inhibiting production of TNFs via the COX-2 pathway.[36][37]
Neutrophils are among the first immune cells recruited by macrophages to exit the blood via extravasation and arrive at the infection site.[35] Macrophages secrete many chemokines such as CXCL1, CXCL2, and CXCL8 (IL-8) that attract neutrophils to the site of infection.[27][35] After neutrophils have finished phagocytosing and clearing the antigen at the end of the immune response, they undergo apoptosis, and macrophages are recruited from blood monocytes to help clear apoptotic debris.[38]
Macrophages also recruit other immune cells such as monocytes, dendritic cells, natural killer cells, basophils, eosinophils, and T cells through chemokines such as CCL2, CCL4, CCL5, CXCL8, CXCL9, CXCL10, and CXCL11.[27][35] Along with dendritic cells, macrophages help activate natural killer (NK) cells through secretion of type I interferons (IFN-α and IFN-β) and IL-12. IL-12 acts with IL-18 to stimulate the production of proinflammatory cytokine interferon gamma (IFN-γ) by NK cells, which serves as an important source of IFN-γ before the adaptive immune system is activated.[35][39] IFN-γ enhances the innate immune response by inducing a more aggressive phenotype in macrophages, allowing macrophages to more efficiently kill pathogens.[35]
Some of the T cell chemoattractants secreted by macrophages include CCL5, CXCL9, CXCL10, and CXCL11.[27]
Role in adaptive immunity
Interactions with CD4+ T Helper Cells
Macrophages are professional antigen presenting cells (APC), meaning they can present peptides from phagocytosed antigens on major histocompatibility complex (MHC) II molecules on their cell surface for T helper cells.[41] Macrophages are not primary activators of naïve T helper cells that have never been previously activated since tissue resident macrophages do not travel to the lymph nodes where naïve T helper cells reside.[42][43] Although macrophages are also found in secondary lymphoid organs like the lymph nodes, they do not reside in T cell zones and are not effective at activating naïve T helper cells.[42] The macrophages in lymphoid tissues are more involved in ingesting antigens and preventing them from entering the blood, as well as taking up debris from apoptotic lymphocytes.[42][44] Therefore, macrophages interact mostly with previously activated T helper cells that have left the lymph node and arrived at the site of infection or with tissue resident memory T cells.[43]
Macrophages supply both signals required for T helper cell activation: 1) Macrophages present antigen peptide-bound MHC class II molecule to be recognized by the corresponding T cell receptor (TCR), and 2) recognition of pathogens by PRRs induce macrophages to upregulate the co-stimulatory molecules CD80 and CD86 (also known as B7) that binds to CD28 on T helper cells to supply the co-stimulatory signal.[35][41] These interactions allow T helper cells to achieve full effector function and provide T helper cells with continued survival and differentiation signals preventing them from undergoing apoptosis due to lack of TCR signaling.[41] For example, IL-2 signaling in T cells upregulates the expression of anti-apoptotic protein Bcl-2, but T cell production of IL-2 and the high-affinity IL-2 receptor IL-2RA both require continued signal from TCR recognition of MHC-bound antigen.[35][45]
Activation
Macrophages can achieve different activation phenotypes through interactions with different subsets of T helper cells, such as TH1 and TH2.[17] Although there is a broad spectrum of macrophage activation phenotypes, there are two major phenotypes that are commonly acknowledged.[17] They are the classically activated macrophages, or M1 macrophages, and the alternatively activated macrophages, or M2 macrophages. M1 macrophages are proinflammatory, while M2 macrophages are mostly anti-inflammatory.[17]
Classical
TH1 cells play an important role in classical macrophage activation as part of type 1 immune response against intracellular pathogens (such as intracellular bacteria) that can survive and replicate inside host cells, especially those pathogens that replicate even after being phagocytosed by macrophages.[46] After the TCR of TH1 cells recognize specific antigen peptide-bound MHC class II molecules on macrophages, TH1 cells 1) secrete IFN-γ and 2) upregulate the expression of CD40 ligand (CD40L), which binds to CD40 on macrophages.[47][35] These 2 signals activate the macrophages and enhance their ability to kill intracellular pathogens through increased production of antimicrobial molecules such as nitric oxide (NO) and superoxide (O2-).[27][35] This enhancement of macrophages' antimicrobial ability by TH1 cells is known as classical macrophage activation, and the activated macrophages are known as classically activated macrophages, or M1 macrophages. The M1 macrophages in turn upregulate B7 molecules and antigen presentation through MHC class II molecules to provide signals that sustain T cell help.[47] The activation of TH1 and M1 macrophage is a positive feedback loop, with IFN-γ from TH1 cells upregulating CD40 expression on macrophages; the interaction between CD40 on the macrophages and CD40L on T cells activate macrophages to secrete IL-12; and IL-12 promotes more IFN-γ secretion from TH1 cells.[35][47] The initial contact between macrophage antigen-bound MHC II and TCR serves as the contact point between the two cells where most of the IFN-γ secretion and CD-40L on T cells concentrate to, so only macrophages directly interacting with TH1 cells are likely to be activated.[35]
In addition to activating M1 macrophages, TH1 cells express Fas ligand (FasL) and lymphotoxin beta (LT-β) to help kill chronically infected macrophages that can no longer kill pathogens.[35] The killing of chronically infected macrophages release pathogens to the extracellular space that can then be killed by other activated macrophages.[35] TH1 cells also help recruit more monocytes, the precursor to macrophages, to the infection site. TH1 secretion TNF-α and LT-α to make blood vessels easier for monocytes to bind to and exit.[35] TH1 secretion of CCL2 as a chemoattractant for monocytes. IL-3 and GM-CSF released by TH1 cells stimulate more monocyte production in the bone marrow.[35]
When intracellular pathogens cannot be eliminated, such as in the case of Mycobacterium tuberculosis, the pathogen is contained through the formation of granuloma, an aggregation of infected macrophages surrounded by activated T cells.[48] The macrophages bordering the activated lymphocytes often fuse to form multinucleated giant cells that appear to have increased antimicrobial ability due to their proximity to TH1 cells, but over time, the cells in the center start to die and form necrotic tissue.[43][48]
Alternative
TH2 cells play an important role in alternative macrophage activation as part of type 2 immune response against large extracellular pathogens like helminths.[35][49] TH2 cells secrete IL-4 and IL-13, which activate macrophages to become M2 macrophages, also known as alternatively activated macrophages.[49][50] M2 macrophages express arginase-1, an enzyme that converts arginine to ornithine and urea.[49] Ornithine help increase smooth muscle contraction to expel the worm and also participates in tissue and wound repair. Ornithine can be further metabolized to proline, which is essential for synthesizing collagen.[49] M2 macrophages can also decrease inflammation by producing IL-1 receptor antagonist (IL-1RA) and IL-1 receptors that do not lead to downstream inflammatory signaling (IL-1RII).[35][51]
