Chemistry:Transition metal chloride complex

In chemistry, a transition metal chloride complex is a coordination complex that consists of a transition metal coordinated to one or more chloride ligand. The class of complexes is extensive.[1]
Bonding
Halides are X-type ligands in coordination chemistry. They are both σ- and π-donors. Chloride is commonly found as both a terminal ligand and a bridging ligand. The halide ligands are weak field ligands. Due to a smaller crystal field splitting energy, the homoleptic halide complexes of the first transition series are all high spin. Only [CrCl6]3− is exchange inert.
Homoleptic metal halide complexes are known with several stoichiometries, but the main ones are the hexahalometallates and the tetrahalometallates. The hexahalides adopt octahedral coordination geometry, whereas the tetrahalides are usually tetrahedral. Square planar tetrahalides are known for Pd(II), Pt(II), and Au(III). Examples with 2- and 3-coordination are common for Au(I), Cu(I), and Ag(I).
Due to the presence of filled pπ orbitals, halide ligands on transition metals are able to reinforce π-backbonding onto a π-acid. They are also known to labilize cis-ligands.[2] [3]
Homoleptic complexes
Homoleptic complexes (complexes with only chloride ligands) are often common reagents. Almost all examples are anions.
1st row
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| TiCl4 | colourless | (t2g)0 | tetrahedral | ||
| [Ti2Cl9]− | white/colourless | d0d0 | face-sharing bioctahedron | Ti-Cl(terminal) = 2.23 Å, 2.45 (terminal) (N(PCl3)2)+ salt)[4] | |
| [Ti2Cl9]3- | orange | (t2g)1(t2g)1 | face-sharing bioctahedron | Ti-Ti =3.22 Å Ti-C1(terminal) = 2.32-2.35 Å, Ti-Cl(bridge) = 2.42-2.55 Å ((NEt4+)3)3 salt)[5] | |
| [Ti2Cl10]2− | colourless | d0d0 | 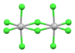
|
bioctahedral | |
| [Ti3Cl12]3- | green | (t2g)1(t2g)1(t2g)1 | face-sharing trioctahedron | Ti-Ti = 3.19, 3.10 Å (terminal) Ti-C1(terminal) = 2.36 Å (terminal), Ti-Cl(bridge) = 2.50 Å ((PPh4+)3)3 salt)[6] | |
| [TiCl6]2− | yellow | d0 | 
|
octahedral | PPh4+ salt Ti-Cl = 2.33 Å[7] |
| VCl4 | red | (t2g)1 | tetrahedral | V1−Cl = 2.29 Å | |
| V2Cl10 | violet | (t2g)0 | 
|
edge-shared bioctahedron | V1−Cl(bridging) = 2.48 Å V1−Cl(terminal) = 2.16-2.21 Å[8] |
| [VCl6]2- | red | (t2g)1 | 
|
octahedral | V1−Cl = 2.29 Å[9] |
| [CrCl6]3− | pink[10] | (t2g)3 | 
|
octahedral[11][10] | |
| [Cr2Cl9]3− | red | (d3)2 | face-sharing bioctahedron | Cr-Cl(terminal) = 2.31 Å, 2.42 (terminal) (Et2NH2+ salt)[12] | |
| [MnCl4]2−[13] | pale pink to while | (eg)2(t2g)3 | tetrahedral | Mn-Cl bond length = 2.3731-2.3830 Å[14] | |
| [MnCl6]2− | dark red | (t2g)3(eg)1 | 
|
octahedral | Mn-Cl distance = 2.28 Å K+ salt[15]) salt is isostructural with K2PtCl6 |
