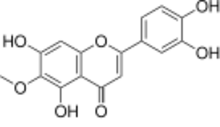
Nepetin
|
|
| Names
|
| IUPAC name
3′,4′,5,7-Tetrahydroxy-6-methoxyflavone
|
Systematic IUPAC name
2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-6-methoxy-4H-1-benzopyran-4-one |
| Other names
6-Methoxyluteolin
|
| Identifiers
|
|
|
|
|
|
|
| ChEMBL
|
|
| ChemSpider
|
|
|
|
|
| UNII
|
|
InChI=1S/C16H12O7/c1-22-16-11(20)6-13-14(15(16)21)10(19)5-12(23-13)7-2-3-8(17)9(18)4-7/h2-6,17-18,20-21H,1H3  N NKey: FHHSEFRSDKWJKJ-UHFFFAOYSA-N  N NInChI=1/C16H12O7/c1-22-16-11(20)6-13-14(15(16)21)10(19)5-12(23-13)7-2-3-8(17)9(18)4-7/h2-6,17-18,20-21H,1H3 Key: FHHSEFRSDKWJKJ-UHFFFAOYAR
|
COC1=C(C2=C(C=C1O)OC(=CC2=O)C3=CC(=C(C=C3)O)O)O
|
| Properties
|
|
|
C16H12O7
|
| Molar mass
|
316.26 g/mol
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
 N verify (what is N verify (what is  Y Y N ?) N ?)
|
| Infobox references
|
|
|
|
Tracking categories (test):
Nepetin is the 6-methoxy derivative of the pentahydroxyflavone 6-hydroxyluteolin, an O-methylated flavone. It can be found in Eupatorium ballotaefolium.[1]
Glycosides
Nepitrin is the 7-glucoside of nepetin.
References
- ↑ Cytotoxic activity of nepetin, a flavonoid from Eupatorium ballotaefolium HBK. Militao G. C. G., Albuquerque M. R. J. R., Pessoa O. D. L., Pessoa C., Moraes M. E. A., De Moraes M. O., Costa-Lotufo L. V., Pharmazie, 2004, vol. 59, no12, pp. 965-966
|
|---|
| Aglycones | | Monohydroxyflavone | |
|---|
| Dihydroxyflavones | |
|---|
| Trihydroxyflavones | |
|---|
| Tetrahydroxyflavones | |
|---|
| Pentahydroxyflavones | |
|---|
| O-methylated flavones | |
|---|
|
|---|
| Glycosides | | of apigenin | |
|---|
| of baicalein | |
|---|
| of hypolaetin | |
|---|
| of luteolin | |
|---|
|
|---|
| Acetylated | |
|---|
| Sulfated glycosides | |
|---|
| Polymers | |
|---|
| Drugs | |
|---|
 | Original source: https://en.wikipedia.org/wiki/Nepetin. Read more |