Interactions with CD8+ cytotoxic t cells
Another part of the adaptive immunity activation involves stimulating CD8+ via cross presentation of antigens peptides on MHC class I molecules. Studies have shown that proinflammatory macrophages are capable of cross presentation of antigens on MHC class I molecules, but whether macrophage cross-presentation plays a role in naïve or memory CD8+ T cell activation is still unclear.[28][52][44]
Interactions with B cells
Macrophages have been shown to secrete cytokines BAFF and APRIL, which are important for plasma cell isotype switching. APRIL and IL-6 secreted by macrophage precursors in the bone marrow help maintain survival of plasma cells homed to the bone marrow.[53]
Subtypes
There are several activated forms of macrophages.[17] In spite of a spectrum of ways to activate macrophages, there are two main groups designated M1 and M2. M1 macrophages: as mentioned earlier (previously referred to as classically activated macrophages),[55] M1 "killer" macrophages are activated by LPS and IFN-gamma, and secrete high levels of IL-12 and low levels of IL-10. M1 macrophages have pro-inflammatory, bactericidal, and phagocytic functions.[56] In contrast, the M2 "repair" designation (also referred to as alternatively activated macrophages) broadly refers to macrophages that function in constructive processes like wound healing and tissue repair, and those that turn off damaging immune system activation by producing anti-inflammatory cytokines like IL-10. M2 is the phenotype of resident tissue macrophages, and can be further elevated by IL-4. M2 macrophages produce high levels of IL-10, TGF-beta and low levels of IL-12. Tumor-associated macrophages are mainly of the M2 phenotype, and seem to actively promote tumor growth.[57]
Macrophages exist in a variety of phenotypes which are determined by the role they play in wound maturation. Phenotypes can be predominantly separated into two major categories; M1 and M2. M1 macrophages are the dominating phenotype observed in the early stages of inflammation and are activated by four key mediators: interferon-γ (IFN-γ), tumor necrosis factor (TNF), and damage associated molecular patterns (DAMPs). These mediator molecules create a pro-inflammatory response that in return produce pro-inflammatory cytokines like Interleukin-6 and TNF. Unlike M1 macrophages, M2 macrophages secrete an anti-inflammatory response via the addition of Interleukin-4 or Interleukin-13. They also play a role in wound healing and are needed for revascularization and reepithelialization. M2 macrophages are divided into four major types based on their roles: M2a, M2b, M2c, and M2d. How M2 phenotypes are determined is still up for discussion but studies have shown that their environment allows them to adjust to whichever phenotype is most appropriate to efficiently heal the wound.[56]
M2 macrophages are needed for vascular stability. They produce vascular endothelial growth factor-A and TGF-β1.[56] There is a phenotype shift from M1 to M2 macrophages in acute wounds, however this shift is impaired for chronic wounds. This dysregulation results in insufficient M2 macrophages and its corresponding growth factors that aid in wound repair. With a lack of these growth factors/anti-inflammatory cytokines and an overabundance of pro-inflammatory cytokines from M1 macrophages chronic wounds are unable to heal in a timely manner. Normally, after neutrophils eat debris/pathogens they perform apoptosis and are removed. At this point, inflammation is not needed and M1 undergoes a switch to M2 (anti-inflammatory). However, dysregulation occurs as the M1 macrophages are unable/do not phagocytose neutrophils that have undergone apoptosis leading to increased macrophage migration and inflammation.[56]
Both M1 and M2 macrophages play a role in promotion of atherosclerosis. M1 macrophages promote atherosclerosis by inflammation. M2 macrophages can remove cholesterol from blood vessels, but when the cholesterol is oxidized, the M2 macrophages become apoptotic foam cells contributing to the atheromatous plaque of atherosclerosis.[58][59]
Role in muscle regeneration
The first step to understanding the importance of macrophages in muscle repair, growth, and regeneration is that there are two "waves" of macrophages with the onset of damageable muscle use– subpopulations that do and do not directly have an influence on repairing muscle. The initial wave is a phagocytic population that comes along during periods of increased muscle use that are sufficient to cause muscle membrane lysis and membrane inflammation, which can enter and degrade the contents of injured muscle fibers.[60][61][62] These early-invading, phagocytic macrophages reach their highest concentration about 24 hours following the onset of some form of muscle cell injury or reloading.[63] Their concentration rapidly declines after 48 hours.[61] The second group is the non-phagocytic types that are distributed near regenerative fibers. These peak between two and four days and remain elevated for several days while muscle tissue is rebuilding.[61] The first subpopulation has no direct benefit to repairing muscle, while the second non-phagocytic group does.
It is thought that macrophages release soluble substances that influence the proliferation, differentiation, growth, repair, and regeneration of muscle, but at this time the factor that is produced to mediate these effects is unknown.[63] It is known that macrophages' involvement in promoting tissue repair is not muscle specific; they accumulate in numerous tissues during the healing process phase following injury.[64]
Role in wound healing
Macrophages are essential for wound healing.[65] They replace polymorphonuclear neutrophils as the predominant cells in the wound by day two after injury.[66] Attracted to the wound site by growth factors released by platelets and other cells, monocytes from the bloodstream enter the area through blood vessel walls.[67] Numbers of monocytes in the wound peak one to one and a half days after the injury occurs. Once they are in the wound site, monocytes mature into macrophages. The spleen contains half the body's monocytes in reserve ready to be deployed to injured tissue.[68][69]
The macrophage's main role is to phagocytize bacteria and damaged tissue,[65] and they also debride damaged tissue by releasing proteases.[70] Macrophages also secrete a number of factors such as growth factors and other cytokines, especially during the third and fourth post-wound days. These factors attract cells involved in the proliferation stage of healing to the area.[71] Macrophages may also restrain the contraction phase.[72] Macrophages are stimulated by the low oxygen content of their surroundings to produce factors that induce and speed angiogenesis[73] and they also stimulate cells that re-epithelialize the wound, create granulation tissue, and lay down a new extracellular matrix.[74] By secreting these factors, macrophages contribute to pushing the wound healing process into the next phase.
