Chemistry:Mesuximide
From HandWiki
Short description: Chemical compound
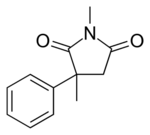 | |
| Clinical data | |
|---|---|
| Trade names | Celontin |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a682028 |
| Pregnancy category |
|
| Routes of administration | By mouth (capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic (demethylation and glucuronidation) |
| Metabolites | N-desmethylmethosuximide |
| Elimination half-life | 1.4–2.6 hours (mesuximide) 28–38 hours (active metabolite) |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C12H13NO2 |
| Molar mass | 203.241 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Mesuximide (or methsuximide, methosuximide) is a succinimide anticonvulsant medication. It is sold as a racemate by Pfizer under the tradenames Petinutin (Switzerland)[1] and Celontin (United States).[2] The therapeutic efficacy of methosuximide is largely due to its pharmacologically active metabolite, N-desmethylmethosuximide, which has a longer half-life and attains much higher plasma levels than its parent.[3]
Medical use
is indicated for the control of absence seizures that are refractory to other drugs.[2]
References
- ↑ Pfizer AG (2005). "Petinutin (Mésuximide)" (in fr). Official Pfizer AG Website. http://www.pfizer.ch/internet/fr/home/products/central_nervous_system/epilepsy/petinutin_mesuximid.html.
- ↑ 2.0 2.1 Pfizer Inc. (2008). "Celontin (methsuximide capsules, USP)". Official Pfizer Inc. Website. http://labeling.pfizer.com/ShowLabeling.aspx?id=555.
- ↑ "Plasma concentrations of phensuximide, methsuximide, and their metabolites in relation to clinical efficacy". Neurology 29 (11): 1509–13. November 1979. doi:10.1212/wnl.29.11.1509. PMID 116142.
 |
