Chemistry:Sodium amide
This article may contain an excessive number of citations. (March 2026) (Learn how and when to remove this template message) |
| File:Sodium amide.png | |
| |
| Names | |
|---|---|
IUPAC name
| |
| Other names
Sodamide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 1390 |
| |
| |
| Properties | |
| NaNH 2 | |
| Molar mass | 39.013 g·mol−1 |
| Appearance | Colourless crystals |
| Odor | Ammonia-like |
| Density | 1.39 g/cm3 |
| Melting point | 210 °C (410 °F; 483 K)[3] |
| Boiling point | 400 °C (752 °F; 673 K) |
| Reacts | |
| Solubility in liquid ammonia | 40 mg/L |
| Acidity (pKa) | 38 (conjugate acid)[2] |
| Structure[citation needed] | |
| orthorhombic | |
| Thermochemistry[citation needed] | |
Heat capacity (C)
|
66.15 J/(mol·K) |
Std molar
entropy (S |
76.9 J/(mol·K) |
Std enthalpy of
formation (ΔfH⦵298) |
−118.8 kJ/mol |
Gibbs free energy (ΔfG˚)
|
−59 kJ/mol |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H261, H314, H412 | |
| P223, P231+232, P260, P264, P273, P280, P301+330+331, P303+361+353, P304+340+310, P305+351+338+310, P335+334, P363, P370+378, P402+404, P405, P501 | |
| NFPA 704 (fire diamond) | |
| 450 °C (842 °F; 723 K)[4] | |
| Related compounds | |
Other anions
|
Sodium bis(trimethylsilyl)amide |
Other cations
|
|
Related compounds
|
Ammonia |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium amide, commonly called sodamide (systematic name sodium azanide), is the inorganic compound with the formula NaNH
2. It is a salt composed of the sodium cation and the azanide anion. It is a white solid which is dangerously reactive toward water, but commercial samples are typically gray due to the presence of small quantities of metallic iron from the manufacturing process. Such impurities do not usually affect the utility of the reagent. NaNH
2 conducts electricity in the fused state, its conductance being similar to that of NaOH in a similar state. NaNH
2 has been widely employed as a strong base in organic synthesis.
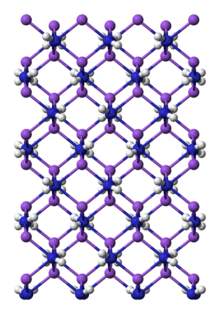
Preparation and structure
Sodium amide can be prepared by the reaction of sodium with ammonia gas,[5] but it is usually prepared by the reaction in liquid ammonia using iron(III) nitrate as a catalyst. The reaction is fastest at the boiling point of the ammonia (−33 °C (−27 °F; 240 K)). An electride, [Na(NH
3)
6]+
e−
, is formed as a reaction intermediate.[6]
- 2 Na + 2 NH
3 → 2 NaNH
2 + H
2
NaNH
2 is a salt-like material and as such, crystallizes as an infinite polymer.[7] The geometry about sodium is tetrahedral.[8] In ammonia, NaNH
2 forms conductive solutions, consistent with the presence of [Na(NH
3)
6]+
and NH−
2 ions.
Uses
Sodium amide is mainly used as a strong base in organic chemistry, often suspended (it is insoluble[9]) in liquid ammonia solution. One of the main advantages to the use of sodium amide is its relatively low nucleophilicity. In the industrial production of indigo, sodium amide is a component of the highly basic mixture that induces cyclisation of N-phenylglycine. The reaction produces ammonia, which is typically recycled.[10]

Dehydrohalogenation
Sodium amide is a standard base for dehydrohalogenations.[11] It induces the loss of two equivalents of hydrogen bromide from a vicinal dibromoalkane to give a carbon–carbon triple bond, as in a preparation of phenylacetylene.[12] Usually two equivalents of sodium amide yields the desired alkyne. Three equivalents are necessary in the preparation of a terminal alkynes because the terminal CH of the resulting alkyne protonates an equivalent amount of base.