Role in limb regeneration
Scientists have elucidated that as well as eating up material debris, macrophages are involved in the typical limb regeneration in the salamander.[75][76] They found that removing the macrophages from a salamander resulted in failure of limb regeneration and a scarring response.[75][76]
Role in iron homeostasis
The iron that is released from the haemoglobin is either stored internally in ferritin or is released into the circulation via ferroportin. In cases where systemic iron levels are raised, or where inflammation is present, raised levels of hepcidin act on macrophage ferroportin channels, leading to iron remaining within the macrophages.[77]
Role in pigment retainment
Melanophages are a subset of tissue-resident macrophages able to absorb pigment, either native to the organism or exogenous (such as tattoos), from extracellular space. In contrast to dendritic juncional melanocytes, which synthesize melanosomes and contain various stages of their development, the melanophages only accumulate phagocytosed melanin in lysosome-like phagosomes.[78][79] This occurs repeatedly as the pigment from dead dermal macrophages is phagocytosed by their successors, preserving the tattoo in the same place.[80]
Role in tissue homeostasis
Every tissue harbors its own specialized population of resident macrophages, which entertain reciprocal interconnections with the stroma and functional tissue.[81][82] These resident macrophages are sessile (non-migratory), provide essential growth factors to support the physiological function of the tissue (e.g. macrophage-neuronal crosstalk in the guts),[83] and can actively protect the tissue from inflammatory damage.[84]
Nerve-associated macrophages
Nerve-associated macrophages or NAMs are those tissue-resident macrophages that are associated with nerves. Some of them are known to have an elongated morphology of up to 200μm [85]
Clinical significance
Due to their role in phagocytosis, macrophages are involved in many diseases of the immune system. For example, they participate in the formation of granulomas, inflammatory lesions that may be caused by a large number of diseases. Some disorders, mostly rare, of ineffective phagocytosis and macrophage function have been described, for example.[86]
As a host for intracellular pathogens
In order to minimize the possibility of becoming the host of an intracellular bacteria, macrophages have evolved defense mechanisms such as induction of nitric oxide and reactive oxygen intermediates,[87] which are toxic to microbes. Macrophages have also evolved the ability to restrict the microbe's nutrient supply and induce autophagy.[88]
Tuberculosis
Once engulfed by a macrophage, the causative agent of tuberculosis, Mycobacterium tuberculosis,[89] avoids cellular defenses and uses the cell to replicate. Recent evidence suggests that in response to the pulmonary infection of Mycobacterium tuberculosis, the peripheral macrophages matures into M1 phenotype. Macrophage M1 phenotype is characterized by increased secretion of pro-inflammatory cytokines (IL-1β, TNF-α, and IL-6) and increased glycolytic activities essential for clearance of infection.[1]
Leishmaniasis
Chikungunya
Infection of macrophages in joints is associated with local inflammation during and after the acute phase of Chikungunya (caused by CHIKV or Chikungunya virus).[90]
Others
Heart disease
Macrophages are the predominant cells involved in creating the progressive plaque lesions of atherosclerosis.[91]
Focal recruitment of macrophages occurs after the onset of acute myocardial infarction. These macrophages function to remove debris, apoptotic cells and to prepare for tissue regeneration.[92] Macrophages protect against ischemia-induced ventricular tachycardia in hypokalemic mice.[93]
HIV infection
Macrophages also play a role in human immunodeficiency virus (HIV) infection. Like T cells, macrophages can be infected with HIV, and even become a reservoir of ongoing virus replication throughout the body. HIV can enter the macrophage through binding of gp120 to CD4 and second membrane receptor, CCR5 (a chemokine receptor). Both circulating monocytes and macrophages serve as a reservoir for the virus.[94] Macrophages are better able to resist infection by HIV-1 than CD4+ T cells, although susceptibility to HIV infection differs among macrophage subtypes.[95]
Cancer
Macrophages can contribute to tumor growth and progression by promoting tumor cell proliferation and invasion, fostering tumor angiogenesis and suppressing antitumor immune cells.[96][97] Inflammatory compounds, such as tumor necrosis factor (TNF)-alpha released by the macrophages activate the gene switch nuclear factor-kappa B. NF-κB then enters the nucleus of a tumor cell and turns on production of proteins that stop apoptosis and promote cell proliferation and inflammation.[98] Moreover, macrophages serve as a source for many pro-angiogenic factors including vascular endothelial factor (VEGF), tumor necrosis factor-alpha (TNF-alpha), macrophage colony-stimulating factor (M-CSF/CSF1) and IL-1 and IL-6,[99] contributing further to the tumor growth.
Macrophages have been shown to infiltrate a number of tumors. Their number correlates with poor prognosis in certain cancers, including cancers of breast, cervix, bladder, brain and prostate.[100][101] Some tumors can also produce factors, including M-CSF/CSF1, MCP-1/CCL2 and Angiotensin II, that trigger the amplification and mobilization of macrophages in tumors.[102][103][104] Additionally, subcapsular sinus macrophages in tumor-draining lymph nodes can suppress cancer progression by containing the spread of tumor-derived materials.[105]
Cancer therapy
Experimental studies indicate that macrophages can affect all therapeutic modalities, including surgery, chemotherapy, radiotherapy, immunotherapy and targeted therapy.[97][106][107] Macrophages can influence treatment outcomes both positively and negatively. Macrophages can be protective in different ways: they can remove dead tumor cells (in a process called phagocytosis) following treatments that kill these cells; they can serve as drug depots for some anticancer drugs;[108] they can also be activated by some therapies to promote antitumor immunity.[109] Macrophages can also be deleterious in several ways: for example they can suppress various chemotherapies,[110][111] radiotherapies[112][113] and immunotherapies.[114][115] Because macrophages can regulate tumor progression, therapeutic strategies to reduce the number of these cells, or to manipulate their phenotypes, are currently being tested in cancer patients.[116][117] However, macrophages are also involved in antibody mediated cytotoxicity (ADCC) and this mechanism has been proposed to be important for certain cancer immunotherapy antibodies.[118] Similarly, studies identified macrophages genetically engineered to express chimeric antigen receptors as promising therapeutic approach to lowering tumor burden.[119]
Obesity
It has been observed that increased number of pro-inflammatory macrophages within obese adipose tissue contributes to obesity complications including insulin resistance and diabetes type 2.[120]
The modulation of the inflammatory state of adipose tissue macrophages has therefore been considered a possible therapeutic target to treat obesity-related diseases.[121] Although adipose tissue macrophages are subject to anti-inflammatory homeostatic control by sympathetic innervation, experiments using ADRB2 gene knockout mice indicate that this effect is indirectly exerted through the modulation of adipocyte function, and not through direct Beta-2 adrenergic receptor activation, suggesting that adrenergic stimulation of macrophages may be insufficient to impact adipose tissue inflammation or function in obesity.[122]
