Chemistry:Primidone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Lepsiral, Mysoline, Resimatil, others |
| Other names | desoxyphenobarbital, desoxyphenobarbitone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682023 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Anticonvulsant, barbiturate |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~100%[3] |
| Protein binding | 25%[3] |
| Metabolism | Liver |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
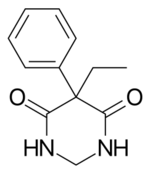
| Formula | C12H14N2O2 |
| Molar mass | 218.256 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Primidone, sold under various brand names, is a barbiturate medication that is used to treat partial and generalized seizures[4] and essential tremors.[5] It is taken by mouth.[4]
Its common side effects include sleepiness, poor coordination, nausea, and loss of appetite.[4] Severe side effects may include suicide and psychosis. [5][4] Use during pregnancy may result in harm to the fetus.[6] Primidone is an anticonvulsant of the barbiturate class;[4] however, its long-term effect in raising the seizure threshold is likely due to its active metabolite, phenobarbital.[7]
Primidone was approved for medical use in the United States in 1954.[4] It is available as a generic medication.[5] In 2020, it was the 269th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[8][9]
Medical uses
Epilepsy
It is licensed for generalized tonic-clonic and complex partial seizures in the United Kingdom.[10] In the United States, primidone is approved for adjunctive (in combination with other drugs) and monotherapy (by itself) use in generalized tonic-clonic seizures, simple partial seizures, complex partial seizures, and myoclonic seizures.[10] In juvenile myoclonic epilepsy, it is a second-line therapy, reserved for when the valproates or lamotrigine do not work and when the other second-line therapy, acetazolamide, does not work.[11]
Open-label case series have suggested that primidone is effective in the treatment of epilepsy.[12][13][14][15][16] Primidone has been compared to phenytoin,[17] phenobarbital,[17] mephobarbital, ethotoin, metharbital, and mephenytoin.[17] In adult comparison trials, primidone has been found to be just as effective.[17]
Essential tremor
Primidone is considered to be a first-line therapy for essential tremor, along with propranolol. In tremor amplitude reduction, it is just as effective as propranolol, reducing it by 50%. Both drugs are well studied for this condition, unlike other therapies, and are recommended for initial treatment. A low-dose therapy (250 mg/day) is just as good as a high-dose therapy (750 mg/day).[18]
Primidone is not the only anticonvulsant used for essential tremor; the others include topiramate and gabapentin. Other pharmacological agents include alprazolam, clonazepam, atenolol, sotalol, nadolol, clozapine, nimodipine, and botulinum toxin A. Many of these drugs were less effective than primidone. Only propranolol has been compared to primidone in a clinical trial.[18]
Psychiatric disorders
In 1965, Monroe and Wise reported using primidone along with a phenothiazine derivative antipsychotic and chlordiazepoxide in treatment-resistant psychosis.[19] What is known is that 10 years later, Monroe went on to publish the results of a meta-analysis of two controlled clinical trials on people displaying out-of-character and situationally inappropriate aggression, who had abnormal EEG readings, and who responded poorly to antipsychotics; one of the studies was specifically mentioned as involving psychosis patients. When they were given various anticonvulsants, not only did their EEGs improve, but so did the aggression.[20]
In March 1993, S.G. Hayes of the University of Southern California School of Medicine reported that 9 out of 27 people (33%) with either treatment-resistant depression or treatment-resistant bipolar disorder had a permanent positive response to primidone. A plurality of subjects was also given methylphenobarbital in addition to or instead of primidone.[21]
Adverse effects
Primidone can cause drowsiness, listlessness, ataxia, visual disturbances, nystagmus, headache, and dizziness. These side effects are the most common, occurring in more than 1% of users.[22] Transient nausea and vomiting are also common side effects.[23]

Dupuytren's contracture, a disease of the fasciae in the palm and fingers that permanently bends the fingers (usually the little and ring fingers) toward the palm, was first noted to be highly prevalent in epileptic people in 1941 by a Dr. Lund, 14 years before primidone was on the market. Lund also noted that it was equally prevalent in individuals with idiopathic and symptomatic epilepsy and that the severity of the epilepsy did not matter. Only one-quarter of the women were affected, though, vs. half of the men.[24] Critcheley et al., 35 years later, reported a correlation between how long a patient had had epilepsy and his or her chance of getting Dupuytren's contracture. They suspected that this was due to phenobarbital therapy, and that the phenobarbital was stimulating peripheral tissue growth factors.[25] Dupuytren's contracture is almost exclusively found in Caucasians, especially those of Viking descent, and highest rates are reported in northern Scotland, Norway , Iceland, and Australia . It has also been associated with alcoholism, heavy smoking, diabetes mellitus, physical trauma (either penetrating in nature or due to manual labor), tuberculosis, and HIV. People with rheumatoid arthritis are less likely to get this, and Drs. Hart and Hooper speculate that this is also true of gout due to the use of allopurinol. This is the only susceptibility factor that is generally agreed upon. Anticonvulsants do not seem to increase the incidence of Dupuytren's contracture in people of color.[24]
Primidone has other cardiovascular effects in beyond shortening the QT interval. Both phenobarbital and it are associated with elevated serum levels (both fasting and six hours after methionine loading) of homocysteine, an amino acid derived from methionine. This is almost certainly related to the low folate levels reported in primidone users. Elevated levels of homocysteine have been linked to coronary heart disease. In 1985, both drugs were also reported to increase serum levels of high-density lipoprotein cholesterol, total cholesterol, and apolipoproteins A and B.[26]
It was first reported to exacerbate hepatic porphyria in 1975. In 1981, phenobarbital, one of primidone's metabolites, was shown to only induced a significant porphyrin level at high concentrations in vitro.[27] It can also cause elevations in hepatic enzymes such as gamma-glutamyl transferase and alkaline phosphatase.[22]