300px
Hydrogen chloride and ethanol can also be eliminated in this way, as in the preparation of 1-ethoxy-1-butyne.[13][14][15][16][17]
500px
Cyclization reactions
Where there is no β-hydrogen to be eliminated, cyclic compounds may be formed, as in the preparation of methylenecyclopropane below.[18]
400px
Cyclopropenes, aziridines and cyclobutanes may be formed in a similar manner.[19][20][21]
Deprotonation of carbon and nitrogen acids
Carbon acids which can be deprotonated by sodium amide in liquid ammonia include:
- Terminal alkynes[22][23][24]
- Methyl ketones[25][26]
- Cyclohexanone[27]
- Phenylacetic acid and its derivatives[28][29][30]
- Diphenylmethane.[31]
Acetylacetone loses two protons to form a dianion.[32][33] Sodium amide will also deprotonate indole and piperidine.[34][35]
Related non-nucleophilic bases
It is however poorly soluble in solvents other than ammonia. Its use has been superseded by the related reagents sodium hydride, sodium bis(trimethylsilyl)amide (NaHMDS), and lithium diisopropylamide (LDA).
Other reactions
- Rearrangement with orthodeprotonation[36]
- Oxirane synthesis[37]
- Indole synthesis[38]
- Chichibabin reaction
Safety
Sodium amide is a common reagent with a long history of laboratory use.[11] It reacts violently on contact with water, producing ammonia and sodium hydroxide:
- NaNH
2 + H
2O → NH
3 + NaOH
When burned in oxygen, it will give oxides of sodium (which react with the produced water, giving sodium hydroxide) along with nitrogen oxides:
- 4 NaNH
2 + 5 O
2 → 4 NaOH + 4 NO + 2 H
2O - 4 NaNH
2 + 7 O
2 → 4 NaOH + 4 NO
2 + 2 H
2O
In the presence of limited quantities of air and moisture, such as in a poorly closed container, explosive mixtures of peroxides may form.[39] This is accompanied by a yellowing or browning of the solid. As such, sodium amide is to be stored in a tightly closed container, under an atmosphere of an inert gas. Sodium amide samples which are yellow or brown in color represent explosion risks.[40]
References
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "amides". doi:10.1351/goldbook.A00266
- ↑ Buncel, E.; Menon, B. (1977). "Carbanion mechanisms: VII. Metallation of hydrocarbon acids by potassium amide and potassium methylamide in tetrahydrofuran and the relative hydride acidities". Journal of Organometallic Chemistry 141 (1): 1–7. doi:10.1016/S0022-328X(00)90661-2.
- ↑ 3.0 3.1 Sigma-Aldrich Co., Sodium amide. Retrieved on 20 March 2026.
- ↑ 4.0 4.1 "SDS - Sodium amide" (pdf). ThermoFisher Scientific. 19 December 2025. pp. 3-5. https://www.fishersci.com/store/msds?partNumber=AC339240250&productDescription=SODIUM+AMIDE+25GR&vendorId=VN00032119&countryCode=US&language=en.
- ↑ Bergstrom, F. W. (1955). "Sodium amide". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=cv3p0778.; Collective Volume, 3, pp. 778
- ↑ Greenlee, K. W.; Henne, A. L.. "Sodium Amide". Inorganic Syntheses. 2. pp. 128–135. doi:10.1002/9780470132333.ch38. ISBN 9780470132333.
- ↑ Zalkin, A.; Templeton, D. H. (1956). "The Crystal Structure Of Sodium Amide". Journal of Physical Chemistry 60 (6): 821–823. doi:10.1021/j150540a042.
- ↑ Wells, A. F. (1984). Structural Inorganic Chemistry. Oxford: Clarendon Press. ISBN 0-19-855370-6.
- ↑ Audrieth, Ludwig F.; Kleinberg, Jacob (1953). Non-aqueous solvents. New York: John Wiley & Sons. p. 79. https://archive.org/details/cftri.2662nonaqueoussolven0000ludw/page/79/.
- ↑ Lange, L.; Treibel, W. (2005). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a24_267.