Within the fat (adipose) tissue of CCR2 deficient mice, there is an increased number of eosinophils, greater alternative macrophage activation, and a propensity towards type 2 cytokine expression. Furthermore, this effect was exaggerated when the mice became obese from a high fat diet.[123] This is partially caused by a phenotype switch of macrophages induced by necrosis of fat cells (adipocytes). In an obese individual some adipocytes burst and undergo necrotic death, which causes the residential M2 macrophages to switch to M1 phenotype. This is one of the causes of a low-grade systemic chronic inflammatory state associated with obesity.[124][125]
Intestinal macrophages
Though very similar in structure to tissue macrophages, intestinal macrophages have evolved specific characteristics and functions given their natural environment, which is in the digestive tract. Macrophages and intestinal macrophages have high plasticity causing their phenotype to be altered by their environments.[126] Like macrophages, intestinal macrophages are differentiated monocytes, though intestinal macrophages have to coexist with the microbiome in the intestines. This is a challenge considering the bacteria found in the gut are not recognized as "self" and could be potential targets for phagocytosis by the macrophage.[127]
To prevent the destruction of the gut bacteria, intestinal macrophages have developed key differences compared to other macrophages. Primarily, intestinal macrophages do not induce inflammatory responses. Whereas tissue macrophages release various inflammatory cytokines, such as IL-1, IL-6 and TNF-α, intestinal macrophages do not produce or secrete inflammatory cytokines. This change is directly caused by the intestinal macrophages environment. Surrounding intestinal epithelial cells release TGF-β, which induces the change from proinflammatory macrophage to noninflammatory macrophage.[127]
Even though the inflammatory response is downregulated in intestinal macrophages, phagocytosis is still carried out. There is no drop off in phagocytosis efficiency as intestinal macrophages are able to effectively phagocytize the bacteria,S. typhimurium and E. coli, but intestinal macrophages still do not release cytokines, even after phagocytosis. Also, intestinal macrophages do not express lipopolysaccharide (LPS), IgA, or IgG receptors.[128] The lack of LPS receptors is important for the gut as the intestinal macrophages do not detect the microbe-associated molecular patterns (MAMPS/PAMPS) of the intestinal microbiome. Nor do they express IL-2 and IL-3 growth factor receptors.[127]
Role in disease
Intestinal macrophages have been shown to play a role in inflammatory bowel disease (IBD), such as Crohn's disease (CD) and ulcerative colitis (UC). In a healthy gut, intestinal macrophages limit the inflammatory response in the gut, but in a disease-state, intestinal macrophage numbers and diversity are altered. This leads to inflammation of the gut and disease symptoms of IBD. Intestinal macrophages are critical in maintaining gut homeostasis. The presence of inflammation or pathogen alters this homeostasis, and concurrently alters the intestinal macrophages.[129] There has yet to be a determined mechanism for the alteration of the intestinal macrophages by recruitment of new monocytes or changes in the already present intestinal macrophages.[128]
Additionally, a new study reveals macrophages limit iron access to bacteria by releasing extracellular vesicles, improving sepsis outcomes.[130]
Media
-
An active J774 macrophage is seen taking up four conidia in a co-operative manner. The J774 cells were treated with 5ng/ml interferon-γ one night before filming with conidia. Observations were made every 30s over a 2.5hr period.
-
Two highly active alveolar macrophages can be seen ingesting conidia. Time lapse is 30s per frame over 2.5hr.
History
Macrophages were first discovered late in the 19th century by zoologist Élie Metchnikoff.[131] Metchnikoff revolutionized the branch of macrophages by combining philosophical insights and the evolutionary study of life.[132] Later on, Van Furth during the 1960s proposed the idea that circulating blood monocytes in adults allowed for the origin of all tissue macrophages.[133] In recent years, publishing regarding macrophages has led people to believe that multiple resident tissue macrophages are independent of the blood monocytes as it is formed during the embryonic stage of development.[134] Within the 21st century, all the ideas concerning the origin of macrophages (present in tissues) were compiled together to suggest that physiologically complex organisms, from macrophages independently by mechanisms that don't have to depend on the blood monocytes.[135]
See also
- Bacteriophage
- Dendritic cell
- Histiocyte
- List of distinct cell types in the adult human body
References
- ↑ 1.0 1.1 "NIX-mediated mitophagy regulate metabolic reprogramming in phagocytic cells during mycobacterial infection". Tuberculosis 126 (January). January 2021. doi:10.1016/j.tube.2020.102046. PMID 33421909.
- ↑ "Regenerative Medicine Partnership in Education". https://www.sepa.duq.edu/regmed/immune/macrophages.html.
- ↑ "Imaging Cardiovascular and Lung Macrophages With the Positron Emission Tomography Sensor 64Cu-Macrin in Mice, Rabbits, and Pigs". Circulation: Cardiovascular Imaging 13 (10). October 2020. doi:10.1161/CIRCIMAGING.120.010586. PMID 33076700.
- ↑ "Macrophages in the embryo and beyond: much more than just giant phagocytes". Genesis 46 (9): 447–462. September 2008. doi:10.1002/dvg.20417. PMID 18781633. "Macrophages are present essentially in all tissues, beginning with embryonic development and, in addition to their role in host defense and in the clearance of apoptotic cells, are being increasingly recognized for their trophic function and role in regeneration.".
- ↑ "M1 and M2 Macrophages: Oracles of Health and Disease". Critical Reviews in Immunology 32 (6): 463–488. 2012. doi:10.1615/CritRevImmunol.v32.i6.10. PMID 23428224.
- ↑ Janeway's immunobiology. Garland Science, New York. 2006. pp. 464, 904. ISBN 978-0-8153-4551-0.
- ↑ "A polarizing question: do M1 and M2 microglia exist?". Nature Neuroscience 19 (8): 987–991. July 2016. doi:10.1038/nn.4338. PMID 27459405.
- ↑ "Cell size of alveolar macrophages: an interspecies comparison". Environmental Health Perspectives 105 (Suppl 5): 1261–1263. September 1997. doi:10.2307/3433544. PMID 9400735.
- ↑ "Expression of macrophage-selective markers in human and rodent adipocytes". FEBS Letters 579 (25): 5631–5634. October 2005. doi:10.1016/j.febslet.2005.09.032. PMID 16213494. Bibcode: 2005FEBSL.579.5631K.
- ↑ Ilya Mechnikov: His Life and Work. Honolulu, Hawaii: University Press of the Pacific. 2001. pp. 78, 210. ISBN 978-0-89875-622-7.
- ↑ "Macrophage plasticity, polarization, and function in health and disease". Journal of Cellular Physiology 233 (9): 6425–6440. September 2018. doi:10.1002/jcp.26429. PMID 29319160.
- ↑ Principles of Renal Physiology, 5th edition. Springer. p. 37.
- ↑ "Topography-mediated immunomodulation in osseointegration; Ally or Enemy". Biomaterials 291. December 2022. doi:10.1016/j.biomaterials.2022.121903. PMID 36410109.
- ↑ "Deciphering the heterogeneity of the Lyve1+ perivascular macrophages in the mouse brain.". Nature Communications 13 (1). 2022. doi:10.1038/s41467-022-35166-9. PMID 36450771. Bibcode: 2022NatCo..13.7366S.
- ↑ "Identification of the lipophilic factor produced by macrophages that stimulates steroidogenesis". Endocrinology 141 (3): 953–958. March 2000. doi:10.1210/endo.141.3.7350. PMID 10698170.
- ↑ "Macrophages Facilitate Electrical Conduction in the Heart". Cell 169 (3): 510–522.e20. April 2017. doi:10.1016/j.cell.2017.03.050. PMID 28431249.
- ↑ 17.0 17.1 17.2 17.3 17.4 "Exploring the full spectrum of macrophage activation". Nature Reviews. Immunology 8 (12): 958–969. December 2008. doi:10.1038/nri2448. PMID 19029990.
