Chemistry:Acetylide
In chemistry, an acetylide is a compound that can be viewed as the result of replacing one or both hydrogen atoms of acetylene (ethyne) HC≡CH by metallic or other cations. Calcium carbide is an important industrial compound, which has long been used to produce acetylene for welding and illumination. It is also a major precursor to vinyl chloride.[1] Other acetylides are reagents in organic synthesis.
Nomenclature
The term acetylide is used loosely. It apply to an acetylene RC≡CM, where R = H or a side chain that is usually organic.[2] The nomenclature can be ambiguous with regards to the distinction between compounds of the type MC
2R and M
2C
2. When both hydrogens of acetylene are replaced by metals, the compound can also be called carbide, e.g. calcium carbide Ca2+[−
C≡C−
], which is calcium acetylide. When only one hydrogen atom is replaced, the anion may be called hydrogen acetylide or the prefix mono- may be attached to the metal, as in monosodium acetylide or sodium hydrogen acetylide, Na+
[−
C≡CH]. Metal acetylide may be described as salts, but that description rarely comports with crystallographic analysis.
Alkali and alkaline earth acetylides
In the absence of additional ligands, metal acetylides adopt polymeric structures wherein the acetylide groups are bridging ligands. Alkali metal acetylides have the general formula [M
2C
2 (M = Li, Na, K, etc).They are sometimes represented as [M+
]
2[−
C≡C−
] but the C---M bonding might also be described as polar covalent. They dissolve without decomposition in ammonia. Such solutions are proposed to contain solvated −
C≡C−
ions.[3]
Alkali metal and alkaline earth metal acetylides have the general formula [M'C
2 (M' = Mg, Ca, etc). Rather than salt-like, they can be considered Zintl phase compounds, containing −
C≡C−
ions, with a triple bond between the two carbon atoms. They undergo ready hydrolysis to form acetylene and metal oxides:
- CaC
2 + 2 H
2O → Ca(OH)
2 + C
2H
2
The −
C≡C−
ion has a closed shell ground state of 1Σ+g, making it isoelectronic to a neutral molecule N
2, which may afford it some gas-phase stability.[4] polymeric structures wherein the acetylide groups are bridging ligands.
-
Structure of sodium acetylide [Na+
]
2[−
C≡C−
].[5] Color code: gray = C, blue = Na. -
Structure and unit cell of potassium acetylide [K+
]
2[−
C≡C−
].[6] -
Structure of the cluster formed from PhC≡CLi complexed to N,N,N′,N′-tetramethyl-1,6-diaminohexane (methylene groups omitted for clarity). Color key: turquoise = Li, blue = N.[7]
Transition metal acetylides
Acetylides of the transition metals, show evidence of covalent character, e. g. for being neither dissolved nor decomposed by water and by radically different chemical reactions. The inventory of such complexes numbers in the hundreds. Even the stoichiometrically simple silver acetylide and copper acetylide appear highly covalent..
File:XAPGIF2.png | Portion of the structure of the polymer copper(I) phenylacetylide Cu+
[−
C≡C–C
6H
5].[8]
Preparation
Of the type MC≡CR
Acetylene and terminal alkynes are weak acids:[9]
- RC≡CH + R″M ⇌ R″H + RC≡CM
Monopotassium and monosodium acetylide can be prepared by reacting acetylene with bases like sodium amide[10] or with the elemental metals, often at room temperature and atmospheric pressure.[9] Copper(I) acetylide can be prepared by passing acetylene through an aqueous solution of copper(I) chloride because of a low solubility equilibrium.[9] Similarly, silver acetylides can be obtained from silver nitrate.
In organic synthesis, acetylides are usually prepared by treating acetylene and alkynes with organometallic[11] or inorganic[10] Classically, liquid ammonia was used for deprotonations, but ethers are now more commonly used.
Lithium amide,[9] LiHMDS,[12] or organolithium reagents, such as butyllithium (BuLi),[11] are frequently used to form lithium acetylides:
- HC≡CH + BuLi → LiC≡CH + BuH
Of the type MC≡CM and CaC
2
Calcium carbide is prepared industrially by heating carbon with lime (calcium oxide) at approximately 2,000 °C.[1] A similar process can be used to produce lithium carbide.