- ↑ 11.0 11.1 Belletire, John L.; Rauh, R. Jeffery (2001). "Sodium Amide". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rs041. ISBN 0-471-93623-5.
- ↑ Campbell, K. N.; Campbell, B. K. (1950). "Phenylacetylene". Organic Syntheses 30: 72. http://www.orgsyn.org/demo.aspx?prep=cv4p0763.; Collective Volume, 4, pp. 763
- ↑ Jones, E. R. H.; Eglinton, G.; Whiting, M. C.; Shaw, B. L. (1954). "Ethoxyacetylene". Organic Syntheses 34: 46. http://www.orgsyn.org/demo.aspx?prep=cv4p0404.; Collective Volume, 4, pp. 404
- ↑ Bou, A.; Pericàs, M. A.; Riera, A.; Serratosa, F. (1987). "Dialkoxyacetylenes: di-tert-butoxyethyne, a valuable synthetic intermediate". Organic Syntheses 65: 58. http://www.orgsyn.org/demo.aspx?prep=cv8p0161.; Collective Volume, 8, pp. 161
- ↑ Magriotis, P. A.; Brown, J. T. (1995). "Phenylthioacetylene". Organic Syntheses 72: 252. http://www.orgsyn.org/demo.aspx?prep=cv9p0656.; Collective Volume, 9, pp. 656
- ↑ Ashworth, P. J.; Mansfield, G. H.; Whiting, M. C. (1955). "2-Butyn-1-ol". Organic Syntheses 35: 20. http://www.orgsyn.org/demo.aspx?prep=cv4p0128.; Collective Volume, 4, pp. 128
- ↑ Newman, M. S.; Stalick, W. M. (1977). "1-Ethoxy-1-butyne". Organic Syntheses 57: 65. http://www.orgsyn.org/demo.aspx?prep=cv6p0564.; Collective Volume, 6, pp. 564
- ↑ Salaun, J. R.; Champion, J.; Conia, J. M. (1977). "Cyclobutanone from methylenecyclopropane via oxaspiropentane". Organic Syntheses 57: 36. http://www.orgsyn.org/demo.aspx?prep=cv6p0320.; Collective Volume, 6, pp. 320
- ↑ Nakamura, M.; Wang, X. Q.; Isaka, M.; Yamago, S.; Nakamura, E. (2003). "Synthesis and (3+2)-cycloaddition of a 2,2-dialkoxy-1-methylenecyclopropane: 6,6-dimethyl-1-methylene-4,8-dioxaspiro(2.5)octane and cis-5-(5,5-dimethyl-1,3-dioxan-2-ylidene)hexahydro-1(2H)-pentalen-2-one". Organic Syntheses 80: 144. http://www.orgsyn.org/demo.aspx?prep=v80p0144.
- ↑ Bottini, A. T.; Olsen, R. E. (1964). "N-Ethylallenimine". Organic Syntheses 44: 53. http://www.orgsyn.org/demo.aspx?prep=cv5p0541.; Collective Volume, 5, pp. 541
- ↑ Skorcz, J. A.; Kaminski, F. E. (1968). "1-Cyanobenzocyclobutene". Organic Syntheses 48: 55. http://www.orgsyn.org/demo.aspx?prep=cv5p0263.; Collective Volume, 5, pp. 263
- ↑ Saunders, J. H. (1949). "1-Ethynylcyclohexanol". Organic Syntheses 29: 47. http://www.orgsyn.org/demo.aspx?prep=cv3p0416.; Collective Volume, 3, pp. 416
- ↑ Peterson, P. E.; Dunham, M. (1977). "(Z)-4-Chloro-4-hexenyl trifluoroacetate". Organic Syntheses 57: 26. http://www.orgsyn.org/demo.aspx?prep=cv6p0273.; Collective Volume, 6, pp. 273
- ↑ Kauer, J. C.; Brown, M. (1962). "Tetrolic acid". Organic Syntheses 42: 97. http://www.orgsyn.org/demo.aspx?prep=cv5p1043.; Collective Volume, 5, pp. 1043