Less than 1% of primidone users experience a rash. Compared to carbamazepine, lamotrigine, and phenytoin, this is very low. The rate is comparable to that of felbamate, vigabatrin, and topiramate.[28] Primidone also causes exfoliative dermatitis, Stevens–Johnson syndrome, and toxic epidermal necrolysis.[22]
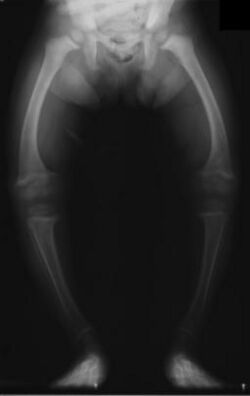
Primidone, along with phenytoin and phenobarbital, is one of the anticonvulsants most heavily associated with bone diseases such as osteoporosis, osteopenia (which can precede osteoporosis), osteomalacia, and fractures.[29][30][31] The populations usually said to be most at risk are institutionalized people, postmenopausal women, older men, people taking more than one anticonvulsant, and children, who are also at risk of rickets.[29] Bone demineralization is suggested to be most pronounced in young people (25–44 years of age),[30] and one 1987 study of institutionalized people found that the rate of osteomalacia in the ones taking anticonvulsants—one out of 19 individuals taking an anticonvulsant (vs. none among the 37 people taking none) —was similar to that expected in elderly people. The authors speculated that this was due to improvements in diet, sun exposure, and exercise in response to earlier findings, and/or that this was because it was sunnier in London than in the Northern European countries, which had earlier reported this effect.[31] In any case, the use of more than one anticonvulsant has been associated with an increased prevalence of bone disease in institutionalized epilepsy patients versus institutionalized people who did not have epilepsy. Likewise, postmenopausal women taking anticonvulsants have a greater risk of fracture than their drug-naive counterparts.[29]
Anticonvulsants affect the bones in many ways. They cause hypophosphatemia, hypocalcemia, low vitamin D levels, and increased parathyroid hormone. Anticonvulsants also contribute to the increased rate of fractures by causing somnolence, ataxia, and tremor, which would cause gait disturbance, further increasing the risk of fractures on top of the increase due to seizures and the restrictions on activity placed on epileptic people. Increased fracture rate has also been reported for carbamazepine, valproate, and clonazepam. The risk of fractures is higher for people taking enzyme-inducing anticonvulsants than for people taking enzyme-non-inducing anticonvulsants.[30] In addition to all of the above, primidone can cause arthralgia.[22]
Granulocytopenia, agranulocytosis, red-cell hypoplasia and aplasia, and megaloblastic anemia are rarely associated with the use of primidone.[32] Megaloblastic anemia is actually a group of related disorders with different causes that share morphological characteristics—enlarged red blood cells with abnormally high nuclear-cytoplasmic ratios resulting from delayed maturation of nuclei combined with normal maturation of cytoplasm, into abnormal megakaryocytes and sometimes hypersegmented neutrophils; regardless of etiology, all of the megaloblastic anemias involve impaired DNA replication.[33] The anticonvulsant users who get this also tend to eat monotonous diets devoid of fruits and vegetables.[34]
This antagonistic effect is not due to the inhibition of dihydrofolate reductase, the enzyme responsible for the reduction of dihydrofolic acid to tetrahydrofolic acid, but rather to defective folate metabolism.[35]
In addition to increasing the risk of megaloblastic anemia, primidone, like other older anticonvulsants, also increases the risk of neural tube defects,[36] and like other enzyme-inducing anticonvulsants, it increases the likelihood of cardiovascular defects, and cleft lip without cleft palate.[6] Epileptic women are generally advised to take folic acid,[36] but there is conflicting evidence regarding the effectiveness of vitamin supplementation in the prevention of such defects.[6][37]
Additionally, a coagulation defect resembling vitamin K deficiency has been observed in newborns of mothers taking primidone.[36] Because of this, primidone is a Category D medication.[38]
Primidone, like phenobarbital and the benzodiazepines, can also cause sedation in the newborn and also withdrawal within the first few days of life; phenobarbital is the most likely out of all of them to do that.[36]
In May 2005, Dr. M. Lopez-Gomez's team reported an association between the use of primidone and depression in epilepsy patients; this same study reported that inadequate seizure control, post-traumatic epilepsy, and polytherapy were also risk factors. Polytherapy was also associated with poor seizure control. Of all of the risk factors, use of primidone and inadequate seizure control were the greatest, with odds ratios of 4.089 and 3.084, respectively. They had been looking for factors associated with depression in epilepsy patients.[39] Schaffer et al. 1999 reported that one of their treatment failures, a 45-year-old woman taking 50 mg a day along with lithium 600 mg/day, clozapine 12.5 mg/day, trazodone 50 mg/day, and alprazolam 4 mg/day for three and a half months experienced auditory hallucinations that led to discontinuation of primidone.[40] It can also cause hyperactivity in children;[41] this most commonly occurs at low serum levels.[42] There is one case of an individual developing catatonic schizophrenia when her serum concentration of primidone went above normal.[43]
Primidone is one of the anticonvulsants associated with anticonvulsant hypersensitivity syndrome, with the others being carbamazepine, phenytoin, and phenobarbital. This syndrome consists of fever, rash, peripheral leukocytosis, lymphadenopathy, and occasionally hepatic necrosis.[44]
Hyperammonemic encephalopathy was reported by Katano Hiroyuki of the Nagoya City Higashi General Hospital in early 2002 in a patient who had been stable on primidone monotherapy for five years before undergoing surgery for astrocytoma, a type of brain tumor. Additionally, her phenobarbital levels were inexplicably elevated after surgery. This is much more common with the valproates than with any of the barbiturates.[45] A randomized, controlled trial w found that primidone was more likely to cause impotence than phenytoin, carbamazepine, or phenobarbital.[23] Like phenytoin, primidone is rarely associated with lymphadenopathy.[46] Primidone can also cause vomiting; this happens in 1.0–0.1% of users.[22]
Overdose
