Chemistry:Gabapentin
Gabapentin, sold under the brand name Neurontin among others, is an anticonvulsant medication used to treat neuropathic pain (postherpetic neuralgia) and partial seizures of epilepsy.[1][2] Gabapentin is a central nervous system (CNS) depressant and derivative of GABA.[2] It is used for the treatment of neuropathic pain caused by diabetic neuropathy, postherpetic neuralgia, and central pain.[3] It is moderately effective: about 30–40% of those given gabapentin for diabetic neuropathy or postherpetic neuralgia have a meaningful benefit.[4]
Gabapentin acts by decreasing activity of the α2δ-1 protein, coded by the CACNA2D1 gene, first known as an auxiliary subunit of voltage-gated calcium channels.[5][6][7] By binding to α2δ-1, gabapentin reduces the release of excitatory neurotransmitters (primarily glutamate) and as a result, reduces excess excitation of neuronal networks in the spinal cord and brain. Sleepiness and dizziness are the most common side effects. Serious side effects include respiratory depression and allergic reactions.[2] On December 16, 2008, the FDA issued gabapentin a class warning for an increased risk of suicide.[2] Approximately two years after this pronouncement, a pharmacoepidemiologic study was conducted that showed there was no outstanding difference in suicide attempt rates between pre and post gabapentin prescription groups.[8]
Gabapentin was first approved for use in the United Kingdom in 1993.[9] It has been available as a generic medication in the United States since 2004.[10] It is the first of several other drugs that are similar in structure and mechanism, called gabapentinoids. In 2023, it was the ninth most commonly prescribed medication in the United States, with more than 45 million prescriptions.[11][12] During the 1990s, Parke-Davis, a subsidiary of Pfizer, used several illegal techniques to encourage physicians in the United States to prescribe gabapentin for unapproved uses.[13] They have paid out millions of dollars to settle lawsuits regarding these activities.[14]
Medical uses
In the US, gabapentin is indicated for the treatment of postherpetic neuralgia; and the adjunctive therapy in the treatment of partial onset seizures without secondary generalization, in people with epilepsy.[2]
Gabapentin is recommended for use in focal seizures and neuropathic pain.[2][1] Gabapentin is prescribed off-label in the US and the UK,[15][16] for example, for the treatment of non-neuropathic pain,[15] anxiety disorders, sleep problems and bipolar disorder.[17] In recent years, gabapentin has seen increased use, particularly in the elderly.[18] There is concern regarding gabapentin's off-label use due to the lack of strong scientific evidence for its efficacy in multiple conditions, its proven side effects and its potential for misuse and physical/psychological dependency.[19][20][21] Some harms, including nervous system harms, have been underreported in published trials of gabapentin, potentially resulting in the underestimation of harms in guidelines for the use of gabapentin.[22]
Seizures
Gabapentin is approved for the treatment of focal seizures;[23] however, it is not effective for generalized epilepsy.[24]
Neuropathic pain
Gabapentin is recommended as a first-line treatment for chronic neuropathic pain by various medical authorities.[1][3][25][26] This is a general recommendation applicable to all neuropathic pain syndromes except for trigeminal neuralgia, where it may be used as a second- or third-line agent.[3][26]
Regarding the specific diagnoses, a systematic review has found evidence for gabapentin to provide pain relief for some people with postherpetic neuralgia and diabetic neuropathy.[4] Gabapentin is approved for the former indication in the US.[2] In addition to these two neuropathies, European Federation of Neurological Societies guideline notes gabapentin effectiveness for central pain.[3] A combination of gabapentin with an opioid or nortriptyline may work better than either drug alone.[3][26]
Evidence finds little or no benefit and significant risk in those with chronic low back pain or sciatica.[27][28] Gabapentin is not effective in HIV-associated sensory neuropathy[29] and neuropathic pain due to cancer.[30]
Anxiety
There is a small amount of research on the use of gabapentin for the treatment of anxiety disorders.[31][32]
Gabapentin is effective for the long-term treatment of social anxiety disorder and in reducing preoperative anxiety.[19][20]
In a controlled trial of breast cancer survivors with anxiety,[32] and a trial for social phobia,[31] gabapentin significantly reduced anxiety levels.
For panic disorder, gabapentin has produced mixed results.[32][31][20]
Sleep
Gabapentin is effective in treating sleep disorders such as insomnia and restless legs syndrome that are the result of an underlying illness, but comes with some risk of discontinuation and withdrawal symptoms after prolonged use at higher doses.[33]
Gabapentin enhances slow-wave sleep in people with primary insomnia. It also improves sleep quality by elevating sleep efficiency and decreasing spontaneous arousal.[34]
Drug dependence
Gabapentin is moderately effective in reducing the symptoms of alcohol withdrawal and associated craving.[35][36][37] The evidence in favor of gabapentin is weak in the treatment of alcoholism: it does not contribute to the achievement of abstinence, and the data on the relapse of heavy drinking and percent of days abstinent do not robustly favor gabapentin; it only decreases the percent days of heavy drinking.[38]
Gabapentin is ineffective in cocaine dependence and methamphetamine use,[39] and it does not increase the rate of smoking cessation.[40] While some studies indicate that gabapentin does not significantly reduce the symptoms of opiate withdrawal, there is increasing evidence that gabapentinoids are effective in controlling some of the symptoms during opiate detoxification. A clinical study in Iran, where heroin dependence is a significant social and public health problem, showed gabapentin produced positive results during an inpatient therapy program, particularly by reducing opioid-induced hyperalgesia and drug craving.[41][39] There is insufficient evidence for its use in cannabis dependence.[42][needs update]
Other
Gabapentin is recommended as a first-line treatment of the acquired pendular nystagmus, torsional nystagmus, and infantile nystagmus; however, it does not work in periodic alternating nystagmus.[43][44][45]
Gabapentin decreases the frequency of hot flashes in both menopausal women and people with breast cancer. However, antidepressants have similar efficacy, and treatment with estrogen more effectively prevents hot flashes.[46]
Gabapentin reduces spasticity in multiple sclerosis and is prescribed as one of the first-line options.[47] It is an established treatment of restless legs syndrome.[48] Gabapentin alleviates itching in kidney failure (uremic pruritus)[49][50] and itching of other causes.[51] It may be an option in essential or orthostatic tremor.[52][53][54]
Gabapentin does not appear to provide benefit for bipolar disorder,[20][36][55] complex regional pain syndrome,[56] post-surgical pain,[57] or tinnitus,[58] or prevent episodic migraine in adults.[59]
Gabapentin is prescribed off-label as a anxiolytic or anti-anxiety medication, although not as effective as pregabalin in this regard, especially for generalized anxiety disorder, as gabapentin is more effective for social anxiety disorder.[19][20]
Contraindications
Gabapentin should be used carefully and at lower doses in people with kidney problems due to possible accumulation and toxicity. It is unclear if it is safe during pregnancy or breastfeeding.[2]
Side effects
In a systematic review analysing data from five cohort studies having 1,085,488 patients, use of gabapentinoids (gabapentin and pregabalin) was associated with an increased risk of thrombotic events (deep venous thrombosis and pulmonary thrombo-embolism) as early as three months of use, and with increased risk of cardiovascular events on prolonged use of more than a year duration. Heart failure was not increased with the use of gabapentinoids.[60]

Dizziness and somnolence are the most frequent side effects.[2] Fatigue, ataxia, peripheral edema (swelling of extremities), and nystagmus are also common.[2] A 2017 meta-analysis found that gabapentin also increased the risk of difficulties in mentation and visual disturbances as compared to a placebo.[27] Gabapentin is associated with a weight gain of 2.2 kg (4.9 lb) after 1.5 months of use.[61] Case studies indicate that it may cause anorgasmia and erectile dysfunction,[62] as well as myoclonus[63][64] that disappear after discontinuing gabapentin or replacing it with other medication. Fever, swollen glands that do not go away, eyes or skin turning yellow, unusual bruises or bleeding, unexpected muscle pain or weakness, rash, long-lasting stomach pain which may indicate an inflamed pancreas, hallucinations, anaphylaxis, respiratory depression, and increased suicidal ideation are rare but serious side effects.[65]
Suicide
The gabapentin prescribing information contains a warning of an increased risk of suicidal thoughts and behaviors.[2] The warning is based on a meta-analysis of all approved antiepileptic drugs in 2008, and not with gabapentin alone.[66] According to an experimental meta-analysis of insurance claims databases, gabapentin use is associated with an approximately 40% increased risk of suicide, suicide attempt, and violent death as compared with a reference anticonvulsant drug topiramate. The risk is increased for people with bipolar disorder or epilepsy.[66] Another study has shown an approximately doubled rate of suicide attempts and self-harm in people with bipolar disorder who are taking gabapentin versus those taking lithium.[67] A large Swedish study suggests that gabapentinoids are associated with an increased risk of suicidal behaviour, unintentional overdoses, head/body injuries, and road traffic incidents and offences.[68] On the other hand, a study published by the Harvard Data Science Review found that gabapentin was associated with a significantly reduced rate of suicide.[69] In addition, a 2010 study published by the Pharmacoepidemiology and Drug Safety journal concluded that although there was no significant suicide rate decrease between the pre-prescription window and the post-prescription window for the non-psychiatric population, significant reductions in suicides rates were observed in psychiatric persons.[8]
Respiratory depression
Serious breathing suppression, potentially fatal, may occur when gabapentin is taken together with opioids, benzodiazepines, or other depressants, or by people with underlying lung problems such as COPD.[70] Gabapentin and opioids are commonly prescribed or abused together, and research indicates that the breathing suppression they cause is additive. For example, gabapentin use before joint replacement or laparoscopic surgery increased the risk of respiratory depression by 30–60%.[70] A Canadian study showed that use of gabapentin and other gabapentinoids, whether for epilepsy, neuropathic pain or other chronic pain was associated with a 35–58% increased risk for severe exacerbation of pre-existing chronic obstructive pulmonary disease.[71]