| [MnCl6]3− | brown[10] | (t2g)3(eg)1 | 
|
octahedral[16][10] | |
| [Mn2Cl6]2− | yellow-green | (eg)2(t2g)3 | 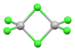
|
bitetrahedral | Mn-Cl(terminal) bond length = 2.24 Å Mn-Cl(terminal) bond length = 2.39 Å[17] (PPN+)2 salt |
| [Mn3Cl12]6− | pink | (t2g)3(eg)2 | cofacial trioctahedron | Mn-Cl distance = --- Å [(C(NH2)3]+6 salt[18] | |
| [FeCl4]2−[13] | cream | (eg)3(t2g)3 | tetrahedral((Et4N+)2 salt)[13] | ||
| [FeCl4]− | (eg)2(t2g)3 | tetrahedral | Fe-Cl bond length = 2.19 Å[19] | ||
| [FeCl6]3− | orange | (t2g)3(eg)2 | 
|
octahedral[10] | |
| [Fe2Cl6]2− | pale yellow | (eg)2(t2g)3 | 
|
bitetrahedral | Fe-Cl(terminal) bond length = 2.24 Å Fe-Cl(terminal) bond length = 2.39 Å[17] (PPN+)2 salt |
| [CoCl4]2−[13] | blue[13] | (eg)4(t2g)3 | tetrahedral | ||
| [Co2Cl6]2− | blue[17] | (eg)4(t2g)3 | 
|
bitetrahedral | Mn-Cl(terminal) bond length = 2.24 Å Co-Cl(terminal) bond length = 2.35 Å[17] (PPN+)2 salt |
| [NiCl4]2−[13] | blue[13] | (eg)4(t2g)4 | tetrahedral | Ni-Cl bond length = 2.28 Å (Et4N+)2 salt[20] | |
| [Ni3Cl12]6− | orange[21] | (t2g)6(eg)2 | confacial trioctahedral | ((Me2NH2+)2)8 salt double salt with two Cl− Ni-Cl bond length = 2.36-2.38 Å[21] | |
| [CuCl4]2−[13] | orange[22] yellow (flattened tetrahedral)[23] green (square planar)[24] |
(t2g)6(eg)3 | flattened tetrahedral or square planar[25][26] |
Cu-Cl bond length = 2.24 Å | |
| [Cu2Cl6]2− | red | [(t2g)6(eg)3]2 | edge-shared bis(square planar)[27] | Cu-Cl(terminal) = 2.24 Å Cu-Cl(bridging) = 2.31 Å | |
| [ZnCl4]2− | white/colorless | d10 | tetrahedral |
2nd row
Some homoleptic complexes of the second row transition metals feature metal-metal bonds.
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| [ZrCl6]2− | yellow | d0 | 
|
octahedral | Zr-Cl distance = 2.460 Å (Me4N+)2 salt[29] |
| [Zr2Cl10]2− | colorless | (d0)2 | 
|
edge-shared bioctahedral | Zr-Cl = 2.36 Å (terminal), 2.43 Å (bridging) N(PCl3)2)+ salt[4] |
| Nb2Cl10 | yellow | (d0)2 | 
|
edge-shared bioctahedral [Nb2Cl10] | 3.99 Å[30] |
| [NbCl6]− | yellow | d0 | 
|
octahedral | Nb-Cl = 2.34 Å N(PCl3)2)+ salt[4] |
| [Nb6Cl18]2− | black | (d2)4(d3)2 (14 cluster electrons) | 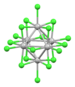
|
cluster Nb---Nb bonding | Nb-Cl = 2.92 Å (K+)2 salt[31] |
| MoCl6 | black | d0 | 
|
octahedron | Mo−Cl = 2.28 -2.31 Å[8] |
| [MoCl6]2− | yellow | (t2g)2 | 
|
octahedron | Mo−Cl = 2.37, 2.38, 2.27 Å[32] |
| [MoCl6]3− | pink | (t2g)3 | 
|
octahedral | |
| [Mo2Cl8]4− | purple[33] | 2(d4) | 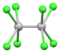
|
Mo-Mo quadruple bond | |
| [Mo2Cl9]3− | 2(d3) | face-shared bioctahedral | Mo-Mo (triple) bond length = 2.65 Å Mo-Cl (terminal) bond length = 2.38 Å Mo-Cl (bridging) bond length = 2.49 Å[34][35] | ||
