Chemistry:Baicalein
 | |
 | |
| Clinical data | |
|---|---|
| Other names | Biacalein; Noroxylin |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C15H10O5 |
| Molar mass | 270.240 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
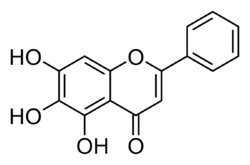
Baicalein (5,6,7-trihydroxyflavone) is a flavone, a type of flavonoid, originally isolated from the roots of Scutellaria baicalensis and Scutellaria lateriflora. It is also reported in Oroxylum indicum (Indian trumpetflower) and Thyme.[1] It is the aglycone of baicalin. Baicalein is one of the active ingredients of Sho-Saiko-To, which is a Chinese classic herbal formula, and listed in Japan as Kampo medicine.[citation needed]
Baicalein, along with its analogue baicalin, is a positive allosteric modulator of the benzodiazepine site and/or a non-benzodiazepine site of the GABAA receptor but with an affinity over 250× lower than diazepam.[2][3][4][5][6][7] It displays subtype selectivity for α2 and α3 subunit-containing GABAA receptors.[8] In accordance, baicalein shows anxiolytic effects in mice without incidence of sedation or myorelaxation.[7][8][9] It is thought that baicalein, along with other flavonoids, may underlie the anxiolytic effects of S. baicalensis and S. lateriflora.[10][11] Baicalein is also an antagonist of the estrogen receptor, or an antiestrogen.[11]
The flavonoid has been shown to inhibit certain types of lipoxygenases[12] and act as an anti-inflammatory agent.[13] It has antiproliferative effects on ET-1-induced proliferation of pulmonary artery smooth muscle cell proliferation via inhibition of TRPC1 channel expression.[14] Possible antidepressant effects have also been attributed to baicalein in animal research.[15]
Baicalein is an inhibitor of CYP2C9,[16] an enzyme of the cytochrome P450 system that metabolizes drugs in the body.
A derivative of baicalin is a known prolyl endopeptidase inhibitor.[17]
Baicalein has been shown to inhibit Staphylococcus aureus biofilm formation and the quorum sensing system in vitro.[18]
It has also been shown to be effective in vitro against all forms of Borrelia burgdorferi and Borrelia garinii.[19]
Glycosides
Tetuin is the 6-glucoside of baicalein.
See also
- List of compounds with carbon number 15
References
- ↑ Phytochemistry Research Progress. Nova Publishers. 2008. ISBN 9781604562323. https://books.google.com/books?id=l5Mo0M5CqQcC&q=%22Baicalein%22+and+%22thymus+vulgaris%22&pg=PA42.
- ↑ "Two flavones from Scutellaria baicalensis Georgi and their binding affinities to the benzodiazepine site of the GABAA receptor complex". Die Pharmazie 57 (12): 857–858. December 2002. PMID 12561253.
- ↑ "Interaction of flavones from the roots of Scutellaria baicalensis with the benzodiazepine site". Planta Medica 66 (1): 91–93. February 2000. doi:10.1055/s-0029-1243121. PMID 10705749.
- ↑ "Baicalein reduces β-amyloid and promotes nonamyloidogenic amyloid precursor protein processing in an Alzheimer's disease transgenic mouse model". Journal of Neuroscience Research 91 (9): 1239–1246. September 2013. doi:10.1002/jnr.23244. PMID 23686791.
- ↑ "Benzodiazepine binding site-interactive flavones from Scutellaria baicalensis root". Planta Medica 64 (6): 571–572. August 1998. doi:10.1055/s-2006-957517. PMID 9776664.
- ↑ "Testing efficacy of natural anxiolytic compounds". Complementary and Alternative Approaches to Biomedicine. Advances in Experimental Medicine and Biology. 546. 2004. pp. 181–191. doi:10.1007/978-1-4757-4820-8_13. ISBN 978-1-4419-3441-3.