Withdrawal and dependence
Withdrawal symptoms typically occur 1–2 days after abruptly stopping gabapentin (almost unambiguously due to extended use and during a very short-term rebound phenomenon) — similar to, albeit less intense than most benzodiazepines.[72] Agitation, confusion and disorientation are the most frequently reported, followed by gastrointestinal complaints and sweating, and more rare tremor, tachycardia, hypertension and insomnia.[72] In some cases, users experience withdrawal seizures after chronic or semi-chronic use in the absence of periodic cycles or breaks during repeating and consecutive use.[73] All these symptoms subside when gabapentin is re-instated[72] or tapered off gradually at an appropriate rate. On its own, gabapentin appears not to have a substantial addictive power. In human and animal experiments, it shows limited to no rewarding effects. The vast majority of people abusing gabapentin are current or former abusers of opioids or sedatives.[73] In these persons, gabapentin can boost the opioid "high" as well as decrease commonly experienced opioid-withdrawal symptoms such as anxiety.[74]
Psychiatric and behavioral adverse effects
Gabapentin is sometimes recognized to cause a range of psychiatric and behavioral adverse effects that extend beyond its more common neurological side effects. Systematic reviews have documented atypical manifestations such as aggression, agitation, irritability, mood instability, and suicidal ideation, with some cases noting the emergence of mania, hallucinations, and psychosis, particularly in pediatric populations and individuals with preexisting psychiatric conditions.[75][76][77]
Large cohort studies and post-marketing surveillance indicate that neuropsychiatric symptoms—including confusion, depression, and behavioral disturbances—can occur in up to 29% of gabapentin users. Most reactions are mild to moderate and often dose-dependent.[78] There is also evidence associating gabapentin with an increased risk of suicidal behavior, especially in younger patients, and rare reports of violent or aggressive behavior. Causality is difficult to establish, and such events remain uncommon.[79][80]
Overdose
Through excessive ingestion, accidental or otherwise, persons may experience overdose symptoms including drowsiness, sedation, blurred vision, slurred speech, somnolence, uncontrollable jerking motions, and anxiety. A very high amount taken is associated with breathing suppression, coma, and possibly death, particularly if combined with alcohol or opioids.[73][81]
Pharmacology

Animal models
Gabapentin prevents seizures in a dose-related manner in several laboratory animal models.[82] These models include spinal extensor seizures from low-intensity electroshock to the forebrain in mice, maximal electroshock in rats, spinal extensor seizures in DBA/2 mice with a genetic sensitivity to seizures induced by loud noise, and in rats "kindled" to produce focal seizures by repeated prior electrical stimulation of the hippocampus. Gabapentin slightly increased spontaneous absence-like seizures in a genetically susceptible strain recorded with electroencephalography. All of these effects of gabapentin were seen at dosages at or below the threshold for producing ataxia.
Gabapentin has been tested in a variety of animal models that are relevant for analgesic actions.[83] Generally, gabapentin is not active to prevent pain-related behaviors in models of acute nociceptive pain. It prevents pain-related behaviors when animals are made sensitive by prior peripheral inflammation or peripheral nerve damage (inflammatory or neuropathic conditions).
Pharmacodynamics
Gabapentin is a ligand of the α2δ calcium channel subunit.[84][85] The α2δ-1 protein is coded by the CACNA2D1 gene. α2δ was first described as an auxiliary protein connected to the main α1 subunit (the channel-forming protein) of high voltage activated voltage-dependent calcium channels (L-type, N-type, P/Q type, and R-type).[5] The same α2δ protein has more recently been shown to interact directly with some NMDA-type and AMPA-type glutamate receptors at presynaptic sites and also with thrombospondin (an extracellular matrix protein secreted by astroglial cells).[86]
Gabapentin is not a direct calcium channel blocker: it exerts its actions by disrupting the regulatory function of α2δ and its interactions with other proteins. Gabapentin reduces delivery of intracellular calcium channels to the cell membrane, reduces the activation of the channels by the α2δ subunit, decreases signaling to lead to neurotransmitters release, and disrupts interactions of α2δ with voltage gated calcium channels but also with NMDA receptors, neurexins, and thrombospondin.[5][6][7] These proteins are found as mutually interacting parts of the presynaptic active zone, where numerous protein molecules interact with each other to enable and to regulate the release of neurotransmitters from presynaptic vesicles into the synaptic space. Out of the four known isoforms of α2δ protein, gabapentin binds with similar high affinity to two: α2δ-1 and α2δ-2.[85] All of the pharmacological properties of gabapentin tested to date are explained by its binding to just one isoform – α2δ-1.[85][6]
The endogenous α-amino acids L-leucine and L-isoleucine, which resemble gabapentin in chemical structure, bind α2δ with similar affinity to gabapentin and are present in human cerebrospinal fluid at micromolar concentrations.[87] They may be the endogenous ligands of the α2δ subunit, and they competitively antagonize the effects of gabapentin.[87][88] Accordingly, while gabapentin has nanomolar affinity for the α2δ subunit, its potency in vivo is in the low micromolar range, and competition for binding by endogenous L-amino acids is likely to be responsible for this discrepancy.[6]
Gabapentin is a potent activator of voltage-gated potassium channels KCNQ3 and KCNQ5, even at low nanomolar concentrations. However, this activation is unlikely to be the dominant mechanism of gabapentin's therapeutic effects.[89]
Gabapentin is structurally similar to the neurotransmitter glutamate and competitively inhibits branched-chain amino acid aminotransferase (BCAT), slowing down the synthesis of glutamate.[90] In particular, it inhibits BCAT-1 at high concentrations (Ki = 1 mM), but not BCAT-2.[91] At very high concentrations, gabapentin can suppress the growth of cancer cells, presumably by affecting mitochondrial catabolism; however, the precise mechanism remains elusive.[91]
Even though gabapentin is a structural GABA analog, and despite its name, it does not bind to the GABA receptors, does not convert into GABA or another GABA receptor agonist in vivo, and does not modulate GABA transport or metabolism within the range of clinical dosing.[84] In vitro gabapentin has been found to very weakly inhibit the GABA aminotransferase enzyme (Ki = 17–20 mM); however, this effect is so weak that it is not clinically relevant at prescribed doses.[90]
Pharmacokinetics
Gabapentin is absorbed from the intestines by an active transport process mediated via an amino acid transporter, presumably, LAT2.[92] As a result, the pharmacokinetics of gabapentin is dose-dependent, with diminished bioavailability and delayed peak levels at higher doses.[85]
The oral bioavailability of gabapentin is approximately 80% at 100 mg administered three times daily once every 8 hours, but decreases to 60% at 300 mg, 47% at 400 mg, 34% at 800 mg, 33% at 1,200 mg, and 27% at 1,600 mg, all with the same dosing schedule.[2][93] Drugs that increase the transit time of gabapentin in the small intestine can increase its oral bioavailability; when gabapentin was co-administered with oral morphine, the oral bioavailability of a 600 mg dose of gabapentin increased by 50%.[93]
Gabapentin at a low dose of 100 mg has a Tmax (time to peak levels) of approximately 1.7 hours, while the Tmax increases to 3 to 4 hours at higher doses.[85] Food does not significantly affect the Tmax of gabapentin and increases the Cmax and area-under-curve levels of gabapentin by approximately 10%.[93]
Gabapentin can cross the blood–brain barrier and enter the central nervous system.[84] Gabapentin concentration in cerebrospinal fluid is approximately 9–14% of its blood plasma concentration.[93] Due to its low lipophilicity,[93] gabapentin requires active transport across the blood–brain barrier.[94][84][95][96] The LAT1 is highly expressed at the blood–brain barrier[97] and transports gabapentin across into the brain.[94][84][95][96] As with intestinal absorption mediated by an amino acid transporter, the transport of gabapentin across the blood–brain barrier by LAT1 is saturable.[94] Gabapentin does not bind to other drug transporters such as P-glycoprotein (ABCB1) or OCTN2 (SLC22A5).[94] It is not significantly bound to plasma proteins (<1%).[93]
Gabapentin undergoes little or no metabolism.[85][93]
Gabapentin is generally safe in people with liver cirrhosis.[98]
Gabapentin is eliminated renally in the urine.[93] It has a relatively short elimination half-life, with the reported average value of 5 to 7 hours.[93] Because of its short elimination half-life, gabapentin must be administered 3 to 4 times per day to maintain therapeutic levels.[99] Gabapentin XR (brand name Gralise) is taken once a day.[100]
Chemistry
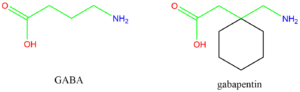
Gabapentin is a 3,3-disubstituted derivative of GABA. Therefore, it is a GABA analog, as well as a γ-amino acid.[101][102] It is similar to several other compounds that collectively are called gabapentinoids. Specifically, it is a derivative of GABA with a pentyl disubstitution at 3 position, hence, the name - gabapentin, in such a way as to form a six-membered ring. After the formation of the ring, the amine and carboxylic groups are not in the same relative positions as they are in the GABA;[103] they are more conformationally constrained.[104]
Although it has been known for some time that gabapentin must bind to the α2δ-1 protein in order to act pharmacologically (see Pharmacodynamics), the three-dimensional structure of the α2δ-1 protein with gabapentin bound (or alternatively, the native amino acid, L-Isoleucine bound) has only recently been obtained by cryo-electron microscopy.[105] A figure of this drug-bound structure is shown in the Chemistry section of the entry on gabapentinoid drugs. This study confirms other findings to show that both compounds alternatively can bind at a single extracellular site (somewhat distant from the calcium conducting pore of the voltage gated calcium channel α1 subunit) on the calcium channel and chemotaxis (Cache1) domain of α2δ-1.