Dilithium acetylide, Li
2C
2, competes with the preparation of the monolithium derivative LiC
2H.[10]
Reactions
Ionic acetylides are typically decomposed by water with evolution of acetylene:
- Ca2+[−
C≡C−
] + 2 H
2O → Ca(OH)
2 + HC≡CH
- RC≡C−
Na+
+ H
2O → RC≡CH + NaOH
Acetylides of the type RC≡CM are widely used in alkynylations in organic chemistry. They are nucleophiles that add to a variety of electrophilic and unsaturated substrates.
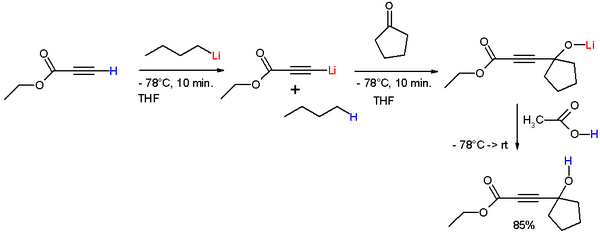
A classic application is the Favorskii reaction, such as in the sequence shown below. Here ethyl propiolate is deprotonated by n-butyllithium to give the corresponding lithium acetylide. This acetylide adds to the carbonyl center of cyclopentanone. Hydrolysis liberates the alkynyl alcohol.[13]
The dimerization of acetylene to vinylacetylene proceeds by insertion of acetylene into a copper(I) acetylide complex.[14]
Coupling reactions
The copper-catalyzed Click reaction of terminal alkynes and azides proceeds via copper(I) acetylide intermediates.[15]
Acetylides are sometimes used as intermediates in coupling reactions. Examples include Sonogashira coupling, Cadiot-Chodkiewicz coupling, Glaser coupling and Eglinton coupling.
Hazards
Some acetylides are notoriously explosive.[16] Formation of acetylides poses a risk in handling of gaseous acetylene in presence of metals such as mercury, silver or copper, or alloys with their high content (brass, bronze, silver solder).
See also
- Sodium hydrogen acetylide
- Calcium acetylide
- Strontium acetylide
- Barium acetylide
- Copper(I) acetylide
- Silver(I) acetylide
- Ethynyl
- Ethynyl radical
- Diatomic carbon (neutral C2)
- Acetylenediol
References
- ↑ 1.0 1.1 Holzrichter, Klaus; Knott, Alfons; Mertschenk, Bernd; Salzinger, Josef (2013). "Calcium Carbide". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–14. doi:10.1002/14356007.a04_533.pub2. ISBN 978-3-527-30673-2.
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "acetylides". doi:10.1351/goldbook.A00067
- ↑ Hamberger, Markus; Liebig, Stefan; Friedrich, Ute; Korber, Nikolaus; Ruschewitz, Uwe (21 December 2012). "Evidence of Solubility of the Acetylide Ion C2−2: Syntheses and Crystal Structures of K2C2·2 NH3, Rb2C2·2 NH3, and Cs2C2·7 NH3". Angewandte Chemie International Edition 51 (52): 13006–13010. doi:10.1002/anie.201206349. PMID 23161511.
- ↑ Sommerfeld, T.; Riss, U.; Meyer, H.-D.; Cederbaum, L. (August 1997). "Metastable C2−2 Dianion". Physical Review Letters 79 (7): 1237–1240. doi:10.1103/PhysRevLett.79.1237. Bibcode: 1997PhRvL..79.1237S.
- ↑ Klöss, Karl-Heinz; Hinz-Hübner, Dirk; Ruschewitz, Uwe (2002). "Über eine neue Modifikation des Na 2 C2". Zeitschrift für Anorganische und Allgemeine Chemie 628 (12): 2701–2704. doi:10.1002/1521-3749(200212)628:12<2701::AID-ZAAC2701>3.0.CO;2-#.