The most common symptoms of primidone overdose are coma with loss of deep tendon reflexes, and during the recovery period, if the patient survives, disorientation, dysarthria, nystagmus, and ataxia,[47] lethargy, somnolence, vomiting, nausea, and occasionally, focal neurological deficits which lessen over time.[48] Complete recovery comes within five to seven days of ingestion.[47] The symptoms of primidone poisoning have generally been attributed to its biotransformation to phenobarbital, but primidone has toxic effects independent of its metabolites in humans.[48] The massive crystalluria that sometimes occurs sets its symptom profile apart from that of phenobarbital.[47][49][50][51] The crystals are white,[48][50] needle-like,[49] shimmering, hexagonal plates consisting mainly of primidone.[48][50]
In the Netherlands alone, 34 cases of suspected primidone poisoning occurred between 1978 and 1982. Of these, primidone poisoning was much less common than phenobarbital poisoning; 27 of those adult cases were reported to the Dutch National Poison Control Center. Of these, one person taking it with phenytoin and phenobarbital died, 12 became drowsy, and four were comatose.[49]
Treatments for primidone overdose have included hemoperfusion with forced diuresis,[49] a combination of bemegride and amiphenazole;[52] and a combination of bemegride, spironolactone, caffeine, pentylenetetrazol, strophanthin, penicillin, and streptomycin.[53]
In the three adults who are reported to have succumbed, the doses were 20–30 g.[47][52][53] However, two adult survivors ingested 30 g[47] 25 g,[52] and 22.5 g.[48] One woman experienced symptoms of primidone intoxication after ingesting 750 mg of her roommate's primidone.[54]
Interactions
Taking primidone with monoamine oxidase inhibitors (MAOIs) such as isocarboxazid (Marplan), phenelzine (Nardil), procarbazine (Matulane), selegiline (Eldepryl), tranylcypromine (Parnate) or within two weeks of stopping any one of them may potentiate the effects of primidone or change one's seizure patterns.[55] Isoniazid, an antitubercular agent with MAOI properties, has been known to strongly inhibit the metabolism of primidone.[56]
Like many anticonvulsants, primidone interacts with other anticonvulsants. Clobazam decreases clearance of primidone,[57] Mesuximide increases plasma levels of phenobarbital in primidone users,[58] both primidone and phenobarbital accelerate the metabolism of carbamazepine via CYP3A4,[59] and lamotrigine's apparent clearance is increased by primidone.[60] In addition to being an inducer of CYP3A4, it is also an inducer of CYP1A2, which causes it to interact with substrates such as fluvoxamine, clozapine, olanzapine, and tricyclic antidepressants.[61] It also interacts with CYP2B6 substrates such as bupropion, efavirenz, promethazine, selegiline, and sertraline; CYP2C8 substrates such as amiodarone, paclitaxel, pioglitazone, repaglinide, and rosiglitazone; and CYP2C9 substrates such as bosentan, celecoxib, dapsone, fluoxetine, glimepiride, glipizide, losartan, montelukast, nateglinide, paclitaxel, phenytoin, sulfonamides, trimethoprim, warfarin, and zafirlukast. It also interacts with estrogens.[55]
Primidone and the other enzyme-inducing anticonvulsants can cut the half-life of antipyrine roughly in half (6.2 ± 1.9 h vs. 11.2 ± 4.2 h), and increases the clearance rate by almost 70%. Phenobarbital reduces the half-life to 4.8 ± 1.3 and increases the clearance by almost 109%.[62] It also interferes with the metabolism of dexamethasone, a synthetic steroid hormone, to the point where its withdrawal from the regimen of a 14-year-old living in the United Kingdom made her hypercortisolemic.[63] Tempelhoff and colleagues at the Washington University School of Medicine's Department of Anesthesiology reported in 1990 that primidone and other anticonvulsant drugs increase the amount of fentanyl needed during craniotomy based on the patient's heart rate.[64]
Mechanism of action
The exact mechanism of primidone's anticonvulsant action is still unknown after over 50 years.[65] It is believed to work via interactions with voltage-gated sodium channels that inhibit high-frequency repetitive firing of action potentials.[66] The effect of primidone in essential tremor is not mediated by phenylethylmalonamide (PEMA).[67] The major metabolite, phenobarbital, is also a potent anticonvulsant in its own right and likely contributes to primidone's effects in many forms of epilepsy. According to Brenner's Pharmacology, it also increases GABA-mediated chloride flux, thereby hyperpolarizing the membrane potential. Primidone was recently shown to directly inhibit the TRPM3 ion channel;[68] whether this effect contributes to its anticonvulsant effect is not known, but gain-of-function mutations in TRPM3 were shown to be associated with epilepsy and intellectual disability in 2021.[69]
Pharmacokinetics
Primidone converts to phenobarbital and PEMA;[70] it is still unknown which exact cytochrome P450 enzymes are responsible.[56] The phenobarbital, in turn, is metabolized to p-hydroxyphenobarbital.[71] The rate of primidone metabolism was greatly accelerated by phenobarbital pretreatment, moderately accelerated by primidone pretreatment, and reduced by PEMA pretreatment.[72] In 1983, a new minor metabolite, p-hydroxyprimidone, was discovered.[73]
Primidone, carbamazepine, phenobarbital, and phenytoin are among the most potent hepatic enzyme-inducing drugs in existence, which occurs at therapeutic doses. In fact, people taking these drugs have displayed the highest degree of hepatic-enzyme induction on record.[62] In addition to being an inducer of CYP3A4, it is also an inducer of CYP1A2, which causes it to interact with substrates such as fluvoxamine, clozapine, olanzapine, and tricyclic antidepressants, as well as potentially increasing the toxicity of tobacco products. Its metabolite, phenobarbital, is a substrate of CYP2C9,[61] CYP2B6,[74] CYP2C8, CYP2C19, CYP2A6, CYP3A5,[75] CYP1E1, and the CYP2E subfamily.[76] The gene expression of these isoenzymes is regulated by human pregnane receptor X (PXR) and constitutive androstane receptor (CAR). Phenobarbital induction of CYP2B6 is mediated by both.[75][77] Primidone does not activate PXR.[78]
The rate of metabolism of primidone into phenobarbital was inversely related to age; the highest rates were in the oldest patients (the maximum age being 55).[79] People aged 70–81, relative to people aged 18–26, have decreased renal clearance of primidone, phenobarbital, and PEMA, in ascending order of significance, and that there was a greater proportion of PEMA in the urine.[80] The clinical significance is unknown.