Synthesis
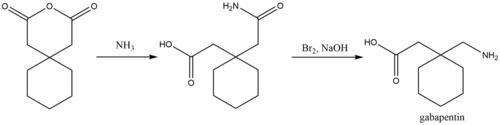
A process for chemical synthesis and isolation of gabapentin with high yield and purity[106] starts with conversion of 1,1-cyclohexanediacetic anhydride to 1,1-cyclohexanediacetic acid monoamide and is followed by a 'Hofmann' rearrangement in an aqueous solution of sodium hypobromite prepared in situ.
History
GABA is the principal inhibitory neurotransmitter in mammalian brains. By the early 1970s, it was appreciated that there are two main classes of GABA receptors, GABAA and GABAB and also that baclofen was an agonist of GABAB receptors. Gabapentin was designed, synthesized, and tested in mice by researchers at the pharmaceutical company Goedecke AG in Freiburg, Germany (a subsidiary of Parke-Davis). It was meant to be a structural analog of the neurotransmitter GABA that could more easily cross the blood–brain barrier. It was first synthesized in 1974/75 and described in 1975[107] by Satzinger and Hartenstein.[103][108]
The first pharmacology findings published were sedating properties and prevention of seizures in mice evoked by the GABA antagonist, thiosemicarbazide.[107] Shortly after, gabapentin was shown in vitro to reduce the release of the neurotransmitter dopamine from slices of rat caudate nucleus (striatum).[109] This study provided evidence that the action of gabapentin, unlike baclofen, did not arise from the GABAB receptor.
Initial clinical trials utilizing small numbers of subjects were for treatment of spasticity[110] and migraine[111] but neither study had statistical power to allow conclusions. In 1987, the first positive results with gabapentin were obtained in a clinical trial using three dose groups versus pre-treatment seizure frequency for 75 days, as add-on treatment in patients who still had seizures despite taking other medications.[112]
Under the brand name Neurontin, it was first approved in the United Kingdom in May 1993, for the treatment of refractory epilepsy.[113] Approval by the US Food and Drug Administration followed in December 1993, also for use as an adjuvant (effective when added to other antiseizure drugs) medication to control partial seizures in adults; that indication was extended to children in 2000.[114][2] Subsequently, gabapentin was approved in the United States for the treatment of pain from postherpetic neuralgia in 2002.[115] A generic version of gabapentin first became available in the United States in 2004.[10] An extended-release formulation of gabapentin for once-daily administration, under the brand name Gralise, was approved in the United States for the treatment of postherpetic neuralgia in January 2011.[116][117]
Gabapentin has been prescribed for a variety of disorders and is one of the more common medications used, particularly in elderly people.[118]
Society and culture
Legal status
United Kingdom
Effective April 2019, the United Kingdom reclassified the drug as a class C controlled substance.[119][120][121][122][123]
United States
Gabapentin is not a controlled substance under the federal Controlled Substances Act.[124] Effective in July 2017, Kentucky classified gabapentin as a Schedule V controlled substance statewide.[125] Gabapentin is a schedule V drug in other states such as West Virginia,[126] Tennessee,[127] Alabama,[128] Utah,[129] and Virginia.[130]
Off-label promotion
Although some small, non-controlled studies in the 1990s—mostly sponsored by gabapentin's manufacturer—suggested that treatment for bipolar disorder with gabapentin may be promising,[131] the preponderance of evidence suggests that it is not effective.[132]
Franklin v. Parke-Davis case
After the corporate acquisition of the original patent holder, the pharmaceutical company Pfizer admitted that there had been violations of FDA guidelines regarding the promotion of unproven off-label uses for gabapentin in the Franklin v. Parke-Davis case.
While off-label prescriptions are common for many drugs, marketing of off-label uses of a drug is not.[13] In 2004, Warner-Lambert (which subsequently was acquired by Pfizer) agreed to plead guilty for activities of its Parke-Davis subsidiary, and to pay $430 million in fines to settle civil and criminal charges regarding the marketing of Neurontin for off-label purposes. The 2004 settlement was one of the largest in U.S. history up to that point, and the first off-label promotion case brought successfully under the False Claims Act.[133]
Kaiser Foundation Hospitals and Kaiser Foundation Health Plan sued Pfizer Inc., alleging that the pharmaceutical company had misled Kaiser by recommending Neurontin as an off-label treatment for certain conditions (including bipolar disorder, migraines, and neuropathic pain).[134][135][136] In 2010, a federal jury in Massachusetts ruled in Kaiser's favor, finding that Pfizer violated the federal Racketeer Influenced and Corrupt Organizations (RICO) Act and was liable for US$47.36 million in damages, which was automatically trebled to just under $142.1 million.[135][134] Aetna, Inc. and a group of employer health plans prevailed in their similar Neurontin-related claims against Pfizer.[137] Pfizer appealed, but the U.S. Court of Appeals for the First Circuit upheld the verdict,[137] and in 2013, the US Supreme Court declined to hear the case.[138][139]
Gabasync
Gabasync, a treatment consisting of a combination of gabapentin and two other medications (flumazenil and hydroxyzine) as well as therapy, is an ineffective treatment promoted for methamphetamine addiction. It had also been claimed to be effective for dependence on alcohol or cocaine.[140] It was marketed as PROMETA. While the FDA has approved individual drugs, they have not approved their off-label use for addiction treatment.[141] Gabasync was marketed by Hythiam, Inc., which is owned by Terren Peizer, a former junk bond salesman who has since been convicted of securities fraud relative to another company.[142][143][144][140] Hythiam charges up to $15,000 per patient to license its use (of which half goes to the prescribing physician, and half to Hythiam).[145]
In a November 2005 article entitled "Curb Your Cravings For This Stock", Barrons wrote: "If the venture works out for patients and the investing public, it'll be a rare success for Peizer, who's promoted a series of disappointing small-cap medical or technology stocks ... since his days at Drexel".[146] 60 Minutes, NBC News, and The Dallas Morning News criticized Peizer after the company bypassed clinical studies and government approval when bringing to market Prometa; the addiction drug proved to be completely ineffective.[147][148][140][149] CBS News journalist Scott Pelley said to Peizer in 2007: "Depending on who you talk to, you're either a revolutionary or a snake oil salesman."[150][146] Journalist Adam Feuerstein opined: "most of what Peizer says is dubious-sounding hype".[151]
In November 2011, the results of a double-blind, placebo-controlled study (financed by Hythiam and carried out at UCLA) were published in the peer-reviewed journal Addiction. It concluded that Gabasync is ineffective: "The PROMETA protocol, consisting of flumazenil, gabapentin, and hydroxyzine, appears to be no more effective than placebo in reducing methamphetamine use, retaining patients in treatment, or reducing methamphetamine craving."[152]
Usage trends
The consumption of gabapentinoids rose significantly between 2008 and 2018.[153] A study published in 2023 highlights this trend, demonstrating a notable escalation in sales of gabapentinoids. The study, which analyzed healthcare data across 65 countries/ regions, found that the consumption rate of gabapentinoids had doubled over the decade, driven by their use in a wide range of indications.[154]
Brand names
Gabapentin was originally sold under the brand name Neurontin. Since it became generic, it has been sold worldwide using over 300 different brand names.[155] An extended-release formulation of gabapentin for once-daily administration was introduced in 2011 for postherpetic neuralgia under the brand name Gralise.[156]
In the US, Neurontin is sold by Viatris after Upjohn was spun off from Pfizer.[157][158][159]
Related drugs
Parke-Davis developed a drug called pregabalin, which is related in structure to gabapentin, as a successor to gabapentin.[160] Another similar drug atagabalin has been unsuccessfully tried by Pfizer as a treatment for insomnia.[161] A prodrug form (gabapentin enacarbil)[162] was approved by the U.S. Food and Drug Administration (FDA).
