Chemistry:Copper aspirinate
From HandWiki
| |

| |
| File:CopperAspirinate.jpg | |
| Names | |
|---|---|
| IUPAC name
dicopper;tetrakis(2-acetyloxybenzoate)
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| Cu 2C 36H 28O 16 | |
| Molar mass | 843.696 g·mol−1 |
| Appearance | bright blue solid |
| Melting point | 255 °C (491 °F; 528 K)[1] (decomposes) |
| Structure[2] | |
| monoclinic | |
| P21/c | |
a = 8.208, b = 10.39, c = 21.56 α = 90°, β = 104.74°, γ = 90°
| |
Formula units (Z)
|
4 units per cell |
| Hazards | |
| NIOSH (US health exposure limits):[3] | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu) |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu) |
IDLH (Immediate danger)
|
TWA 100 mg/m3 (as Cu) |
| Related compounds | |
Other anions
|
|
Related compounds
|
Aspirin |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
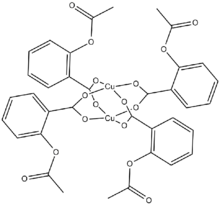
Copper(II) aspirinate is an aspirin chelate of copper(II) cations (Cu2+). It is a bright blue solid, forming monoclinic crystals.
Preparation
Copper aspirinate can be prepared treating a solution of aspirin and potassium bicarbonate with copper(II) sulfate solution.[1]
Heating an equimolar mixture of aspirin and copper salicylate tetrahydrate in 50% aqueous ethanol results in dark blue crystals.[4]
Research
Copper aspirinate has been investigated for its claimed anti-inflammatory,[5][6] antioxidative,[1] and antithrombotic[7] properties as well as a potential treatment for skin disorders.[8]
The use of copper aspirinate as a pigment in PVC and polystyrene has also been investigated.[9] [needs update]
External links
Footnotes
- ↑ 1.0 1.1 1.2 Fujimori, T.; Yamada, S.; Yasui, H.; Sakurai, H.; In, Y.; Ishida, T. (2005-12-01). "Orally active antioxidative copper(II) aspirinate: synthesis, structure characterization, superoxide scavenging activity, and in vitro and in vivo antioxidative evaluations" (in en). Journal of Biological Inorganic Chemistry 10 (8): 831–841. doi:10.1007/s00775-005-0031-3. ISSN 1432-1327. https://doi.org/10.1007/s00775-005-0031-3.
- ↑ Manojlović-Muir, L. (1973-10-15). "The crystal structure of copper(II) aspirinate" (in en). Acta Crystallographica Section B: Structural Crystallography and Crystal Chemistry 29 (10): 2033–2037. doi:10.1107/S0567740873006060. ISSN 0567-7408. https://journals.iucr.org/b/issues/1973/10/00/a10234/.
- ↑ NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/npg/npgd0150.html.
- ↑ Manojlović-Muir, Ljubica (1967-01-01). "Copper(II) aspirinate" (in en). Chemical Communications (20): 1057b–1058. doi:10.1039/C1967001057B. ISSN 0009-241X. https://pubs.rsc.org/en/content/articlelanding/1967/c1/c1967001057b.
- ↑ Zhiqiang, S.; Lei, W. Y.; Li, L.; Chen, Z. H.; Liu, W. P. (1998-12-01). "Coordination of copper with aspirin improves its anti-inflammatory activity" (in en). InflammoPharmacology 6 (4): 357–362. doi:10.1007/s10787-998-0018-0. ISSN 1568-5608. https://doi.org/10.1007/s10787-998-0018-0.
- ↑ Iqbal, Mohammad S.; Sher, Muhammad; Pervez, Humayun; Saeed, Maryiam (September 2008). "Pharmacokinetic Study of Copper (II) Acetylsalicylate". Biological Trace Element Research 124 (3): 283–288. doi:10.1007/s12011-008-8146-3. PMID 18478192. Bibcode: 2008BTER..124..283I.
- ↑ Liu, Weiping; Xiong, Huizhou; Yang Ling Li, Yikun; Shen, Zhiqiang; Chen, Zhihe (1998). "Potential Application of Copper Aspirinate in Preventing and Treating Thromboembolic Diseases". Metal-Based Drugs 5 (3): 123–126. doi:10.1155/MBD.1998.123. PMID 18475833.
- ↑ Duncan, Clare; White, Anthony R. (2012). "Copper complexes as therapeutic agents" (in en). Metallomics 4 (2): 134–135. doi:10.1039/C2MT00174H. ISSN 1756-5901. https://academic.oup.com/metallomics/article/4/2/127-138/6016075.
- ↑ Allan, JR; Renton, A; Smith, WE; Gerrard, DL; Birnie, J (1991). "A Study of the Performance of Bis(acetylsalicylate) Copper(II) and the Cobalt(II), Nickel(II) and Copper(II) Complexes of Pyridine-3,4-dicarboxylic Acid as Colouring Materials for Poly(vinyl chloride) and Polystyrene". Eur. Polym. J. 27 (7): 669–672. doi:10.1016/0014-3057(91)90155-H. Bibcode: 1991EurPJ..27..669A.
 |