The percentage of primidone converted to phenobarbital has been estimated to be 5% in dogs and 15% in humans. Work done 12 years later found that the serum phenobarbital 0.111 mg/100 mL for every mg/kg of primidone ingested. Authors publishing a year earlier estimated that 24.5% of primidone was metabolized to phenobarbital, but the patient reported by Kappy and Buckley would have had a serum level of 44.4 mg/100 mL instead of 8.5 mg/100 mL if this were true for individuals who have ingested a large dose. The patient reported by Morley and Wynne would have had serum barbiturate levels of 50 mg/100 mL, which would have been fatal.[47]
History
Primidone is a congener of phenobarbital, where the carbonyl oxygen of the urea moiety is replaced by two hydrogen atoms.[81] The effectiveness of Primidone for epilepsy was first demonstrated in 1949 by Yule Bogue.[12] He found it to have a similar anticonvulsant effect, but more specific, i.e. with fewer associated sedative effects.[82]
It was brought to market a year later by the Imperial Chemical Industry, now known as AstraZeneca in the United Kingdom[52][83] and Germany.[53] In 1952, it was approved in the Netherlands.[49]
Also in 1952, Drs. Handley and Stewart demonstrated its effectiveness in the treatment of patients who failed to respond to other therapies; it was noted to be more effective in people with idiopathic generalized epilepsy than in people whose epilepsy had a known cause.[12] Dr. Whitty noted in 1953 that it benefitted patients with psychomotor epilepsy, who were often treatment-resistant. Toxic effects were reported to be mild.[13] That same year, it was approved in France.[84] Primidone was introduced in 1954 under the brandname Mysoline by Wyeth in the United States.[85]
Association with megaloblastic anemia
In 1954, Chalmers and Boheimer reported that the drug was associated with megaloblastic anemia.[86] Between 1954 and 1957, 21 cases of megaloblastic anemia associated with primidone and/or phenytoin were reported.[87] In most of these cases, the anemia was due to vitamin deficiencies - usually folic acid deficiency, in one case vitamin B12 deficiency,[86] and in one case vitamin C deficiency.[87] Some cases were associated with deficient diets - one patient ate mostly bread and butter,[86] another ate bread, buns, and hard candy, and another could rarely be persuaded to eat in the hospital.[87]
The idea that folic acid deficiency could cause megaloblastic anemia was not new. What was new was the idea that drugs could cause this in well-nourished people with no intestinal abnormalities.[86] In many cases, it was not clear which drug had caused it.[88] This might be related to the structural similarity between folic acid, phenytoin, phenobarbital, and primidone.[89] Folic acid had been found to alleviate the symptoms of megaloblastic anemia in the 1940s, not long after it was discovered, but the typical patient only made a full recovery—cessation of CNS and PNS symptoms as well as anemia—on B12 therapy.[90] Five years earlier, folic acid deficiency was linked to birth defects in rats.[91] Primidone was seen by some as too valuable to withhold based on the slight possibility of this rare side effect[86] and by others as dangerous enough to be withheld unless phenobarbital or some other barbiturate failed to work for this and other reasons (i.e., reports of permanent psychosis).[92]
Available forms
Primidone is available as a 250 mg/5mL suspension, and in the form of 50 mg, 125 mg, and 250 mg tablets. It is also available in a chewable tablet formulation in Canada.[93]
It is marketed as several different brands, including Mysoline (Canada,[94] Ireland,[95] Japan,[96] the United Kingdom,[97] the United States[94] and Turkey[98]), Prysoline (Israel, Rekah Pharmaceutical Products, Ltd.),[99] Apo-Primidone,[93][100] Liskantin (Germany, Desitin),[101] Resimatil (Germany, Sanofi-Synthélabo GmbH),[102] Mylepsinum (Germany, AWD.pharma GmbH & Co., KG).,[103] and Sertan (Hungary, 250 mg tablets, ICN Pharmaceuticals Inc.[1])
Veterinary uses
Primidone has veterinary uses, including the prevention of aggressive behavior and cannibalism in gilt pigs, and treatment of nervous disorders in dogs and other animals.[104][105]
References
- ↑ "Primidone (Mysoline) Use During Pregnancy". 18 February 2019. https://www.drugs.com/pregnancy/primidone.html.
- ↑ "Primidone SERB 50mg Tablets - Summary of Product Characteristics (SmPC)". 18 August 2014. https://www.medicines.org.uk/emc/product/2941/smpc.
- ↑ 3.0 3.1 "Antiepileptic Drugs: An Overview". eMedicine. eMedicine, Inc.. 2005. http://www.emedicine.com/neuro/topic692.htm.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 "Primidone Monograph for Professionals". American Society of Health-System Pharmacists. https://www.drugs.com/monograph/primidone.html.