Recreational use
When taken in excess, gabapentin can induce euphoria, a sense of calm, improved sociability, and reduced alcohol or cocaine cravings.[163][164][165] Also known on the streets as "Gabbies",[166] gabapentin was reported in 2017 to be increasingly abused and misused for these euphoric effects.[167][168] About 1 percent of the responders to an Internet poll and 22 percent of those attending addiction facilities had a history of abuse of gabapentin.[72][169] Gabapentin misuse, toxicity, and use in suicide attempts among adults in the US increased from 2013 to 2017.[170]
After Kentucky implemented stricter legislation regarding opioid prescriptions in 2012, there was an increase in gabapentin-only and multi-drug use from 2012 to 2015. The majority of these cases were from overdose in suspected suicide attempts. Increases in abuse and recreational use accompanied these rates.[171]
Withdrawal symptoms, often resembling those of benzodiazepine withdrawal, play a role in the physical dependence some users experience.[73] Its misuse predominantly coincides with the usage of other CNS depressant drugs, namely opioids, benzodiazepines, and alcohol.[172]
Veterinary use
In cats, gabapentin can be used as an analgesic in multi-modal pain management,[173] anxiety medication to reduce stress during travel or vet visits,[174] and anticonvulsant.[175]
Veterinarians may prescribe gabapentin as an anticonvulsant and pain reliever in dogs.[176][175] It has beneficial effects for treating epilepsy, different kinds of pain (chronic, neuropathic, and post-operative pain), and anxiety, lip-licking behavior, storm phobia, fear-based aggression.[177][178]
It is also used to treat chronic pain-associated nerve inflammation in horses and dogs. Side effects include tiredness and loss of coordination, but these effects generally resolve within 24 hours of starting the medication.[176][175]
References
- ↑ 1.0 1.1 1.2 "Neuropathic pain in adults: pharmacological management in non-specialist settings". 20 November 2013. https://www.nice.org.uk/guidance/cg173/chapter/1-Recommendations.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 "Neurontin- gabapentin capsule Neurontin- gabapentin tablet, film coated Neurontin- gabapentin solution". 11 April 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ee9ad9ed-6d9f-4ee1-9d7f-cfad438df388.
- ↑ 3.0 3.1 3.2 3.3 3.4 "EFNS guidelines on the pharmacological treatment of neuropathic pain: 2010 revision". European Journal of Neurology 17 (9): 1113–1e88. September 2010. doi:10.1111/j.1468-1331.2010.02999.x. PMID 20402746.
- ↑ 4.0 4.1 "Gabapentin for chronic neuropathic pain in adults". The Cochrane Database of Systematic Reviews 6 (6). June 2017. doi:10.1002/14651858.CD007938.pub4. PMID 28597471.
- ↑ 5.0 5.1 5.2 "Emerging roles for α2δ subunits in calcium channel function and synaptic connectivity". Current Opinion in Neurobiology 63: 162–169. August 2020. doi:10.1016/j.conb.2020.04.007. PMID 32521436.
- ↑ 6.0 6.1 6.2 6.3 "The diverse therapeutic actions of pregabalin: is a single mechanism responsible for several pharmacological activities?". Trends in Pharmacological Sciences 34 (6): 332–339. June 2013. doi:10.1016/j.tips.2013.04.001. PMID 23642658.
- ↑ 7.0 7.1 "Analgesia with Gabapentin and Pregabalin May Involve N-Methyl-d-Aspartate Receptors, Neurexins, and Thrombospondins". The Journal of Pharmacology and Experimental Therapeutics 374 (1): 161–174. July 2020. doi:10.1124/jpet.120.266056. PMID 32321743.
- ↑ 8.0 8.1 "Gabapentin and suicide attempts". Pharmacoepidemiology and Drug Safety 19 (12). Oct 2010. doi:10.1002/pds.2036. PMID 20922708.
- ↑ Models of Seizures and Epilepsy. Burlington: Elsevier. 2005. p. 539. ISBN 978-0-08-045702-4. https://books.google.com/books?id=Qw6KqLjwtZQC&pg=PA539.
- ↑ 10.0 10.1 The Other End of the Stethoscope: The Physician's Perspective on the Health Care Crisis. AuthorHouse. 2 March 2012. pp. 63–. ISBN 978-1-4685-4410-7. https://books.google.com/books?id=HkICcDDz0qQC&pg=PA63.
- ↑ "The Top 300 of 2023". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Gabapentin Drug Usage Statistics, United States, 2014 - 2023". https://clincalc.com/DrugStats/Drugs/Gabapentin.
- ↑ 13.0 13.1 "Safeguarding patient welfare: who's in charge?". Annals of Internal Medicine 145 (4): 305–307. August 2006. doi:10.7326/0003-4819-145-4-200608150-00013. PMID 16908923.
- ↑ "Pfizer to pay $325 million in Neurontin settlement". Reuters. 2 June 2014. https://www.reuters.com/article/us-pfizer-neurontin-settlement/pfizer-to-pay-325-million-in-neurontin-settlement-idUSKBN0ED1IS20140602.
- ↑ 15.0 15.1 "Trends in First Gabapentin and Pregabalin Prescriptions in Primary Care in the United Kingdom, 1993-2017". JAMA 320 (20): 2149–2151. November 2018. doi:10.1001/jama.2018.12358. PMID 30480717.
- ↑ "A Clinical Overview of Off-label Use of Gabapentinoid Drugs". JAMA Internal Medicine 179 (5): 695–701. May 2019. doi:10.1001/jamainternmed.2019.0086. PMID 30907944.
- ↑ Successful Psychopharmacology: Evidence-Based Treatment Solutions for Achieving Remission. W. W. Norton. 5 November 2012. p. 124. ISBN 978-0-393-70857-8. https://books.google.com/books?id=dnAlO_Veu2QC&pg=PA124.
- ↑ "The Painkiller Used for Just About Anything". The New York Times. 17 August 2024. https://www.nytimes.com/2024/08/17/health/gabapentin-seniors-pain.html.
- ↑ 19.0 19.1 19.2 "Review finds little evidence to support gabapentinoid use in bipolar disorder or insomnia". NIHR Evidence (National Institute for Health and Care Research). 17 October 2022. doi:10.3310/nihrevidence_54173. https://evidence.nihr.ac.uk/alert/review-finds-little-evidence-support-gabapentinoid-use-bipolar-disorder-or-insomnia/.
- ↑ 20.0 20.1 20.2 20.3 20.4 "Gabapentin and pregabalin in bipolar disorder, anxiety states, and insomnia: Systematic review, meta-analysis, and rationale". Molecular Psychiatry 27 (3): 1339–1349. March 2022. doi:10.1038/s41380-021-01386-6. PMID 34819636.
- ↑ "Gabapentin withdrawal syndrome in the presence of a taper". Bipolar Disorders 7 (3): 302–4. June 2005. doi:10.1111/j.1399-5618.2005.00200.x. PMID 15898970.
- ↑ "Harms were detected but not reported in six clinical trials of gabapentin". Journal of Clinical Epidemiology 164: 76–87. December 2023. doi:10.1016/j.jclinepi.2023.10.014. PMID 37871835. https://osf.io/ej7fp.
- ↑ "Management of focal-onset seizures: an update on drug treatment". Drugs 66 (13): 1701–1725. 2006. doi:10.2165/00003495-200666130-00004. PMID 16978035.
