Medicine:Octreotide scan
| Octreotide scan | |
|---|---|
| Medical diagnostics | |
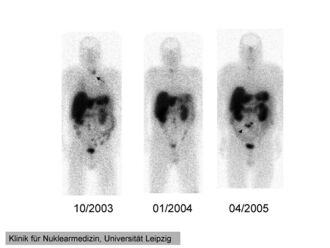 111In-pentetreotide scintigraphy of a 41-year-old man with ectopic Cushing's syndrome caused by a neuroendocrine carcinoma of the mesentery. Radiotracer accumulation in the left thyroid in 10/2003 (arrow). The mesenterial neuroendocrine tumor became clearly visible in 4/2005 (arrow). | |
| Synonyms | ocreoscan |
| ICD-9-CM | 92.18 |
| OPS-301 code | 3-70c |
An octreotide scan is a type of SPECT scintigraphy used to find carcinoid, pancreatic neuroendocrine tumors, and to localize sarcoidosis. It is also called somatostatin receptor scintigraphy (SRS). Octreotide, a drug similar to somatostatin, is radiolabeled with indium-111,[1] and is injected into a vein and travels through the bloodstream. The radioactive octreotide attaches to tumor cells that have receptors for somatostatin (i.e. gastrinoma, glucagonoma, etc.). A gamma camera detects the radioactive octreotide, and makes pictures showing where the tumor cells are in the body, typically by a SPECT technique. A technetium-99m based radiopharmaceutical kit is also available.[2][3]
Octreotide scanning is reported to have a sensitivity between 75% and 100% for detecting pancreatic neuroendocrine tumors.[4]
Instead of gamma-emitting 111In, certain octreotide derivatives such as edotreotide (DOTATOC) or DOTATATE are able to be linked by chelation to positron-emitting isotopes such as gallium-68 and copper-64 which in turn can be evaluated with more precise (compared with SPECT) scanning techniques such as PET-CT. Thus, the octreotide scan is now being replaced in most centers with gallium-68 DOTATATE and copper-64 DOTATATE scans. Somatostatin receptor imaging can now be performed with positron emission tomography (PET) which offers higher resolution and more rapid imaging.[5]
Indications
An octreotide scan may be used to locate suspected primary neuroendocrine tumours (NET) or for follow-up or staging after treatment.[6][7][8]
Where indicated, octreotide scanning for NET tumors is being increasingly replaced by gallium-68 DOTA and copper-64 DOTATATE scans.[9]
Procedure
Indium-111
 | |
| Clinical data | |
|---|---|
| Trade names | Octreoscan |
| Other names | MP-1727 |
| License data | |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| Chemical and physical data | |
| 3D model (JSmol) | |
| |
| |
The indium-111 pentetreotide radiopharmaceutical is prepared from a kit in a radiopharmacy. Pentetreotide is a DTPA conjugate of octreotide.[6][11]
Approximately 200 megabecquerels (MBq) of indium-111 is injected intravenously. Imaging takes place 24 hours after injection, but may also be carried out at 4 and 48 hours.[7][12]
Technetium-99m
The 99mTc product is supplied as a kit with two vials, one containing the chelating agent ethylenediaminediacetic acid (EDDA) and the other the HYNIC-Tyr3-octreotide chelator and somatostatin analog.[13] Approximately 400-700 MBq may be administered, with imaging at 2, 4, and occasionally 24 hours post administration.[14] 99mTc based octreotide imaging shows slightly higher sensitivity than 111In.[2][15]
References
- ↑ medicinenet.com > Carcinoid Syndrome (cont.) By Dennis Lee and Jay Marks. Retrieved Mars 2011
- ↑ 2.0 2.1 "99mTc-EDDA/HYNIC-TOC is a New Opportunity in Neuroendocrine Tumors of the Lung (and in other Malignant and Benign Pulmonary Diseases)". Current Radiopharmaceuticals 13 (3): 166–176. 30 November 2020. doi:10.2174/1874471013666191230143610. PMID 31886756.
- ↑ "Limitations and pitfalls of 99mTc-EDDA/HYNIC-TOC (Tektrotyd) scintigraphy" (in de). Nuclear Medicine Review. Central & Eastern Europe 19 (2): 93–98. November 2019. doi:10.5603/NMR.2016.0019. PMID 27479887.
- ↑ "Somatostatin receptor imaging". Seminars in Nuclear Medicine 32 (2): 84–91. April 2002. doi:10.1053/snuc.2002.31022. PMID 11965603.
- ↑ "High management impact of Ga-68 DOTATATE (GaTate) PET/CT for imaging neuroendocrine and other somatostatin expressing tumours". Journal of Medical Imaging and Radiation Oncology 56 (1): 40–47. February 2012. doi:10.1111/j.1754-9485.2011.02327.x. PMID 22339744.
- ↑ 6.0 6.1 "ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: somatostatin receptor imaging with (111)In-pentetreotide". Neuroendocrinology 90 (2): 184–189. 2009. doi:10.1159/000225946. PMID 19713709. https://boris.unibe.ch/32122/.
- ↑ 7.0 7.1 "111In-pentetreotide scintigraphy: procedure guidelines for tumour imaging". European Journal of Nuclear Medicine and Molecular Imaging 37 (7): 1441–1448. July 2010. doi:10.1007/s00259-010-1473-6. PMID 20461371.
- ↑ (in en) Fundamentals of Nuclear Pharmacy. Springer. 2010. p. 145. ISBN 9781441958600. https://books.google.com/books?id=bEXqI4ACk-AC&pg=PA145.
- ↑ "Management of Small Bowel Neuroendocrine Tumors". Journal of Oncology Practice 14 (8): 471–482. August 2018. doi:10.1200/JOP.18.00135. PMID 30096273.
- ↑ "Octreoscan- indium in -111 pentetreotide kit". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=93d8f3b2-1216-41dc-a63d-0e812b33891d.
- ↑ "In 111 pentetreotide" (in en). National Cancer Institute. 2011-02-02. https://www.cancer.gov/publications/dictionaries/cancer-drug?cdrid=43127.
- ↑ "The SNM practice guideline for somatostatin receptor scintigraphy 2.0". Journal of Nuclear Medicine Technology 39 (4): 317–324. December 2011. doi:10.2967/jnmt.111.098277. PMID 22068564.
- ↑ "TEKTROTYD". http://www.rotop-pharmaka.de/en/our-products/99m-tc-kits/tektrotyd/.
- ↑ "Limitations and pitfalls of 99mTc-EDDA/HYNIC-TOC (Tektrotyd) scintigraphy". Nuclear Medicine Review. Central & Eastern Europe 19 (2): 93–98. 2016. doi:10.5603/NMR.2016.0019. PMID 27479887.
- ↑ "TEKTROTYD Public Assessment Report (PAR)". Heads of Medicines Agencies. 6 May 2020. https://mri.cts-mrp.eu/Human/Product/Details/45950.
External links
- "Indium In 111 pentetreotide". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/rn/139096-04-1.
- Octreotide scan entry in the public domain NCI Dictionary of Cancer Terms
![]() This article incorporates public domain material from the U.S. National Cancer Institute document "Dictionary of Cancer Terms".
This article incorporates public domain material from the U.S. National Cancer Institute document "Dictionary of Cancer Terms".
 |