- ↑ "The development and maintenance of resident macrophages". Nature Immunology 17 (1): 2–8. January 2016. doi:10.1038/ni.3341. PMID 26681456.
- ↑ "Tissue-Resident Macrophage Ontogeny and Homeostasis". Immunity 44 (3): 439–449. March 2016. doi:10.1016/j.immuni.2016.02.024. PMID 26982352.
- ↑ "The journey from stem cell to macrophage". Annals of the New York Academy of Sciences 1319 (1): 1–18. June 2014. doi:10.1111/nyas.12393. PMID 24673186. Bibcode: 2014NYASA1319....1P.
- ↑ "Two populations of self-maintaining monocyte-independent macrophages exist in adult epididymis and testis". Proceedings of the National Academy of Sciences of the United States of America 118 (1). January 2021. doi:10.1073/pnas.2013686117. PMID 33372158. Bibcode: 2021PNAS..11813686W.
- ↑ 22.0 22.1 22.2 "Inflammation in wound repair: molecular and cellular mechanisms". The Journal of Investigative Dermatology 127 (3): 514–25. March 2007. doi:10.1038/sj.jid.5700701. PMID 17299434. "=Monocytes/macrophages. Unless stimuli for neutrophil recruitment persist at the wound site, the neutrophil infiltration ceases after few days, and expended neutrophils are themselves phagocytosed by macrophages, which are present at the wound side within 2 days after injury.".
- ↑ "Neutrophil extracellular traps enhance macrophage killing of bacterial pathogens" (in EN). Science Advances 7 (37). September 2021. doi:10.1126/sciadv.abj2101. PMID 34516771. Bibcode: 2021SciA....7.2101M.
- ↑ "A Role of Macrophages: An Overview" (in en). Journal of Drug Delivery and Therapeutics 7 (6): 91–103. 2017-11-15. doi:10.22270/jddt.v7i6.1521. ISSN 2250-1177. http://jddtonline.info/index.php/jddt/article/view/1521.
- ↑ "Hijacking of Macrophages by Salmonella (310r) Through 'Types III' Secretion Like Exocytotic Signalling: A Mechanism for Infection of Chicken Ileum". Indian Journal of Poultry Science 35 (3): 276–281. 2000. https://www.researchgate.net/publication/230823526.
- ↑ Kirschnek, Susanne; Ying, Songmin; Fischer, Silke F.; Häcker, Hans; Villunger, Andreas; Hochrein, Hubertus; Häcker, Georg (2005-01-15). "Phagocytosis-Induced Apoptosis in Macrophages Is Mediated by Up-Regulation and Activation of the Bcl-2 Homology Domain 3-Only Protein Bim1". The Journal of Immunology 174 (2): 671–679. doi:10.4049/jimmunol.174.2.671. ISSN 0022-1767. PMID 15634885. https://journals.aai.org/jimmunol/article/174/2/671/72667/Phagocytosis-Induced-Apoptosis-in-Macrophages-Is?.
- ↑ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 "Macrophage cytokines: involvement in immunity and infectious diseases". Frontiers in Immunology 5: 491. 2014-10-07. doi:10.3389/fimmu.2014.00491. PMID 25339958.
- ↑ 28.0 28.1 28.2 28.3 28.4 (in en) Kuby Immunology (8th ed.). New York, New York: W. H. Freeman. May 25, 2018. ISBN 978-1-4641-8978-4.
- ↑ 29.0 29.1 29.2 "Microbial Phagocytic Receptors and Their Potential Involvement in Cytokine Induction in Macrophages". Frontiers in Immunology 12. 2021-04-29. doi:10.3389/fimmu.2021.662063. PMID 33995386.
- ↑ 30.0 30.1 "The Phagocytic Function of Macrophage-Enforcing Innate Immunity and Tissue Homeostasis". International Journal of Molecular Sciences 19 (1): 92. December 2017. doi:10.3390/ijms19010092. PMID 29286292.
- ↑ "Phagocytosis: Our Current Understanding of a Universal Biological Process". Frontiers in Immunology 11. 2020-06-02. doi:10.3389/fimmu.2020.01066. PMID 32582172.
- ↑ "C3 receptors on macrophages". Journal of Cell Science. Supplement 9 (Supplement_9): 67–97. 1988-01-01. doi:10.1242/jcs.1988.Supplement_9.4. PMID 2978518.
- ↑ "Reactive oxygen species and cell signaling: respiratory burst in macrophage signaling". American Journal of Respiratory and Critical Care Medicine 166 (12 Pt 2): S4–S8. December 2002. doi:10.1164/rccm.2206007. PMID 12471082.
- ↑ "NF-κB signaling in inflammation". Signal Transduction and Targeted Therapy 2 (1): 17023–. 2017-07-14. doi:10.1038/sigtrans.2017.23. PMID 29158945.
- ↑ 35.00 35.01 35.02 35.03 35.04 35.05 35.06 35.07 35.08 35.09 35.10 35.11 35.12 35.13 35.14 35.15 35.16 35.17 (in en) Janeway's Immunobiology (10th ed.). New York, New York: W. W. Norton & Company. 2022. ISBN 978-0-393-88487-6.
- ↑ "Macrophage responses to lipopolysaccharide are modulated by a feedback loop involving prostaglandin E2, dual specificity phosphatase 1 and tristetraprolin". Scientific Reports 7 (4350). 2017-07-28. doi:10.1038/s41598-017-04100-1. PMID 28659609.
- ↑ "Major role of adipocyte prostaglandin E2 in lipolysis-induced macrophage recruitment". Journal of Lipid Research 57 (4): 663–73. 2016-02-24. doi:10.1194/jlr.m066530. PMID 26912395.
- ↑ "Inflammation in wound repair: molecular and cellular mechanisms". The Journal of Investigative Dermatology 127 (3): 514–525. March 2007. doi:10.1038/sj.jid.5700701. PMID 17299434.
- ↑ "Changing the paradigm of IFN-γ at the interface between innate and adaptive immunity: Macrophage-derived IFN-γ". Journal of Leukocyte Biology 108 (1): 419–426. July 2020. doi:10.1002/JLB.4MIR0420-619RR. PMID 32531848.
- ↑ "Filopodia act as phagocytic tentacles and pull with discrete steps and a load-dependent velocity". Proceedings of the National Academy of Sciences of the United States of America 104 (28): 11633–11638. July 2007. doi:10.1073/pnas.0702449104. PMID 17620618. Bibcode: 2007PNAS..10411633K.
- ↑ 41.0 41.1 41.2 "Macrophages: Their Untold Story in T Cell Activation and Function". International Review of Cell and Molecular Biology (Elsevier) 342: 73–93. 2019. doi:10.1016/bs.ircmb.2018.07.001. ISBN 978-0-12-815381-9. PMID 30635094.