- ↑ 5.0 5.1 5.2 British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 332. ISBN 9780857113382.
- ↑ 6.0 6.1 6.2 "Folic acid antagonists during pregnancy and the risk of birth defects". The New England Journal of Medicine 343 (22): 1608–1614. November 2000. doi:10.1056/NEJM200011303432204. PMID 11096168.
- ↑ Goodman and Gilman's The Pharmacological Basis of Therapeutics (7th ed.). New York: Macmillan. 1985.
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Primidone - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Primidone.
- ↑ 10.0 10.1 Acorus Therapeutics, Ltd. (2005). "Mysoline 250 mg Tablets". electronic Medicines Compendium. Datapharm Communications and the Association of the British Pharmaceutical Industry (ABPI). http://emc.medicines.org.uk/emc/assets/c/html/displaydoc.asp?documentid=16760.
- ↑ "Juvenile Myoclonic Epilepsy of Janz (JME)". The Childhood Seizure e-Book. Valhalla, New York. 200. http://www.pediatricneurology.com/new_page_1.htm. Retrieved 3 July 2005.
- ↑ 12.0 12.1 12.2 "DISCUSSION on recent advances in treatment". Proceedings of the Royal Society of Medicine 49 (8): 583–594. August 1956. doi:10.1177/003591575604900813. PMID 13359420.
- ↑ 13.0 13.1 "Value of primidone in epilepsy". British Medical Journal 2 (4835): 540–541. September 1953. doi:10.1136/bmj.2.4835.540. PMID 13082031.
- ↑ "Primidone (mysoline) in the treatment of epilepsy; results of treatment of 486 patients and review of the literature". The New England Journal of Medicine 254 (7): 327–329. February 1956. doi:10.1056/NEJM195602162540706. PMID 13288784.
- ↑ "Mysoline, a new anticonvulsant drug; its value in refractory cases of epilepsy". Canadian Medical Association Journal 68 (5): 464–467. May 1953. PMID 13042720.
- ↑ "Primidone therapy in refractory neonatal seizures". The Journal of Pediatrics 105 (4): 651–654. October 1984. doi:10.1016/S0022-3476(84)80442-4. PMID 6481545.
- ↑ 17.0 17.1 17.2 17.3 "Comparison of the effectiveness of phenobarital, mephobarbital, primidone, diphenylhydantoin, ethotoin, metharbital, and methylphenylethylhydantoin in motor seizures". Clinical Pharmacology and Therapeutics 3: 23–28. January–February 1962. doi:10.1002/cpt19623123. PMID 13902356.
- ↑ 18.0 18.1 "Practice Parameter: Therapies for essential tremor: Report of the Quality Standards Subcommittee of the American Academy of Neurology". Neurology 64 (12): 2008–20. 28 June 2005. doi:10.1212/01.WNL.0000163769.28552.CD. PMID 15972843.
- ↑ "Combined phenothiazine, chlordiazepoxide and primidone therapy for uncontrolled psychotic patients". The American Journal of Psychiatry 122 (6): 694–698. December 1965. doi:10.1176/ajp.122.6.694. PMID 5320821.
- ↑ "Anticonvulsants in the treatment of aggression". The Journal of Nervous and Mental Disease 160 (2–1): 119–126. February 1975. doi:10.1097/00005053-197502000-00006. PMID 1117287.
- ↑ "Barbiturate anticonvulsants in refractory affective disorders". Annals of Clinical Psychiatry 5 (1): 35–44. March 1993. doi:10.3109/10401239309148922. PMID 8348197.
- ↑ 22.0 22.1 22.2 22.3 22.4 "Summary of Product Characteristics". Official Acorus Therapeutics Site. Acorus Therapeutics. 1 June 2007. pp. 3–4. http://www.acorus-therapeutics.com/pdfs/products/MysolineSPCJune%202007.pdf. [|permanent dead link|dead link}}]
- ↑ 23.0 23.1 "Comparison of carbamazepine, phenobarbital, phenytoin, and primidone in partial and secondarily generalized tonic-clonic seizures". The New England Journal of Medicine 313 (3): 145–151. July 1985. doi:10.1056/NEJM198507183130303. PMID 3925335.
- ↑ 24.0 24.1 "Clinical associations of Dupuytren's disease". Postgraduate Medical Journal 81 (957): 425–428. July 2005. doi:10.1136/pgmj.2004.027425. PMID 15998816.
- ↑ "Dupuytren's disease in epilepsy: result of prolonged administration of anticonvulsants". Journal of Neurology, Neurosurgery, and Psychiatry 39 (5): 498–503. May 1976. doi:10.1136/jnnp.39.5.498. PMID 932769.
- ↑ "Elevated plasma concentrations of homocysteine in antiepileptic drug treatment". Epilepsia 40 (3): 345–350. March 1999. doi:10.1111/j.1528-1157.1999.tb00716.x. PMID 10080517.
- ↑ "Safety of anticonvulsants in hepatic porphyrias". Neurology 31 (4): 480–484. April 1981. doi:10.1212/wnl.31.4.480. PMID 7194443.
- ↑ "Comparison and predictors of rash associated with 15 antiepileptic drugs". Neurology 68 (20): 1701–9. 15 May 2007. doi:10.1212/01.wnl.0000261917.83337.db. PMID 17502552. http://www.neurology.org/cgi/content/full/68/20/1701. Retrieved 25 September 2007.
- ↑ 29.0 29.1 29.2 "Adverse effects of antiepileptic drugs on bone structure: epidemiology, mechanisms and therapeutic implications". CNS Drugs 15 (8): 633–642. 2001. doi:10.2165/00023210-200115080-00006. PMID 11524035.
- ↑ 30.0 30.1 30.2 "Antiepileptic drugs and bone metabolism". Nutrition & Metabolism 3 (36): 36. September 2006. doi:10.1186/1743-7075-3-36. PMID 16956398.