- ↑ "Pharmacotherapy for tonic-clonic seizures". Expert Opinion on Pharmacotherapy 15 (10): 1417–1426. July 2014. doi:10.1517/14656566.2014.915029. PMID 24798217.
- ↑ "Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society". Pain Research & Management 12 (1): 13–21. 2007. doi:10.1155/2007/730785. PMID 17372630.
- ↑ 26.0 26.1 26.2 "Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis". The Lancet. Neurology 14 (2): 162–173. February 2015. doi:10.1016/S1474-4422(14)70251-0. PMID 25575710.
- ↑ 27.0 27.1 "Benefits and safety of gabapentinoids in chronic low back pain: A systematic review and meta-analysis of randomized controlled trials". PLOS Medicine 14 (8). August 2017. doi:10.1371/journal.pmed.1002369. PMID 28809936.
- ↑ "Anticonvulsants in the treatment of low back pain and lumbar radicular pain: a systematic review and meta-analysis". CMAJ 190 (26): E786–E793. July 2018. doi:10.1503/cmaj.171333. PMID 29970367.
- ↑ "Pharmacological treatment of painful HIV-associated sensory neuropathy: a systematic review and meta-analysis of randomised controlled trials". PLOS ONE 5 (12). December 2010. doi:10.1371/journal.pone.0014433. PMID 21203440. Bibcode: 2010PLoSO...514433P.
- ↑ "Gabapentin for Chronic Neuropathic Pain". JAMA 319 (8): 818–819. February 2018. doi:10.1001/jama.2017.21547. PMID 29486015.
- ↑ 31.0 31.1 31.2 "The role of anticonvulsant drugs in anxiety disorders: a critical review of the evidence". Journal of Clinical Psychopharmacology 27 (3): 263–272. June 2007. doi:10.1097/jcp.0b013e318059361a. PMID 17502773.
- ↑ 32.0 32.1 32.2 "Gabapentin and Pregabalin for the Treatment of Anxiety Disorders". Clinical Pharmacology in Drug Development 7 (3): 228–232. March 2018. doi:10.1002/cpdd.446. PMID 29579375.
- ↑ "Efficacy and Tolerability of Gabapentin in Adults with Sleep Disturbance in Medical Illness: A Systematic Review and Meta-analysis". Frontiers in Neurology 8. 2017. doi:10.3389/fneur.2017.00316. PMID 28769860.
- ↑ "Treatment effects of gabapentin for primary insomnia". Clinical Neuropharmacology 33 (2): 84–90. 2010. doi:10.1097/WNF.0b013e3181cda242. PMID 20124884.
- ↑ "Outpatient management of alcohol withdrawal syndrome". American Family Physician 88 (9): 589–595. November 2013. PMID 24364635.
- ↑ 36.0 36.1 "Gabapentin Therapy in Psychiatric Disorders: A Systematic Review". The Primary Care Companion for CNS Disorders 17 (5). 2015. doi:10.4088/PCC.15r01821. PMID 26835178.
- ↑ "Effectiveness of Gabapentin in Reducing Cravings and Withdrawal in Alcohol Use Disorder: A Meta-Analytic Review". The Primary Care Companion for CNS Disorders 21 (4). August 2019. doi:10.4088/PCC.19r02465. PMID 31461226.
- ↑ "A meta-analysis of the efficacy of gabapentin for treating alcohol use disorder". Addiction 114 (9): 1547–1555. September 2019. doi:10.1111/add.14655. PMID 31077485.
- ↑ 39.0 39.1 "Gabapentin for the treatment of alcohol use disorder". Expert Opinion on Investigational Drugs 27 (1): 113–124. January 2018. doi:10.1080/13543784.2018.1417383. PMID 29241365.
- ↑ "Gabapentin for smoking cessation". Nicotine & Tobacco Research 12 (3): 300–304. March 2010. doi:10.1093/ntr/ntp195. PMID 20081039.
- ↑ "Gabapentin Effect on Pain Associated with Heroin Withdrawal in Iranian Crack: a Randomized Double-blind Clinical Trial". Iranian Journal of Pharmaceutical Research 11 (3): 979–983. 2012. PMID 24250527.
- ↑ "Pharmacotherapies for cannabis dependence". The Cochrane Database of Systematic Reviews 1 (1). January 2019. doi:10.1002/14651858.CD008940.pub3. PMID 30687936.
- ↑ "The pharmacological treatment of nystagmus: a review". Expert Opinion on Pharmacotherapy 10 (11): 1805–1816. August 2009. doi:10.1517/14656560902978446. PMID 19601699.
- ↑ "Treatment of nystagmus". Current Treatment Options in Neurology 14 (1): 60–72. February 2012. doi:10.1007/s11940-011-0154-5. PMID 22072056.
- ↑ "The pharmacological treatment of acquired nystagmus". Practical Neurology 12 (3): 147–153. June 2012. doi:10.1136/practneurol-2011-000181. PMID 22661344.
- ↑ "Efficacy and safety of gabapentin and pregabalin in patients with vasomotor symptoms: a systematic review and meta-analysis". American Journal of Obstetrics and Gynecology 222 (6): 564–579.e12. June 2020. doi:10.1016/j.ajog.2019.12.011. PMID 31870736.
- ↑ "Pharmacological management of spasticity in multiple sclerosis: Systematic review and consensus paper". Multiple Sclerosis 22 (11): 1386–1396. October 2016. doi:10.1177/1352458516643600. PMID 27207462. https://discovery.ucl.ac.uk/id/eprint/1496196/.
- ↑ "Treatment of restless legs syndrome: Evidence-based review and implications for clinical practice (Revised 2017)§". Movement Disorders 33 (7): 1077–1091. July 2018. doi:10.1002/mds.27260. PMID 29756335. https://push-zb.helmholtz-muenchen.de/frontdoor.php?source_opus=54361.
- ↑ "Pruritus and renal failure". Seminars in Cutaneous Medicine and Surgery 30 (2): 99–100. June 2011. doi:10.1016/j.sder.2011.04.005. PMID 21767770.
- ↑ "Interventions for itch in people with advanced chronic kidney disease". The Cochrane Database of Systematic Reviews 2020 (12). December 2020. doi:10.1002/14651858.CD011393.pub2. PMID 33283264.
- ↑ "Gabapentin for pruritus in palliative care". The American Journal of Hospice & Palliative Care 30 (2): 192–196. March 2013. doi:10.1177/1049909112445464. PMID 22556282.
- ↑ "The treatment of tremor". Neurotherapeutics 11 (1): 128–138. January 2014. doi:10.1007/s13311-013-0230-5. PMID 24142589.
- ↑ "Evidence-based guideline update: treatment of essential tremor: report of the Quality Standards subcommittee of the American Academy of Neurology". Neurology 77 (19): 1752–1755. November 2011. doi:10.1212/WNL.0b013e318236f0fd. PMID 22013182.
- ↑ "Pharmacological management of essential tremor". Drugs 70 (17): 2215–2228. December 2010. doi:10.2165/11538180-000000000-00000. PMID 21080739.
- ↑ "A Systematic Review of the Clinical Use of Gabapentin and Pregabalin in Bipolar Disorder". Pharmaceuticals 14 (9): 834. August 2021. doi:10.3390/ph14090834. PMID 34577534.
- ↑ "Treatment of complex regional pain syndrome: a review of the evidence". Canadian Journal of Anaesthesia 57 (2): 149–166. February 2010. doi:10.1007/s12630-009-9237-0. PMID 20054678.
- ↑ "A Meta-Analysis on the Use of Gabapentinoids for the Treatment of Acute Postoperative Pain Following Total Knee Arthroplasty". The Journal of Bone and Joint Surgery. American Volume 98 (16): 1340–1350. August 2016. doi:10.2106/jbjs.15.01202. PMID 27535436. https://ora.ox.ac.uk/objects/uuid:cbe29197-14a9-47dc-b961-2ad3e24fd1c0. Retrieved 22 August 2020.
- ↑ "Gabapentin for tinnitus: a systematic review". American Journal of Audiology 20 (2): 151–158. December 2011. doi:10.1044/1059-0889(2011/10-0041). PMID 21940981.
- ↑ "Gabapentin or pregabalin for the prophylaxis of episodic migraine in adults". The Cochrane Database of Systematic Reviews 2013 (6). June 2013. doi:10.1002/14651858.CD010609. PMID 23797675.
- ↑ "Cardiovascular safety of gabapentinoids gabapentin & pregabalin: A systematic review.". Indian J Med Res 161 (4): 363–374. Apr 2025. doi:10.25259/IJMR_1990_2024. PMID 40536375.
- ↑ "Clinical review: Drugs commonly associated with weight change: a systematic review and meta-analysis". The Journal of Clinical Endocrinology and Metabolism 100 (2): 363–370. February 2015. doi:10.1210/jc.2014-3421. PMID 25590213.