- ↑ 42.0 42.1 42.2 "Antigen presentation to naive CD4 T cells in the lymph node". Nature Immunology 4 (8): 733–739. August 2003. doi:10.1038/ni957. PMID 12888794.
- ↑ 43.0 43.1 43.2 (in en) Janeway's Immunobiology (9th ed.). New York, New York: Garland Science. 2016. pp. 363–364. ISBN 978-0-8153-4505-3.
- ↑ 44.0 44.1 "Lymph node macrophages". Journal of Innate Immunity 4 (5–6): 424–436. 2012. doi:10.1159/000337007. PMID 22488251.
- ↑ "The Surprising Story of IL-2" (in en). The American Journal of Pathology 190 (9): 1776–1781. September 2020. doi:10.1016/j.ajpath.2020.05.007. PMID 32828360.
- ↑ "The 3 major types of innate and adaptive cell-mediated effector immunity". The Journal of Allergy and Clinical Immunology 135 (3): 626–635. March 2015. doi:10.1016/j.jaci.2014.11.001. PMID 25528359.
- ↑ 47.0 47.1 47.2 "Defects in Macrophage Reprogramming in Cancer Therapy: The Negative Impact of PD-L1/PD-1". Frontiers in Immunology 12. 2021-06-23. doi:10.3389/fimmu.2021.690869. PMID 34248982.
- ↑ 48.0 48.1 "T cell-macrophage interactions and granuloma formation in vasculitis". Frontiers in Immunology 5: 432. 2014. doi:10.3389/fimmu.2014.00432. PMID 25309534.
- ↑ 49.0 49.1 49.2 49.3 "Macrophage Activation and Functions during Helminth Infection: Recent Advances from the Laboratory Mouse". Journal of Immunology Research 2018. 2018-07-02. doi:10.1155/2018/2790627. PMID 30057915.
- ↑ "Alternative activation of macrophages". Nature Reviews. Immunology 3 (1): 23–35. January 2003. doi:10.1038/nri978. PMID 12511873.
- ↑ "IL-1 receptor 2 (IL-1R2) and its role in immune regulation". Brain, Behavior, and Immunity 32: 1–8. August 2013. doi:10.1016/j.bbi.2012.11.006. PMID 23195532.
- ↑ "Antigen Cross-Presentation by Macrophages". Frontiers in Immunology 11. 2020-07-08. doi:10.3389/fimmu.2020.01276. PMID 32733446.
- ↑ "The antigen presenting cells instruct plasma cell differentiation". Frontiers in Immunology 4: 504. January 2014. doi:10.3389/fimmu.2013.00504. PMID 24432021.
- ↑ Robbins Pathologic Basis of Disease. Philadelphia: W.B Saunders Company. 1999. ISBN 978-0-7216-7335-6.
- ↑ "The lymphocyte story". New Scientist (1605). https://www.newscientist.com/channel/health/hiv/mg11716050.100. Retrieved 2007-09-13.
- ↑ 56.0 56.1 56.2 56.3 "Macrophage Phenotypes Regulate Scar Formation and Chronic Wound Healing". International Journal of Molecular Sciences 18 (7): 1545. July 2017. doi:10.3390/ijms18071545. PMID 28714933.
- ↑ "Tumor associated macrophages and neutrophils in tumor progression". Journal of Cellular Physiology 228 (7): 1404–1412. July 2013. doi:10.1002/jcp.24260. PMID 23065796.
- ↑ "Endoplasmic reticulum stress and atherosclerosis". Nature Medicine 16 (4): 396–399. April 2010. doi:10.1038/nm0410-396. PMID 20376052.
- ↑ "Endoplasmic reticulum stress controls M2 macrophage differentiation and foam cell formation". The Journal of Biological Chemistry 287 (15): 11629–11641. April 2012. doi:10.1074/jbc.M111.338673. PMID 22356914.
- ↑ "Distinguishing unloading- versus reloading-induced changes in rat soleus muscle". Muscle & Nerve 16 (1): 99–108. January 1993. doi:10.1002/mus.880160116. PMID 8423838.
- ↑ 61.0 61.1 61.2 "Differential response of macrophage subpopulations to soleus muscle reloading after rat hindlimb suspension". Journal of Applied Physiology 77 (1): 290–297. July 1994. doi:10.1152/jappl.1994.77.1.290. PMID 7961247.
- ↑ "Macrophage invasion does not contribute to muscle membrane injury during inflammation". Journal of Leukocyte Biology 65 (4): 492–498. April 1999. doi:10.1002/jlb.65.4.492. PMID 10204578.
- ↑ 63.0 63.1 Skeletal Muscle Repair and Regeneration. Advances in Muscle Research. 3. 2008.
- ↑ "Modulation of macrophage activation state protects tissue from necrosis during critical limb ischemia in thrombospondin-1-deficient mice". PLOS ONE 3 (12). 2008. doi:10.1371/journal.pone.0003950. PMID 19079608. Bibcode: 2008PLoSO...3.3950B.
- ↑ 65.0 65.1 de la Torre J., Sholar A. (2006). Wound healing: Chronic wounds. Emedicine.com. Accessed 20 January 2008.
- ↑ "The phases of cutaneous wound healing". Expert Reviews in Molecular Medicine (Cambridge University Press) 5. 21 March 2003. http://www-ermm.cbcu.cam.ac.uk/03005829a.pdf.
- ↑ "Wounds: biology, pathology, and management.". Essential practice of surgery. New York, NY: Springer. 2003. pp. 77–88. ISBN 978-0-387-22744-3. http://recon.stanford.edu/Articles/LorenzWH.pdf.
- ↑ "Identification of splenic reservoir monocytes and their deployment to inflammatory sites". Science 325 (5940): 612–616. July 2009. doi:10.1126/science.1175202. PMID 19644120. Bibcode: 2009Sci...325..612S.
- ↑ "Immunology. Dispensable but not irrelevant". Science 325 (5940): 549–550. July 2009. doi:10.1126/science.1178329. PMID 19644100. Bibcode: 2009Sci...325..549J.
- ↑ "Surgical physiology of wound healing: a review". Journal of Postgraduate Medicine 43 (2): 52–56. 1997. PMID 10740722. http://www.jpgmonline.com/article.asp?issn=0022-3859;year=1997;volume=43;issue=2;spage=52;epage=6;aulast=Deodhar.
- ↑ Rosenberg L., de la Torre J. (2006). Wound Healing, Growth Factors. Emedicine.com. Accessed 20 January 2008.
- ↑ "Macrophages restrain contraction of an in vitro wound healing model". Inflammation 28 (4): 207–214. August 2004. doi:10.1023/B:IFLA.0000049045.41784.59. PMID 15673162.
- ↑ "The role of apoptosis in wound healing". The International Journal of Biochemistry & Cell Biology 30 (9): 1019–1030. September 1998. doi:10.1016/S1357-2725(98)00058-2. PMID 9785465.