- ↑ S. Hemmersbach, B. Zibrowius, U. Ruschewitz (1999): "Na2C2 und K2C2: Synthese, Kristallstruktur und spektroskopische Eigenschaften". Zeitschrift für anorganische und allgemeine Chemie, volume 625, issue 9, pages 1440-1446. doi:10.1002/(SICI)1521-3749(199909)625:9<1440::AID-ZAAC1440>3.0.CO;2-R
- ↑ Schubert, Bernd; Weiss, Erwin (1983). "(PHCCLi)4(tmhda)2, A Polymeric Organolithium Compound with Cubic Li4C4 Structural Units". Angewandte Chemie International Edition in English 22 (6): 496–497. doi:10.1002/anie.198304961.
- ↑ Chui, Stephen S. Y.; Ng, Miro F. Y.; Che, Chi-Ming (2005). "Structure Determination of Homoleptic AuI, AgI, and CuI Aryl/Alkylethynyl Coordination Polymers by X-ray Powder Diffraction". Chemistry: A European Journal 11 (6): 1739–1749. doi:10.1002/chem.200400881. PMID 15669067.
- ↑ 9.0 9.1 9.2 9.3 Viehe, Heinz Günter (1969). "Chemistry of Acetylenes". Angewandte Chemie (New York: Marcel Dekker) 84 (8): 170–179 & 225–241. doi:10.1002/ange.19720840843.
- ↑ 10.0 10.1 10.2 Coffman, Donald D. (1940). "Dimethylethhynylcarbinol". Organic Syntheses 40: 20. doi:10.15227/orgsyn.020.0040.
- ↑ 11.0 11.1 Midland, M. M.; McLoughlin, J. I.; Werley, Ralph T. Jr. (1990). "Preparation and Use of Lithium Acetylide: 1-Methyl-2-ethynyl-endo-3,3-dimethyl-2-norbornanol". Organic Syntheses 68: 14. doi:10.15227/orgsyn.068.0014.
- ↑ Reich, Melanie (August 24, 2001). "Addition of a lithium acetylide to an aldehyde; 1-(2-pentyn-4-ol)-cyclopent-2-en-1-ol". ChemSpider Synthetic Pages: 137. doi:10.1039/SP137.
- ↑ Midland, M. Mark; Tramontano, Alfonso; Cable, John R. (1980). "Synthesis of alkyl 4-hydroxy-2-alkynoates". The Journal of Organic Chemistry 45 (1): 28–29. doi:10.1021/jo01289a006.
- ↑ Trotuş, Ioan-Teodor; Zimmermann, Tobias; Schüth, Ferdi (2014). "Catalytic Reactions of Acetylene: A Feedstock for the Chemical Industry Revisited". Chemical Reviews 114 (3): 1761–1782. doi:10.1021/cr400357r. PMID 24228942.
- ↑ Worrell, B. T.; Malik, J. A.; Fokin, V. V. (2013). "Direct Evidence of a Dinuclear Copper Intermediate in Cu(I)-Catalyzed Azide-Alkyne Cycloadditions". Science 340 (6131): 457–460. doi:10.1126/science.1229506. PMID 23558174. Bibcode: 2013Sci...340..457W.
- ↑ Cataldo, Franco; Casari, Carlo S. (2007). "Synthesis, Structure and Thermal Properties of Copper and Silver Polyynides and Acetylides". Journal of Inorganic and Organometallic Polymers and Materials 17 (4): 641–651. doi:10.1007/s10904-007-9150-3. ISSN 1574-1443.
 |
![Structure of sodium acetylide [Na+ ] 2[− C≡C− ].[5] Color code: gray = C, blue = Na.](/wiki/images/thumb/b/be/Na2C2structure.jpg/250px-Na2C2structure.jpg%3Futm_source%3Dcommons.wikimedia.org%26utm_campaign%3Dimageinfo%26utm_content%3Dthumbnail)
![Structure and unit cell of potassium acetylide [K+ ] 2[− C≡C− ].[6]](/wiki/images/thumb/8/86/K2C2_unit_cell.png/330px-K2C2_unit_cell.png%3Futm_source%3Dcommons.wikimedia.org%26utm_campaign%3Dimageinfo%26utm_content%3Dthumbnail)
![Structure of the cluster formed from PhC≡CLi complexed to N,N,N′,N′-tetramethyl-1,6-diaminohexane (methylene groups omitted for clarity). Color key: turquoise = Li, blue = N.[7]](/wiki/images/thumb/0/09/CAMNEJ.svg/242px-CAMNEJ.svg.png)