- ↑ 31.0 31.1 "Anticonvulsant drugs and bone disease in the elderly". Journal of the Royal Society of Medicine 80 (7): 425–427. July 1987. doi:10.1177/014107688708000710. PMID 3656313.
- ↑ "Mysoline". RxList. pp. 3. http://www.rxlist.com/cgi/generic/primid_ad.htm.
- ↑ "Megaloblastic Anemia". eMedicine. 2005. http://www.emedicine.com/MED/topic1420.htm.
- ↑ "Reversible absorptive defects in anticonvulsant megaloblastic anaemia". Journal of Clinical Pathology 18 (5): 593–598. September 1965. doi:10.1136/jcp.18.5.593. PMID 5835440.
- ↑ "Drug-induced anaemias". Drugs 11 (5): 394–404. 1976. doi:10.2165/00003495-197611050-00003. PMID 782836.
- ↑ 36.0 36.1 36.2 36.3 "Management of epilepsy in women". Postgraduate Medical Journal 81 (955): 278–285. May 2005. doi:10.1136/pgmj.2004.030221. PMID 15879038.
- ↑ "Effect of folic acid supplementation on congenital malformations due to anticonvulsive drugs". European Journal of Obstetrics, Gynecology, and Reproductive Biology 18 (4): 211–216. November 1984. doi:10.1016/0028-2243(84)90119-9. PMID 6519344.
- ↑ "Epilepsy in Pregnant Women". Current Treatment Options in Neurology 4 (1): 31–40. January 2002. doi:10.1007/s11940-002-0003-7. PMID 11734102.
- ↑ "Primidone is associated with interictal depression in patients with epilepsy". Epilepsy & Behavior 6 (3): 413–416. May 2005. doi:10.1016/j.yebeh.2005.01.016. PMID 15820351.
- ↑ "The use of primidone in the treatment of refractory bipolar disorder". Annals of Clinical Psychiatry 11 (2): 61–66. June 1999. doi:10.3109/10401239909147050. PMID 10440522.
- ↑ "Behavioural effects of anti-epileptic drugs". Developmental Medicine and Child Neurology 17 (5): 647–658. October 1975. doi:10.1111/j.1469-8749.1975.tb03536.x. PMID 241674.
- ↑ "Clinical side effects of phenobarbital, primidone, phenytoin, carbamazepine, and valproate during monotherapy in children". Epilepsia 29 (6): 794–804. November–December 1988. doi:10.1111/j.1528-1157.1988.tb04237.x. PMID 3142761.
- ↑ "Primidone-induced catatonic schizophrenia". Drug Intelligence & Clinical Pharmacy 17 (7–8): 551–552. July–August 1983. doi:10.1177/106002808301700715. PMID 6872851.
- ↑ "Antiepileptic drug hypersensitivity syndrome". Epilepsia 39 (Suppl 7): S3–S7. 1998. doi:10.1111/j.1528-1157.1998.tb01678.x. PMID 9798755.
- ↑ "Primidone-induced hyperammonemic encephalopathy in a patient with cerebral astrocytoma". Journal of Clinical Neuroscience 9 (1): 79–81. January 2002. doi:10.1054/jocn.2001.1011. PMID 11749025.
- ↑ "Lymphadenopathy and megaloblastic anaemia in patient receiving primidone". British Medical Journal 1 (5534): 215–217. January 1967. doi:10.1136/bmj.1.5534.215. PMID 4959849.
- ↑ 47.0 47.1 47.2 47.3 47.4 47.5 "Primidone intoxication in a child". Archives of Disease in Childhood 44 (234): 282–284. April 1969. doi:10.1136/adc.44.234.282. PMID 5779436.
- ↑ 48.0 48.1 48.2 48.3 48.4 "Acute primidone intoxication". Archives of Neurology 30 (3): 255–258. March 1974. doi:10.1001/archneur.1974.00490330063011. PMID 4812959.
- ↑ 49.0 49.1 49.2 49.3 49.4 "Coma and crystalluria: a massive primidone intoxication treated with haemoperfusion". Journal of Toxicology. Clinical Toxicology 20 (4): 307–318. June 1983. doi:10.3109/15563658308990598. PMID 6655772.
- ↑ 50.0 50.1 50.2 "Chemical analysis of massive crystalluria following primidone overdose". American Journal of Clinical Pathology 58 (5): 583–589. November 1972. doi:10.1093/ajcp/58.5.583. PMID 4642162.
- ↑ "Primidone intoxication and massive crystalluria". Clinical Pediatrics 19 (10): 706–707. October 1980. doi:10.1177/000992288001901015. PMID 7408374.
- ↑ 52.0 52.1 52.2 52.3 "Treatment of acute primidone poisoning with bemegride and amiphenazole". British Medical Journal 2 (5042): 451–452. August 1957. doi:10.1136/bmj.2.5042.451. PMID 13446511.
- ↑ 53.0 53.1 53.2 "[Fatal poisoning (suicide) with mysoline and phenobarbital]" (in de). Archiv für Toxikologie 18 (4): 213–233. January 1960. doi:10.1007/BF00577226. PMID 13698457.
- ↑ "An unusual case of primidone intoxication". Diseases of the Nervous System 27 (10): 660–661. October 1966. PMID 5919666.
- ↑ 55.0 55.1 "Primidone". The Merck Manual's Online Medical Library. Lexi-Comp. http://www.merck.com/mmpe/lexicomp/primidone.html.
- ↑ 56.0 56.1 "Inhibition of cytochrome P450 (CYP450) isoforms by isoniazid: potent inhibition of CYP2C19 and CYP3A". Antimicrobial Agents and Chemotherapy 45 (2): 382–392. February 2001. doi:10.1128/AAC.45.2.382-392.2001. PMID 11158730.