- ↑ "Sexual dysfunction related to antiepileptic drugs in patients with epilepsy". Expert Opinion on Drug Safety 15 (1): 31–42. January 2016. doi:10.1517/14740338.2016.1112376. PMID 26559937.
- ↑ "Negative myoclonus induced by gabapentin and pregabalin: A case series and systematic literature review". Journal of the Neurological Sciences 382: 36–39. November 2017. doi:10.1016/j.jns.2017.09.019. PMID 29111014.
- ↑ "Gabapentin or pregabalin induced myoclonus: A case series and literature review". Journal of Clinical Neuroscience 61: 225–234. March 2019. doi:10.1016/j.jocn.2018.09.019. PMID 30381161.
- ↑ "Side effects of gabapentin". National Health Service. 16 September 2021. https://www.nhs.uk/medicines/gabapentin/side-effects-of-gabapentin/.
- ↑ 66.0 66.1 "Anticonvulsant medications and the risk of suicide, attempted suicide, or violent death". JAMA 303 (14): 1401–1409. April 2010. doi:10.1001/jama.2010.410. PMID 20388896.
- ↑ "The association between gabapentin and suicidality in bipolar patients". International Clinical Psychopharmacology 34 (1): 27–32. January 2019. doi:10.1097/YIC.0000000000000242. PMID 30383553.
- ↑ "Associations between gabapentinoids and suicidal behaviour, unintentional overdoses, injuries, road traffic incidents, and violent crime: population based cohort study in Sweden". BMJ 365. June 2019. doi:10.1136/bmj.l2147. PMID 31189556.
- ↑ Gibbons, R., Hur, K., Lavigne, J., Wang, J., & Mann, J. J. (2019). Medications and Suicide: High Dimensional Empirical Bayes Screening (iDEAS). Harvard Data Science Review, 1(2). https://doi.org/10.1162/99608f92.6fdaa9de
- ↑ 70.0 70.1 "FDA warns about serious breathing problems with seizure and nerve pain medicines gabapentin (Neurontin, Gralise, Horizant) and pregabalin (Lyrica, Lyrica CR)". 19 December 2019. https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-serious-breathing-problems-seizure-and-nerve-pain-medicines-gabapentin-neurontin.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ "Gabapentinoids and Risk for Severe Exacerbation in Chronic Obstructive Pulmonary Disease: A Population-Based Cohort Study". Annals of Internal Medicine 177 (Online ahead of print): 144–154. January 2024. doi:10.7326/M23-0849. PMID 38224592.
- ↑ 72.0 72.1 72.2 72.3 "Gabapentin: Abuse, Dependence, and Withdrawal". The Annals of Pharmacotherapy 50 (3): 229–233. March 2016. doi:10.1177/1060028015620800. PMID 26721643.
- ↑ 73.0 73.1 73.2 73.3 "How addictive are gabapentin and pregabalin? A systematic review". European Neuropsychopharmacology 27 (12): 1185–1215. December 2017. doi:10.1016/j.euroneuro.2017.08.430. PMID 28988943.
- ↑ "On the addictive power of gabapentinoids: a mini-review". Psychiatria Danubina 30 (2): 142–149. June 2018. doi:10.24869/psyd.2018.142. PMID 29930223. http://www.psychiatria-danubina.com/UserDocsImages/pdf/dnb_vol30_no2/dnb_vol30_no2_142.pdf.
- ↑ "A comprehensive review of the typical and atypical side effects of gabapentin". Pain Practice 24 (8): 1051–1058. November 2024. doi:10.1111/papr.13400. PMID 38949515.
- ↑ "Gabapentin (Oral Route)". Mayo Clinic. https://www.mayoclinic.org/drugs-supplements/gabapentin-oral-route/description/drg-20064011.
- ↑ "What are the side effects of gabapentin?". Medical News Today. March 2024. https://www.medicalnewstoday.com/articles/323753.
- ↑ "The association between Gabapentin or Pregabalin use and the risk of dementia: an analysis of the National Health Insurance Research Database in Taiwan". Frontiers in Pharmacology 14. 2023. doi:10.3389/fphar.2023.1128601. PMID 37324474.
- ↑ "Associations between gabapentinoids and suicidal behaviour, unintentional overdoses, injuries, road traffic incidents, and violent crime: population based cohort study in Sweden". BMJ (Clinical Research Ed.) 365. June 2019. doi:10.1136/bmj.l2147. PMID 31189556.
- ↑ "Gabapentinoids: a therapeutic review". Australian Prescriber 46 (4): 80–85. December 2023. doi:10.18773/austprescr.2023.025. PMID 38152314.
- ↑ R.C. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 677–8. ISBN 978-0-9626523-7-0.
- ↑ "Activity profile of pregabalin in rodent models of epilepsy and ataxia". Epilepsy Research 68 (3): 189–205. March 2006. doi:10.1016/j.eplepsyres.2005.11.001. PMID 16337109.
- ↑ "Mechanisms of the antinociceptive action of gabapentin". Journal of Pharmacological Sciences 100 (5): 471–486. 2006. doi:10.1254/jphs.CR0050020. PMID 16474201.
- ↑ 84.0 84.1 84.2 84.3 84.4 "The mechanisms of action of gabapentin and pregabalin". Current Opinion in Pharmacology 6 (1): 108–113. February 2006. doi:10.1016/j.coph.2005.11.003. PMID 16376147.
- ↑ 85.0 85.1 85.2 85.3 85.4 85.5 "Alpha2delta ligands, gabapentin, pregabalin and mirogabalin: a review of their clinical pharmacology and therapeutic use". Expert Review of Neurotherapeutics 16 (11): 1263–1277. November 2016. doi:10.1080/14737175.2016.1202764. PMID 27345098.
- ↑ "Analgesia with Gabapentin and Pregabalin May Involve N-Methyl-d-Aspartate Receptors, Neurexins, and Thrombospondins". The Journal of Pharmacology and Experimental Therapeutics 374 (1): 161–174. July 2020. doi:10.1124/jpet.120.266056. PMID 32321743.
- ↑ 87.0 87.1 "Ca2+ channel alpha2delta ligands: novel modulators of neurotransmission". Trends in Pharmacological Sciences 28 (2): 75–82. February 2007. doi:10.1016/j.tips.2006.12.006. PMID 17222465.
- ↑ "Functional biology of the alpha(2)delta subunits of voltage-gated calcium channels". Trends in Pharmacological Sciences 28 (5): 220–228. May 2007. doi:10.1016/j.tips.2007.03.005. PMID 17403543.
- ↑ "Gabapentin Is a Potent Activator of KCNQ3 and KCNQ5 Potassium Channels". Molecular Pharmacology 94 (4): 1155–1163. October 2018. doi:10.1124/mol.118.112953. PMID 30021858.
- ↑ 90.0 90.1 "Effects of anticonvulsant drug gabapentin on the enzymes in metabolic pathways of glutamate and GABA". Epilepsy Research 22 (1): 1–11. September 1995. doi:10.1016/0920-1211(95)00028-9. PMID 8565962.
- ↑ 91.0 91.1 "Gabapentin Can Suppress Cell Proliferation Independent of the Cytosolic Branched-Chain Amino Acid Transferase 1 (BCAT1)". Biochemistry 57 (49): 6762–6766. December 2018. doi:10.1021/acs.biochem.8b01031. PMID 30427175.
- ↑ "Pharmacokinetic role of L-type amino acid transporters LAT1 and LAT2". European Journal of Pharmaceutical Sciences 35 (3): 161–174. October 2008. doi:10.1016/j.ejps.2008.06.015. PMID 18656534.
- ↑ 93.0 93.1 93.2 93.3 93.4 93.5 93.6 93.7 93.8 "A comparison of the pharmacokinetics and pharmacodynamics of pregabalin and gabapentin". Clinical Pharmacokinetics 49 (10): 661–669. October 2010. doi:10.2165/11536200-000000000-00000. PMID 20818832.
- ↑ 94.0 94.1 94.2 94.3 "Transport of gabapentin by LAT1 (SLC7A5)". Biochemical Pharmacology 85 (11): 1672–1683. June 2013. doi:10.1016/j.bcp.2013.03.022. PMID 23567998.
- ↑ 95.0 95.1 "Molecular determinants of blood-brain barrier permeation". Therapeutic Delivery 6 (8): 961–971. 2015. doi:10.4155/tde.15.32. PMID 26305616.
- ↑ 96.0 96.1 "Prodrug approaches for enhancing the bioavailability of drugs with low solubility". Chemistry & Biodiversity 6 (11): 2071–2083. November 2009. doi:10.1002/cbdv.200900114. PMID 19937841.