- ↑ "Update on wound dressings: Indications and best use". Clinical Techniques in Equine Practice 3 (2): 148–163. June 2004. doi:10.1053/j.ctep.2004.08.006.
- ↑ 75.0 75.1 "Scientists identify cell that could hold the secret to limb regeneration". the verge.com. 2013-05-23. https://www.theverge.com/2013/5/23/4358418/salamander-macrophages-could-aid-limb-regeneration. "Researchers have identified a cell that aids limb regrowth in Salamanders. Macrophages are a type of repairing cell that devour dead cells and pathogens, and trigger other immune cells to respond to pathogens."
- ↑ 76.0 76.1 "Macrophages are required for adult salamander limb regeneration". Proceedings of the National Academy of Sciences of the United States of America 110 (23): 9415–9420. June 2013. doi:10.1073/pnas.1300290110. PMID 23690624. Bibcode: 2013PNAS..110.9415G.
- ↑ "Iron metabolism and iron disorders revisited in the hepcidin era". Haematologica 105 (2): 260–272. 2020. doi:10.3324/haematol.2019.232124. PMID 31949017.
- ↑ "Lysosomes in malanin phagocytosis and synthesis" (in En). Nature 216 (5110): 67. October 1967. doi:10.1038/216067a0. PMID 6050674. Bibcode: 1967Natur.216...67M.
- ↑ "Cellular and subcellular differentiation of melanin phagocytosis and synthesis by lysosomal and melanosomal activity". The Journal of Investigative Dermatology 46 (1): 70–75. January 1966. doi:10.1038/jid.1966.11. PMID 5905254.
- ↑ "Unveiling skin macrophage dynamics explains both tattoo persistence and strenuous removal". The Journal of Experimental Medicine 215 (4): 1115–1133. April 2018. doi:10.1084/jem.20171608. PMID 29511065.
- ↑ "Tissue-specific signals control reversible program of localization and functional polarization of macrophages". Cell 157 (4): 832–844. May 2014. doi:10.1016/j.cell.2014.04.016. PMID 24792964.
- ↑ "Environment drives selection and function of enhancers controlling tissue-specific macrophage identities". Cell 159 (6): 1327–1340. December 2014. doi:10.1016/j.cell.2014.11.023. PMID 25480297.
- ↑ "Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility". Cell 158 (2): 300–313. July 2014. doi:10.1016/j.cell.2014.04.050. PMID 25036630.
- ↑ "Resident Macrophages Cloak Tissue Microlesions to Prevent Neutrophil-Driven Inflammatory Damage". Cell 177 (3): 541–555.e17. April 2019. doi:10.1016/j.cell.2019.02.028. PMID 30955887.
- ↑ "Origin and Differentiation of Nerve-Associated Macrophages". Journal of Immunology 204 (2): 271–279. January 2020. doi:10.4049/jimmunol.1901077. PMID 31907269.
- ↑ "Phagocytosis". Macrophages: Biology and Role in the Pathology of Diseases. Springer New York. 2014. pp. 91–109. doi:10.1007/978-1-4939-1311-4_5. ISBN 978-1-4939-1310-7.
- ↑ "Functions of ROS in Macrophages and Antimicrobial Immunity". Antioxidants 10 (2): 313. February 2021. doi:10.3390/antiox10020313. PMID 33669824.
- ↑ "Macrophage defense mechanisms against intracellular bacteria". Immunological Reviews 264 (1): 182–203. March 2015. doi:10.1111/imr.12266. PMID 25703560.
- ↑ Sherris Medical Microbiology (4th ed.). McGraw Hill. 2004. ISBN 978-0-8385-8529-0.
- ↑ "Chikungunya disease: infection-associated markers from the acute to the chronic phase of arbovirus-induced arthralgia". PLOS Neglected Tropical Diseases 6 (3). 2012. doi:10.1371/journal.pntd.0001446. PMID 22479654.
- ↑ "Atherosclerosis: role of chemokines and macrophages". Expert Reviews in Molecular Medicine 3 (25): 1–18. November 2001. doi:10.1017/S1462399401003696. PMID 14585150.
- ↑ "Cardiac macrophages and their role in ischaemic heart disease". Cardiovascular Research 102 (2): 240–248. May 2014. doi:10.1093/cvr/cvu025. PMID 24501331.
- ↑ "Neutrophils incite and macrophages avert electrical storm after myocardial infarction". Nature Cardiovascular Research 1 (7): 649–664. July 2022. doi:10.1038/s44161-022-00094-w. PMID 36034743.
- ↑ "Macrophage". Future Virology 6 (2): 187–208. February 2011. doi:10.2217/fvl.10.93.
- ↑ "Macrophages and their relevance in Human Immunodeficiency Virus Type I infection". Retrovirology 9 (1). October 2012. doi:10.1186/1742-4690-9-82. PMID 23035819.
- ↑ "Macrophage diversity enhances tumor progression and metastasis". Cell 141 (1): 39–51. April 2010. doi:10.1016/j.cell.2010.03.014. PMID 20371344.
- ↑ 97.0 97.1 "The role of myeloid cells in cancer therapies". Nature Reviews. Cancer 16 (7): 447–462. July 2016. doi:10.1038/nrc.2016.54. PMID 27339708.
- ↑ "A malignant flame. Understanding chronic inflammation, which contributes to heart disease, Alzheimer's and a variety of other ailments, may be a key to unlocking the mysteries of cancer". Scientific American 297 (1): 60–67. July 2007. doi:10.1038/scientificamerican0707-60. PMID 17695843. Bibcode: 2007SciAm.297a..60S.
- ↑ "Macrophages regulate the angiogenic switch in a mouse model of breast cancer". Cancer Research 66 (23): 11238–11246. December 2006. doi:10.1158/0008-5472.can-06-1278. PMID 17114237.
- ↑ Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 2002; 196:254–65.
- ↑ "In vitro human tumor-associated macrophage model implicates macrophage proliferation as a mechanism for maintaining tumor-associated macrophage populations". Cancer Research 78 (13 Supplement): 4060. July 2018. doi:10.1158/1538-7445.AM2018-4060. http://cancerres.aacrjournals.org/content/78/13_Supplement/4060.short.
- ↑ "Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy". The Journal of Experimental Medicine 193 (6): 727–740. March 2001. doi:10.1084/jem.193.6.727. PMID 11257139.
- ↑ "CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis". Nature 475 (7355): 222–225. June 2011. doi:10.1038/nature10138. PMID 21654748.
- ↑ "Angiotensin II drives the production of tumor-promoting macrophages". Immunity 38 (2): 296–308. February 2013. doi:10.1016/j.immuni.2012.10.015. PMID 23333075.
- ↑ "SCS macrophages suppress melanoma by restricting tumor-derived vesicle-B cell interactions". Science 352 (6282): 242–246. April 2016. doi:10.1126/science.aaf1328. PMID 26989197. Bibcode: 2016Sci...352..242P.