- ↑ "Interactions of clobazam with conventional antiepileptics in children". Journal of Child Neurology 12 (3): 208–213. April 1997. doi:10.1177/088307389701200311. PMID 9130097.
- ↑ "Methsuximide for complex partial seizures: efficacy, toxicity, clinical pharmacology, and drug interactions". Neurology 33 (4): 414–418. April 1983. doi:10.1212/WNL.33.4.414. PMID 6403891.
- ↑ "Clinically significant pharmacokinetic drug interactions with carbamazepine. An update". Clinical Pharmacokinetics 31 (3): 198–214. September 1996. doi:10.2165/00003088-199631030-00004. PMID 8877250.
- ↑ GlaxoSmithKline (2005). "LAMICTAL Prescribing Information". http://us.gsk.com/products/assets/us_lamictal.pdf.
- ↑ 61.0 61.1 "Clinical significance of pharmacokinetic interactions between antiepileptic and psychotropic drugs". Epilepsia 43 (Suppl 2): 37–44. February 2002. doi:10.1046/j.1528-1157.2002.043s2037.x. PMID 11903482.
- ↑ 62.0 62.1 "A comparative study of the relative enzyme inducing properties of anticonvulsant drugs in epileptic patients". British Journal of Clinical Pharmacology 18 (3): 401–410. September 1984. doi:10.1111/j.1365-2125.2004.02311.x. PMID 6435654.
- ↑ "Loss of therapeutic control in congenital adrenal hyperplasia due to interaction between dexamethasone and primidone". Acta Paediatrica Scandinavica 80 (1): 120–124. January 1991. doi:10.1111/j.1651-2227.1991.tb11744.x. PMID 2028784.
- ↑ "Anticonvulsant therapy increases fentanyl requirements during anaesthesia for craniotomy". Canadian Journal of Anaesthesia 37 (3): 327–332. April 1990. doi:10.1007/BF03005584. PMID 2108815.
- ↑ "Mysoline: Clinical Pharmacology". RxList. http://www.rxlist.com/cgi/generic/primid_cp.htm.
- ↑ "Antiepileptic drug mechanisms of action". Epilepsia 36 (Suppl 2): S2-12. 1995. doi:10.1111/j.1528-1157.1995.tb05996.x. PMID 8784210.
- ↑ "Phenylethylmalonamide in essential tremor. A double-blind controlled study". Journal of Neurology, Neurosurgery, and Psychiatry 44 (10): 932–934. October 1981. doi:10.1136/jnnp.44.10.932. PMID 7031184.
- ↑ "Primidone inhibits TRPM3 and attenuates thermal nociception in vivo". Pain 158 (5): 856–867. May 2017. doi:10.1097/j.pain.0000000000000846. PMID 28106668.
- ↑ "The newest TRP channelopathy: Gain of function TRPM3 mutations cause epilepsy and intellectual disability". Channels 15 (1): 386–397. December 2021. doi:10.1080/19336950.2021.1908781. PMID 33853504.
- ↑ "Interindividual variability in the metabolism of anti-epileptic drugs and its clinical application". Interindividual Variability in Human Drug Metabolism. CRC Press. 1 July 2001. pp. 168. ISBN 978-0-7484-0864-1.
- ↑ "Microassay for primidone and its metabolites phenylethylmalondiamide, phenobarbital and p-hydroxyphenobarbital in human serum, saliva, breast milk and tissues by gas chromatography--mass spectrometry using selected ion monitoring". Journal of Chromatography 182 (1): 71–79. April 1980. doi:10.1016/S0378-4347(00)81652-7. PMID 7380904. http://torpedo.nrl.navy.mil/tu/ps/openurl.html?issn=0021-9673&volume=182&issue=1&spage=71. Retrieved 16 October 2005.
- ↑ "Study of the hepatic metabolism of primidone by improved methodology". The Journal of Pharmacology and Experimental Therapeutics 194 (1): 117–125. July 1975. PMID 1151744.
- ↑ "Identification of p-hydroxyprimidone as a minor metabolite of primidone in rat and man". Drug Metabolism and Disposition 11 (6): 607–610. November–December 1983. PMID 6140148. http://dmd.aspetjournals.org/cgi/reprint/11/6/607. Retrieved 3 August 2005.
- ↑ "Phenobarbital increases monkey in vivo nicotine disposition and induces liver and brain CYP2B6 protein". British Journal of Pharmacology 148 (6): 786–794. July 2006. doi:10.1038/sj.bjp.0706787. PMID 16751792.
- ↑ 75.0 75.1 "Broad but distinct role of pregnane x receptor on the expression of individual cytochrome p450s in human hepatocytes". Drug Metabolism and Pharmacokinetics 22 (4): 276–286. August 2007. doi:10.2133/dmpk.22.276. PMID 17827782. http://www.jstage.jst.go.jp/article/dmpk/22/4/276/_pdf. Retrieved 7 October 2007.
- ↑ "Effects of prototypical microsomal enzyme inducers on cytochrome P450 expression in cultured human hepatocytes". Drug Metabolism and Disposition 31 (4): 421–431. April 2003. doi:10.1124/dmd.31.4.421. PMID 12642468.
- ↑ "DHA down-regulates phenobarbital-induced cytochrome P450 2B1 gene expression in rat primary hepatocytes by attenuating CAR translocation". Toxicology and Applied Pharmacology 225 (3): 329–336. December 2007. doi:10.1016/j.taap.2007.08.009. PMID 17904175.
- ↑ "Key structural features of ligands for activation of human pregnane X receptor". Drug Metabolism and Disposition 32 (4): 468–472. April 2004. doi:10.1124/dmd.32.4.468. PMID 15039302.