- ↑ 7.0 7.1 "Involvement of GABAergic non-benzodiazepine sites in the anxiolytic-like and sedative effects of the flavonoid baicalein in mice". Behavioural Brain Research 221 (1): 75–82. August 2011. doi:10.1016/j.bbr.2011.02.038. PMID 21377498.
- ↑ 8.0 8.1 "GABA A receptor subtype selectivity underlying selective anxiolytic effect of baicalin". Neuropharmacology 55 (7): 1231–1237. December 2008. doi:10.1016/j.neuropharm.2008.07.040. PMID 18723037.
- ↑ "Anxiolytic-like effects of baicalein and baicalin in the Vogel conflict test in mice". European Journal of Pharmacology 464 (2–3): 141–146. March 2003. doi:10.1016/s0014-2999(03)01422-5. PMID 12620506.
- ↑ "Phytochemical and biological analysis of skullcap (Scutellaria lateriflora L.): a medicinal plant with anxiolytic properties". Phytomedicine 10 (8): 640–649. November 2003. doi:10.1078/0944-7113-00374. PMID 14692724.
- ↑ 11.0 11.1 "Native American Psychoactive Herbs". Psychoactive Herbs in Veterinary Behavior Medicine. John Wiley & Sons. 9 January 2008. pp. 139–. ISBN 978-0-470-34434-7. https://books.google.com/books?id=ZP6QVep-x24C&pg=PA139.
- ↑ "Baicalein is a potent in vitro inhibitor against both reticulocyte 15-human and platelet 12-human lipoxygenases". Bioorganic & Medicinal Chemistry 14 (12): 4295–4301. June 2006. doi:10.1016/j.bmc.2006.01.057. PMID 16500106. https://www.escholarship.org/uc/item/8mj512rb.
- ↑ "Baicalein inhibits IL-1beta- and TNF-alpha-induced inflammatory cytokine production from human mast cells via regulation of the NF-kappaB pathway". Clinical and Molecular Allergy 5 (1): 5. November 2007. doi:10.1186/1476-7961-5-5. PMID 18039391.
- ↑ "Baicalein, isolated from Scutellaria baicalensis, protects against endothelin-1-induced pulmonary artery smooth muscle cell proliferation via inhibition of TRPC1 channel expression". Journal of Ethnopharmacology 138 (2): 373–381. November 2011. doi:10.1016/j.jep.2011.09.014. PMID 21963569.
- ↑ "Antidepressant effects of a plant-derived flavonoid baicalein involving extracellular signal-regulated kinases cascade". Biological & Pharmaceutical Bulletin 34 (2): 253–259. 2011. doi:10.1248/bpb.34.253. PMID 21415537.
- ↑ "Mechanism of CYP2C9 inhibition by flavones and flavonols". Drug Metabolism and Disposition 37 (3): 629–634. March 2009. doi:10.1124/dmd.108.023416. PMID 19074529. http://p4502c.googlepages.com/dmd2.pdf. Retrieved 2009-02-19.
- ↑ "Baicalin, a prodrug able to reach the CNS, is a prolyl oligopeptidase inhibitor". Bioorganic & Medicinal Chemistry 16 (15): 7516–7524. August 2008. doi:10.1016/j.bmc.2008.04.067. PMID 18650094.
- ↑ "Baicalein Inhibits Staphylococcus aureus Biofilm Formation and the Quorum Sensing System In Vitro". PLOS ONE 11 (4): e0153468. April 2016. doi:10.1371/journal.pone.0153468. PMID 27128436. Bibcode: 2016PLoSO..1153468C.
- ↑ "In vitro evaluation of antibacterial activity of phytochemicals and micronutrients against Borrelia burgdorferi and Borrelia garinii". Journal of Applied Microbiology 119 (6): 1561–1572. December 2015. doi:10.1111/jam.12970. PMID 26457476.
 |