- ↑ Coffman, D. D. (1940). "Dimethylethynylcarbinol". Organic Syntheses 20: 40. http://www.orgsyn.org/demo.aspx?prep=cv3p0320.; Collective Volume, 3, pp. 320
- ↑ Hauser, C. R.; Adams, J. T.; Levine, R. (1948). "Diisovalerylmethane". Organic Syntheses 28: 44. http://www.orgsyn.org/demo.aspx?prep=cv3p0291.; Collective Volume, 3, pp. 291
- ↑ Vanderwerf, C. A.; Lemmerman, L. V. (1948). "2-Allylcyclohexanone". Organic Syntheses 28: 8. http://www.orgsyn.org/demo.aspx?prep=cv3p0044.; Collective Volume, 3, pp. 44
- ↑ Hauser, C. R.; Dunnavant, W. R. (1960). "α,β-Diphenylpropionic acid". Organic Syntheses 40: 38. http://www.orgsyn.org/demo.aspx?prep=cv5p0526.; Collective Volume, 5, pp. 526
- ↑ Kaiser, E. M.; Kenyon, W. G.; Hauser, C. R. (1967). "Ethyl 2,4-diphenylbutanoate". Organic Syntheses 47: 72. http://www.orgsyn.org/demo.aspx?prep=cv5p0559.; Collective Volume, 5, pp. 559
- ↑ Wawzonek, S.; Smolin, E. M. (1951). "α,β-Diphenylcinnamonitrile". Organic Syntheses 31: 52. http://www.orgsyn.org/demo.aspx?prep=cv4p0387.; Collective Volume, 4, pp. 387
- ↑ Murphy, W. S.; Hamrick, P. J.; Hauser, C. R. (1968). "1,1-Diphenylpentane". Organic Syntheses 48: 80. http://www.orgsyn.org/demo.aspx?prep=cv5p0523.; Collective Volume, 5, pp. 523
- ↑ Hampton, K. G.; Harris, T. M.; Hauser, C. R. (1971). "Phenylation of diphenyliodonium chloride: 1-phenyl-2,4-pentanedione". Organic Syntheses 51: 128. http://www.orgsyn.org/demo.aspx?prep=cv6p0928.; Collective Volume, 6, pp. 928
- ↑ Hampton, K. G.; Harris, T. M.; Hauser, C. R. (1967). "2,4-Nonanedione". Organic Syntheses 47: 92. http://www.orgsyn.org/demo.aspx?prep=cv5p0848.; Collective Volume, 5, pp. 848
- ↑ Potts, K. T.; Saxton, J. E. (1960). "1-Methylindole". Organic Syntheses 40: 68. http://www.orgsyn.org/demo.aspx?prep=cv5p0769.; Collective Volume, 5, pp. 769
- ↑ "N-β-Naphthylpiperidine". Organic Syntheses 40: 74. 1960. http://www.orgsyn.org/demo.aspx?prep=cv5p0816.; Collective Volume, 5, pp. 816
- ↑ Brazen, W. R.; Hauser, C. R. (1954). "2-Methylbenzyldimethylamine". Organic Syntheses 34: 61. http://www.orgsyn.org/demo.aspx?prep=cv4p0585.; Collective Volume, 4, pp. 585
- ↑ Allen, C. F. H.; VanAllan, J. (1944). "Phenylmethylglycidic ester". Organic Syntheses 24: 82. http://www.orgsyn.org/demo.aspx?prep=cv3p0727.; Collective Volume, 3, pp. 727
- ↑ Allen, C. F. H.; VanAllan, J. (1942). "2-Methylindole". Organic Syntheses 22: 94. http://www.orgsyn.org/demo.aspx?prep=cv3p0597.; Collective Volume, 3, pp. 597
- ↑ Clark, Donald E (2001). "Peroxides and peroxide-forming compounds". Chemical Health and Safety 8 (5): 12–22. doi:10.1016/S1074-9098(01)00247-7. ISSN 1074-9098.
- ↑ "Sodium amide Handling". Princeton. https://ehs.princeton.edu/laboratory-research/chemical-safety/chemical-specific-protocols/sodium-amide.
 |