- ↑ "Plasma levels of primidone and its metabolite phenobarbital: effect of age and associated therapy". Therapeutic Drug Monitoring 5 (1): 73–9. 1983. doi:10.1097/00007691-198303000-00006. PMID 6845402.
- ↑ "The disposition of primidone in elderly patients". British Journal of Clinical Pharmacology 30 (4): 607–11. 1990. doi:10.1111/j.1365-2125.1990.tb03820.x. PMID 2291873.
- ↑ Goodman and Gilman's The Pharmacological Basis of Therapeutics. 1964. p. 226.
- ↑ "The evaluation of "mysoline," a new anticonvulsant drug". British Journal of Pharmacology and Chemotherapy 8 (2): 230–236. June 1953. doi:10.1111/j.1476-5381.1953.tb00784.x. PMID 13066728.
- ↑ "Acute primidone poisoning in a child". British Medical Journal 1 (5010): 90. January 1957. doi:10.1136/bmj.1.5010.90. PMID 13383203.
- ↑ "Clinical experience with new antiepileptic drugs: antiepileptic drugs in Europe". Epilepsia 40 (Suppl 6): S3–8. June 1999. doi:10.1111/j.1528-1157.1999.tb00925.x. PMID 10530675.
- ↑ Wyeth. "Wyeth Timeline". About Wyeth. http://www.wyeth.com/aboutwyeth/history.
- ↑ 86.0 86.1 86.2 86.3 86.4 "Megaloblastic anemia following the use of primidone". Blood 12 (2): 183–188. February 1957. doi:10.1182/blood.V12.2.183.183. PMID 13403983.
- ↑ 87.0 87.1 87.2 "Megaloblastic anaemia and vitamin-B12 deficiency after anticonvulsant therapy; report of two cases". British Medical Journal 2 (5051): 974–976. October 1957. doi:10.1136/bmj.2.2689.97. PMID 13472024.
- ↑ "Observations on megaloblastic anaemias after primidone". British Medical Journal 1 (4974): 1021–1023. May 1956. doi:10.1136/bmj.1.4974.1021. PMID 13304415.
- ↑ "Megaloblastic anaemia occurring during primidone therapy". British Medical Journal 1 (4959): 146–147. January 1956. doi:10.1136/bmj.1.4959.146. PMID 13276653.
- ↑ "Folic acid in the treatment of pernicious anemia". Blood 2 (1): 50–62. January 1947. doi:10.1182/blood.V2.1.50.50. PMID 20278334.
- ↑ "Production of multiple congenital abnormalities in young by maternal pteroylglutamic acid deficiency during gestation". The Journal of Nutrition 48 (1): 61–79. September 1952. doi:10.1093/jn/48.1.61. PMID 13000492.
- ↑ "Drugs used in the management of epilepsy". Proceedings of the Royal Society of Medicine 50 (8): 611–615. August 1957. PMID 13465742.
- ↑ 93.0 93.1 "Mysoline". Epilepsy.com. Epilepsy Therapy Development Project. February 2004. http://www.epilepsy.com/medications/b_mysoline_intro.html.
- ↑ 94.0 94.1 "Valeant Pharmaceuticals International: Products". http://www.valeant.com/products/index.jspf.
- ↑ "Service List". http://www.acorus-therapeutics.com/index_Page910.htm.
- ↑ Dainippon Sumitomo Pharma (2005). "Primidone 250 mg Tablets & Primidone 99.5% Powder". http://med.ds-pharma.co.jp/medicine/ryouiki/seishin/doc/primidone_tabfgr_tenpu.pdf.[yes|permanent dead link|dead link}}]
- ↑ "Acorus Therapeutics Ltd. - Ordering - UK". acorus-therapeutics.com. Acorus Therapeutics. http://www.acorus-therapeutics.com/index_orderinguk.htm.
- ↑ "Emre Ecza - History". https://www.emreecza.com/en/tarihce.php. "MYSOLINE tablet ( primidone ) is a registered product of our company in Turkey..."
- ↑ "Prysoline Tablets". The Israel Drug Registry. The State of Israel. 2005. http://www.health.gov.il/units/pharmacy/trufot/PerutTrufa.asp?Reg_Number=024%2079%2021145%2000.
- ↑ "APO-PRIMIDONE". Apotex. 10 January 2007. http://www.epilepsy.com/medications/b_mysoline_intro.html.
- ↑ "Liskantin". Desitin. http://www.desitin.de/index.php/article/detail/766.
- ↑ "Resimatil Tabletten". Deutsche Krankenversicherung AG. http://www.dkv.com/content_appl.phtml?content=http://dkv.netdoktor.de/redesign/medikamente/100001437.htm.[yes|permanent dead link|dead link}}]
- ↑ "Mylepsinum Tabletten". Deutsche Krankenversicherung AG. http://www.dkv.com/content_appl.phtml?content=http://dkv.netdoktor.de/redesign/medikamente/100001217.htm.[yes|permanent dead link|dead link}}]
- ↑ National Office of Animal Health. "Compendium of Veterinary Medicine". http://www.noahcompendium.co.uk/Schering-Plough_Animal_Health/Mysoline_Veterinary_Tablets/-34475.html.
- ↑ The Pig Site. "Savaging of Piglets". http://www.thepigsite.com/pighealth/article/260/savaging-of-piglets-cannibalism.
Further reading
- "Toxicology and Carcinogenesis Studies of Primidone in F344/N Rats and B6C3F1 Mice (Feed Studies)". September 2000. https://ntp.niehs.nih.gov/ntp/htdocs/lt_rpts/tr476.pdf.
- Lay summary in: "Abstract for TR-476". September 2000. https://ntp.niehs.nih.gov/go/tr476abs.
External links
- "Testing Status of Primidone (primaclone) 10270-A". https://ntp.niehs.nih.gov/whatwestudy/testpgm/status/ts-10270-a.html.
 |