- ↑ "Selective expression of the large neutral amino acid transporter at the blood-brain barrier". Proceedings of the National Academy of Sciences of the United States of America 96 (21): 12079–12084. October 1999. doi:10.1073/pnas.96.21.12079. PMID 10518579. Bibcode: 1999PNAS...9612079B.
- ↑ "The Safe Use of Analgesics in Patients with Cirrhosis: A Narrative Review". Am J Med 137 (2): 99–106. February 2024. doi:10.1016/j.amjmed.2023.10.022. PMID 37918778.
- ↑ "Gabapentin enacarbil - clinical efficacy in restless legs syndrome". Neuropsychiatric Disease and Treatment 6: 151–158. May 2010. doi:10.2147/NDT.S5712. PMID 20505847.
- ↑ Pharmacology, An Issue of Anesthesiology Clinics E-Book. Elsevier Health Sciences. 5 June 2017. pp. 98–. ISBN 978-0-323-52998-3. https://books.google.com/books?id=lsUmDwAAQBAJ&pg=PT98.
- ↑ Wyllie's Treatment of Epilepsy: Principles and Practice. Lippincott Williams & Wilkins. 17 February 2012. p. 423. ISBN 978-1-4511-5348-4. https://books.google.com/books?id=j9t6Qg0kkuUC&pg=RA1-PA423.
- ↑ Practical Management of Pain. Elsevier Health Sciences. 11 September 2013. p. 1006. ISBN 978-0-323-17080-2. https://books.google.com/books?id=kfcDAQAAQBAJ&pg=PA1006.
- ↑ 103.0 103.1 Drug Discovery: A History. John Wiley & Sons. 2005. pp. 219–220. ISBN 978-0-470-01552-0. https://books.google.com/books?id=jglFsz5EJR8C&pg=PA219.
- ↑ "Conformationally restricted GABA analogs: from rigid carbocycles to cage hydrocarbons". Future Medicinal Chemistry 3 (2): 223–241. February 2011. doi:10.4155/fmc.10.287. PMID 21428817.
- ↑ "Structural basis for CaVα2δ:gabapentin binding". Nature Structural & Molecular Biology 30 (6): 735–739. June 2023. doi:10.1038/s41594-023-00951-7. PMID 36973510.
- ↑ "Process For Synthesis Of Gabapentin". https://patents.google.com/patent/US20080103334A1/en.
- ↑ 107.0 107.1 Satzinger G, Hartenstein J, Herrmann M, Heldt W, "Cyclic amino acids", patent US4024175A, issued 1977-05-17
- ↑ The Art of Drug Synthesis. John Wiley & Sons. 26 February 2013. pp. 13–. ISBN 978-1-118-67846-6. https://books.google.com/books?id=zvruBDAulWEC&pg=SA13-PA41.
- ↑ "Inhibition by GABA, baclofen and gabapentin of dopamine release from rabbit caudate nucleus: are there common or different sites of action?". European Journal of Pharmacology 94 (3–4): 341–344. October 1983. doi:10.1016/0014-2999(83)90425-9. PMID 6653664.
- ↑ "A new antispastic agent: gabapentin: its effect on EMG analysis during voluntary movement in hemiplegia". Clinical Neurophysiology 62 (3): S221. 1985. doi:10.1016/0013-4694(85)90838-7. https://www.sciencedirect.com/journal/electroencephalography-and-clinical-neurophysiology.
- ↑ "Preliminary Results Of A Double Blind Study With The New Migraine Prophylactic Drug Gabapentin". Cephalalgia 7 (6_suppl): 477–478. 1987. doi:10.1177/03331024870070S6214. ISSN 0333-1024.
- ↑ "Gabapentin as an antiepileptic drug in man". Journal of Neurology, Neurosurgery, and Psychiatry 50 (6): 682–686. June 1987. doi:10.1136/jnnp.50.6.682. PMID 3302110. PMC 1032070. https://jnnp.bmj.com/content/50/6/682.
- ↑ "Drug Profile: Gabapentin". Adis Insight. https://adisinsight.springer.com/drugs/800002421.
- ↑ "Examination of the evidence for off-label use of gabapentin". Journal of Managed Care Pharmacy 9 (6): 559–568. 2003. doi:10.18553/jmcp.2003.9.6.559. PMID 14664664. PMC 10437292. http://www.amcp.org/data/jmcp/Contemporary%20Subject-559-568.pdf. Retrieved 15 August 2006.
- ↑ "Once-daily gastroretentive gabapentin for the management of postherpetic neuralgia: an update for clinicians". Therapeutic Advances in Chronic Disease 3 (5): 211–218. September 2012. doi:10.1177/2040622312452905. PMID 23342236.
- ↑ "Yabba Dabba Gabapentin: Are Gralise and Horizant Worth the Cost?". GoodRx, Inc.. 31 May 2013. https://www.goodrx.com/blog/yabba-dabba-gabapentin-are-gralise-and-horizant-worth-the-cost/.
- ↑ "Gabapentin controlled release – Depomed". Adis Insight. http://adisinsight.springer.com/drugs/800019682.
- ↑ "The Painkiller Used for Just About Anything". The New York Times. 17 August 2024. https://www.nytimes.com/2024/08/17/health/gabapentin-seniors-pain.html.
- ↑ "Pregabalin and gabapentin will become controlled drugs in April". 17 October 2018. https://nursingnotes.co.uk/pregabalin-gabapentin-will-become-controlled-drugs-april/.
- ↑ "Re: Pregabalin and Gabapentin advice". 14 January 2016. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/491854/ACMD_Advice_-_Pregabalin_and_gabapentin.pdf.
- ↑ "Pregabalin and gabapentin: proposal to schedule under the Misuse of Drugs Regulations 2001". 10 November 2017. https://www.gov.uk/government/consultations/pregabalin-and-gabapentin-proposal-to-schedule-under-the-misuse-of-drugs-regulations-2001.
- ↑ "Pregabalin and gabapentin become controlled drugs to cut deaths from misuse". BMJ 363. October 2018. doi:10.1136/bmj.k4364. PMID 30327316.
- ↑ "Pregabalin and gabapentin to be controlled as Class C drugs". 15 October 2018. https://www.gov.uk/government/news/pregabalin-and-gabapentin-to-be-controlled-as-class-c-drugs.
- ↑ "Gabapentin (Neurontin)". Drug Enforcement Administration. January 2023. https://www.deadiversion.usdoj.gov/drug_chem_info/gabapentin.pdf.
- ↑ "Important Notice: Gabapentin Becomes a Schedule 5 Controlled Substance in Kentucky". March 2017. https://pharmacy.ky.gov/Documents/Gabapentin%20-%20Schedule%20V%20Controlled%20Substance.pdf.
- ↑ "WV Code 212". http://www.wvlegislature.gov/WVCODE/ChapterEntire.cfm?chap=60a&art=2§ion=212.
- ↑ "Gabapentin will be a Schedule V controlled substance in Tennessee effective July 1, 2018". https://www.tn.gov/content/dam/tn/health/healthprofboards/New%20Statue%20Gabapentin%2006-18.pdf.
- ↑ "Pharmacy Division". http://www.alabamapublichealth.gov/pharmacy/.
- ↑ "Controlled Substances Amendments". https://le.utah.gov/~2024/bills/static/HB0260.html.
- ↑ "Scheduling of Gabapentin". Virginia Department of Health Professions. http://www.dhp.virginia.gov/Pharmacy/docs/gabapentin06172019.pdf.
- ↑ "Examination of the evidence for off-label use of gabapentin". Journal of Managed Care Pharmacy 9 (6): 559–568. 2003. doi:10.18553/jmcp.2003.9.6.559. PMID 14664664.
- ↑ "A systematic review on the role of anticonvulsants in the treatment of acute bipolar depression". The International Journal of Neuropsychopharmacology 16 (2): 485–496. March 2013. doi:10.1017/S1461145712000491. PMID 22575611.
- ↑ "Huge penalty in drug fraud, Pfizer settles felony case in Neurontin off-label promotion". San Francisco Chronicle: p. C-1. 14 May 2004. http://www.sfgate.com/cgi-bin/article.cgi?f=/c/a/2004/05/14/BUGKK6L0LB1.DTL.
- ↑ 134.0 134.1 "US jury's Neurontin ruling to cost Pfizer $141 mln". Reuters. 25 March 2010. https://www.reuters.com/article/pfizer-neurontin-idUSN259778920100325/.
- ↑ 135.0 135.1 "Pfizer faces $142M in damages for drug fraud". Bloomberg Businessweek. 25 March 2010. http://www.businessweek.com/ap/financialnews/D9ELVKG80.htm.
- ↑ "Pfizer Told to Pay $142.1 Million for Neurontin Marketing Fraud". 26 March 2010. https://www.bloomberg.com/apps/news?pid=email_en&sid=a_9aVylZQGjU.