- ↑ "The interaction of anticancer therapies with tumor-associated macrophages". The Journal of Experimental Medicine 212 (4): 435–445. April 2015. doi:10.1084/jem.20150295. PMID 25753580.
- ↑ "Macrophage regulation of tumor responses to anticancer therapies". Cancer Cell 23 (3): 277–286. March 2013. doi:10.1016/j.ccr.2013.02.013. PMID 23518347.
- ↑ "Tumour-associated macrophages act as a slow-release reservoir of nano-therapeutic Pt(IV) pro-drug". Nature Communications 6. October 2015. doi:10.1038/ncomms9692. PMID 26503691. Bibcode: 2015NatCo...6.8692M.
- ↑ "Low-dose irradiation programs macrophage differentiation to an iNOS⁺/M1 phenotype that orchestrates effective T cell immunotherapy". Cancer Cell 24 (5): 589–602. November 2013. doi:10.1016/j.ccr.2013.09.014. PMID 24209604.
- ↑ "Macrophage IL-10 blocks CD8+ T cell-dependent responses to chemotherapy by suppressing IL-12 expression in intratumoral dendritic cells". Cancer Cell 26 (5): 623–637. November 2014. doi:10.1016/j.ccell.2014.09.006. PMID 25446896.
- ↑ "Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy". Cancer Discovery 1 (1): 54–67. June 2011. doi:10.1158/2159-8274.CD-10-0028. PMID 22039576.
- ↑ "TH2-Polarized CD4(+) T Cells and Macrophages Limit Efficacy of Radiotherapy". Cancer Immunology Research 3 (5): 518–525. May 2015. doi:10.1158/2326-6066.CIR-14-0232. PMID 25716473.
- ↑ "Recruitment of myeloid but not endothelial precursor cells facilitates tumor regrowth after local irradiation". Cancer Research 70 (14): 5679–5685. July 2010. doi:10.1158/0008-5472.CAN-09-4446. PMID 20631066.
- ↑ "In vivo imaging reveals a tumor-associated macrophage-mediated resistance pathway in anti-PD-1 therapy". Science Translational Medicine 9 (389). May 2017. doi:10.1126/scitranslmed.aal3604. PMID 28490665.
- ↑ "CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models". Cancer Research 74 (18): 5057–5069. September 2014. doi:10.1158/0008-5472.CAN-13-3723. PMID 25082815.
- ↑ "Targeting tumor-associated macrophages with anti-CSF-1R antibody reveals a strategy for cancer therapy". Cancer Cell 25 (6): 846–859. June 2014. doi:10.1016/j.ccr.2014.05.016. PMID 24898549.
- ↑ "Macrophages and therapeutic resistance in cancer". Cancer Cell 27 (4): 462–472. April 2015. doi:10.1016/j.ccell.2015.02.015. PMID 25858805.
- ↑ "TLR1/2 ligand enhances antitumor efficacy of CTLA-4 blockade by increasing intratumoral Treg depletion". Proceedings of the National Academy of Sciences of the United States of America 116 (21): 10453–10462. May 2019. doi:10.1073/pnas.1819004116. PMID 31076558. Bibcode: 2019PNAS..11610453S.
- ↑ Klichinsky, Michael; Ruella, Marco; Shestova, Olga; Lu, Xueqing Maggie; Best, Andrew; Zeeman, Martha; Schmierer, Maggie; Gabrusiewicz, Konrad et al. (August 2020). "Human chimeric antigen receptor macrophages for cancer immunotherapy" (in en). Nature Biotechnology 38 (8): 947–953. doi:10.1038/s41587-020-0462-y. ISSN 1087-0156. PMID 32361713.
- ↑ Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW. Obesity is associated with macrophage accumulation in adipose tissue" Journal of Clinical Investigation 2003; 112:1796–808.
- ↑ "Molecular pathways linking adipose innervation to insulin action in obesity and diabetes mellitus". Nature Reviews. Endocrinology 15 (4): 207–225. April 2019. doi:10.1038/s41574-019-0165-y. PMID 30733616.
- ↑ "Macrophage beta2-adrenergic receptor is dispensable for the adipose tissue inflammation and function". Molecular Metabolism 48. June 2021. doi:10.1016/j.molmet.2021.101220. PMID 33774223.
- ↑ "CCR2 deficiency leads to increased eosinophils, alternative macrophage activation, and type 2 cytokine expression in adipose tissue". Journal of Leukocyte Biology 98 (4): 467–477. October 2015. doi:10.1189/jlb.3HI0115-018R. PMID 25934927.
- ↑ "Adipose tissue macrophages: going off track during obesity". Diabetologia 59 (5): 879–894. May 2016. doi:10.1007/s00125-016-3904-9. PMID 26940592.
- ↑ "Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans". Journal of Lipid Research 46 (11): 2347–2355. November 2005. doi:10.1194/jlr.M500294-JLR200. PMID 16150820.
- ↑ "Diversity of Intestinal Macrophages in Inflammatory Bowel Diseases". Frontiers in Immunology 6: 613. 2015-12-07. doi:10.3389/fimmu.2015.00613. PMID 26697009.
- ↑ 127.0 127.1 127.2 "Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity". The Journal of Clinical Investigation 115 (1): 66–75. January 2005. doi:10.1172/JCI19229. PMID 15630445.
- ↑ 128.0 128.1 "Mucosal macrophages in intestinal homeostasis and inflammation". Journal of Innate Immunity 3 (6): 550–564. 2011. doi:10.1159/000329099. PMID 22025201.
- ↑ "Macrophages in intestinal homeostasis and inflammation". Immunological Reviews 260 (1): 102–117. July 2014. doi:10.1111/imr.12192. PMID 24942685.
- ↑ "Macrophage vesicles starve bacteria of iron". Nature Metabolism 5 (1): 10–12. January 2023. doi:10.1038/s42255-022-00719-1. PMID 36658401.
- ↑ "Origin and functions of tissue macrophages". Immunity 41 (1): 21–35. July 2014. doi:10.1016/j.immuni.2014.06.013. PMID 25035951.
- ↑ "From macrophage biology to macrophage-based cellular immunotherapies". Gene Therapy 28 (9): 473–476. September 2021. doi:10.1038/s41434-021-00221-5. PMID 33542457.
- ↑ "Origin and functions of tissue macrophages". Immunity 41 (1): 21–35. July 2014. doi:10.1016/j.immuni.2014.06.013. PMID 25035951.
- ↑ "New Insights Into Tissue Macrophages: From Their Origin to the Development of Memory". Immune Network 15 (4): 167–176. August 2015. doi:10.4110/in.2015.15.4.167. PMID 26330802.
- ↑ "Physiology and diseases of tissue-resident macrophages". Nature 618 (7966): 698–707. June 2023. doi:10.1038/s41586-023-06002-x. PMID 37344646. Bibcode: 2023Natur.618..698L.
 |