- ↑ 137.0 137.1 "Pfizer Appeal Targets Fraudulent Drug Marketing Claims Brought Under Civil RICO Statute". Healthcare Law Insights (Hugh Blackwell). 3 October 2013. https://www.healthcarelawinsights.com/2013/10/pfizer-appeal-targets-fraudulent-drug-marketing-claims-brought-under-civil-rico-statute/.
- ↑ "US high court leaves intact $142 million verdict against Pfizer". Reuters. 9 December 2013. https://www.reuters.com/article/idUSBRE9B80K1/.
- ↑ "Pfizer Inc. v. Kaiser Foundation Health Plan, Inc.: Petition for certiorari denied on December 9, 2013". SCOTUSBlog. https://www.scotusblog.com/case-files/cases/pfizer-inc-v-kaiser-foundation-health-plan-inc/.
- ↑ 140.0 140.1 140.2 "Prescription For Addiction". 60 Minutes. 7 December 2007. http://www.cbsnews.com/news/prescription-for-addiction/.
- ↑ "Prometa Founder's Spotty Background Explored". 3 November 2006. http://www.drugfree.org/join-together/prometa-founders-spotty-background-explored/.
- ↑ Dave Michaels (21 June 2024). "Jury Convicts Milken Protégé Terren Peizer of Insider Trading," The Wall Street Journal.
- ↑ "Santa Monica man used anti-insider-trading measure to commit fraud: DOJ". 1 March 2023. https://ktla.com/news/local-news/santa-monica-man-used-anti-insider-trading-measure-to-commit-fraud-doj/.
- ↑ "United States V. Terren S. Peizer". 1 March 2023. https://www.justice.gov/criminal-vns/case/united-states-v-terren-s-peizer.
- ↑ "Prometa under fire in Washington drug court program". Alcoholism & Drug Abuse Weekly 20 (3). 21 January 2008. doi:10.1002/adaw.20121.
- ↑ 146.0 146.1 "Curb Your Cravings For This Stock". The Wall Street Journal. 7 November 2005. https://www.wsj.com/articles/SB113115427427989094.
- ↑ "Unproven meth, cocaine 'remedy' hits market". 5 February 2007. http://www.nbcnews.com/health/health-news/unproven-meth-cocaine-remedy-hits-market-flna1C9444547.
- ↑ "The Rise and Fall of a "Miracle Cure" for Drug Addiction". Washington Monthly. 24 January 2012. http://washingtonmonthly.com/2012/01/24/the-rise-and-fall-of-a-miracle-cure-for-drug-addiction/.
- ↑ "Texas' Prometa program for treating meth addicts draws skeptics". Dallas Morning News. 20 January 2008. https://www.dallasnews.com/sharedcontent/dws/dn/latestnews/stories/012108dntexprometa.2c2f801.html.
- ↑ "Prescription For Addiction". 7 December 2007. https://www.cbsnews.com/news/prescription-for-addiction/.
- ↑ "Hythiam, Shire, Genentech; Talk is proving cheap at Hythiam". Biotech Notebook. TheStreet. 13 November 2007. https://www.thestreet.com/investing/stocks/biotech-notebook-hythiam-shire-genentech-10389850.
- ↑ "Double-blind placebo-controlled evaluation of the PROMETA™ protocol for methamphetamine dependence". Addiction 107 (2): 361–369. February 2012. doi:10.1111/j.1360-0443.2011.03619.x. PMID 22082089.
- ↑ "The Hidden Risks of America's Most Popular Prescription Painkiller". December 25, 2025. https://www.wsj.com/health/gabapentin-painkiller-hidden-risks-603e6130.
- ↑ "Gabapentinoid consumption in 65 countries and regions from 2008 to 2018: a longitudinal trend study". Nature Communications 14 (1). August 2023. doi:10.1038/s41467-023-40637-8. PMID 37591833. PMC 10435503. Bibcode: 2023NatCo..14.5005C. https://pure.rug.nl/ws/files/799417606/s41467-023-40637-8.pdf.
- ↑ "International listings for Gabapentin". Drugs.com. https://www.drugs.com/international/gabapentin.html.
- ↑ "Gralise Approval History". Drugs.com. https://www.drugs.com/history/gralise.html.
- ↑ "Pfizer Completes Transaction to Combine Its Upjohn Business with Mylan". Pfizer. 16 November 2020. https://www.businesswire.com/news/home/20201116005378/en/.
- ↑ "Neurontin". https://www.pfizer.com/products/product-detail/neurontin.
- ↑ "Brands". 16 November 2020. https://www.viatris.com/en/products/brands.
- ↑ "The mechanism of action of gabapentin in neuropathic pain". Current Opinion in Investigational Drugs 7 (1): 33–39. January 2006. PMID 16425669.
- ↑ "Modeling sleep data for a new drug in development using markov mixed-effects models". Pharmaceutical Research 28 (10): 2610–2627. October 2011. doi:10.1007/s11095-011-0490-x. PMID 21681607.
- ↑ "Modifications of antiepileptic drugs for improved tolerability and efficacy". Perspectives in Medicinal Chemistry 2: 21–39. February 2008. doi:10.1177/1177391X0800200001. PMID 19787095.
- ↑ "Substance misuse of gabapentin". The British Journal of General Practice 62 (601): 406–407. August 2012. doi:10.3399/bjgp12X653516. PMID 22867659.
- ↑ "Gabapentin abuse and overdose: a case report". J Subst Abus Alcohol 2: 1018. 2014. https://pdfs.semanticscholar.org/9bab/627065e943e1508057231c865569bf20f5d3.pdf.
- ↑ "A Call for Caution in Prescribing Gabapentin to Individuals With Concurrent Polysubstance Abuse: A Case Report". Journal of Psychiatric Practice 25 (4): 308–312. July 2019. doi:10.1097/PRA.0000000000000403. PMID 31291212.
- ↑ Oxford Textbook of Correctional Psychiatry. Oxford University Press. April 2015. p. 167. ISBN 978-0-19-936057-4.
- ↑ "Gabapentin and Pregabalin for Pain - Is Increased Prescribing a Cause for Concern?". The New England Journal of Medicine 377 (5): 411–414. August 2017. doi:10.1056/NEJMp1704633. PMID 28767350.
- ↑ "Abuse and Misuse of Pregabalin and Gabapentin". Drugs 77 (4): 403–426. March 2017. doi:10.1007/s40265-017-0700-x. PMID 28144823.
- ↑ "[On the risk of dependence on gabapentinoids]" (in de). Fortschritte der Neurologie-Psychiatrie 86 (2): 82–105. February 2018. doi:10.1055/s-0043-122392. PMID 29179227.
- ↑ "Trends in gabapentin and baclofen exposures reported to U.S. poison centers". Clinical Toxicology 58 (7): 763–772. July 2020. doi:10.1080/15563650.2019.1687902. PMID 31786961.
- ↑ "Trending gabapentin exposures in Kentucky after legislation requiring the use of the state prescription drug monitoring program for all opioid prescriptions". Clinical Toxicology 57 (6): 398–403. June 2019. doi:10.1080/15563650.2018.1538518. PMID 30676102.
- ↑ "Gabapentin misuse, abuse and diversion: a systematic review". Addiction 111 (7): 1160–1174. July 2016. doi:10.1111/add.13324. PMID 27265421.
- ↑ "Gabapentin as part of multi-modal analgesia in two cats suffering multiple injuries". Veterinary Anaesthesia and Analgesia 38 (5): 518–520. September 2011. doi:10.1111/j.1467-2995.2011.00638.x. PMID 21831060.
- ↑ "Effects of a single preappointment dose of gabapentin on signs of stress in cats during transportation and veterinary examination". Journal of the American Veterinary Medical Association 251 (10): 1175–1181. November 2017. doi:10.2460/javma.251.10.1175. PMID 29099247.
- ↑ 175.0 175.1 175.2 "Gabapentin". https://www.plumbsveterinarydrugs.com/#!/veterinarymedicationguides.
- ↑ 176.0 176.1 "Gabapentin for Dogs: Uses and Side Effects". 31 October 2022. https://www.akc.org/expert-advice/health/gabapentin-for-dogs/.
- ↑ "Gabapentin: Clinical Use and Pharmacokinetics in Dogs, Cats, and Horses". Animals 13 (12): 2045. June 2023. doi:10.3390/ani13122045. PMID 37370556.
- ↑ "Effects of Gabapentin on the Treatment of Behavioral Disorders in Dogs: A Retrospective Evaluation". Animals 14 (10): 1462. May 2024. doi:10.3390/ani14101462. PMID 38791679.
External links
| Wikimedia Commons has media related to Gabapentin. |
| Scholia has a topic profile for Gabapentin. |
 |