| Mo2Cl10 | green | (d1)2 | 
|
edge-sharing bioctahedra[36] | |
| [Mo2Cl10]2− | (d2)2 | 
|
edge-sharing bioctahedra[37] | ||
| [Mo5Cl13]2− | brown[33] | d2d2d2d2d3 | 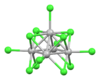
|
incomplete octahedron[38] | |
| [Mo6Cl14]2− | yellow | d4 | 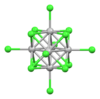
|
octahedral cluster | (4-HOPyH+)2 salt[39] |
| [TcCl6]2− | yellow | (t2g)3 | 
|
octahedron | Tc-Cl = 2.35 Å for As(C6H5)4+ salt[40] |
| [Tc2Cl8]2− | green | (t2g)4 | 
|
Tc-Tc quadruple bond | Tc-Tc = 2.16, Tc-Cl = 2.34 Å for NBu4+ salt[41] |
| [RuCl6]2− | brown | (t2g)4 | 
|
octahedral | (EtPPh3+)2 salt[42] |
| [Ru2Cl9]3− | red | [(t2g)5]2 | cofacial bioctahedral | Ru-Ru bond length = 2.71 Å; Ru-Cl(terminal) = 2.35 Å, Ru-Cl(bridging) = 2.36 Å ((Et4N)+)3 salt[43] | |
| [Ru3Cl12]4− | green | (d5)2(d6) | cofacial trioctahedral | Ru-Ru bond lengths = 2.86 Å Ru-Cl bond lengths = 2.37-2.39 Å (Et4N+)2(H7O3+)2 salt[44] | |
| [RhCl6]3− | red | (t2g)6 | 
|
octahedral | H2N+(CH2CH2NH3+)2 salt)[45] |
| [Rh2Cl9]3− | red-brown | (t2g)6 | octahedral | Rh-Cl(terminal) = 2.30 Å, Rh-Cl(terminal) = 2.40 Å ((Me3CH2Ph)+)3 salt)[34] | |
| [PdCl4]2− | brown | d8 | square planar | ||
| [Pd2Cl6]2−[46] | red ((Et4N+)2 salt) | d8 | square planar | ||
| [Pd3Cl8]2−[47] | orange brown ((Bu4N+)2 salt) | d8 | square planar | ||
| [PdCl6]2− | brown | d6 | 
|
octahedral | Pd(IV) |
| [Pd6Cl12] | yellow-brown | d8 | 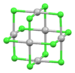
|
square planar[48] | |
| [AgCl2]− | white/colorless | d10 | linear | salt of [K(2.2.2-crypt)]+[49] | |
| [CdCl4]2− | white/colorless | d10 | tetrahedral | Et4N+ salt, Cd-Cl distance is 2.43 Å[28] | |
| [Cd2Cl6]2− | white/colorless | d10 | 
|
edge-shared bitetrahedron | (C6N3(4-C5H4N)33+ salt[50] |
| [Cd3Cl12]6− | white/colorless | d10 | octahedral (central Cd) pentacoordinate (terminal Cd's) cofactial trioctahedral |
(C6N3(4-C5H4N)33+ salt[50] (3,8-Diammonium-6-phenylphenanthridine3+)2[51] | |
| [Cd6Cl19]7− | white/colorless | d10 | 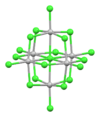
|
octahedron of octahedra | 4,4'-(C6H3(2-Et)NH3+)2 salt[52] |
3rd row
| Complex | colour | electron config. | structure | geometry | comments |
|---|---|---|---|---|---|
| [HfCl6]2− | white | d0 | 
|
octahedral | Hf-Cl distance = 2.448 A ((Me4N+)2 salt)[29] |
| [Hf2Cl10]2− | colorless/white | d0 | 
|
edge-shared bioctahedral[53] | |
| [Hf2Cl9]− | colorless/white | (d0)2 | face-shared bioctahedral[54] | ||
| [TaCl5] | white | d0 | 
|
edge-shared bioctahedral | |
| [TaCl6]− | white/colourless | d0 | 
|
octahedral | Ta-Cl = 2.34 Å (N(PCl3)2)+ salt)[4] |
| [Ta6Cl18]2- | green | d0 | 
|
octahedral | Ta-Ta = 2.34 Å (H+2 salt hexahydrate[55] |
| WCl6 | blue | d0 | 
|
octahedral | 2.24–2.26 Å[56] |
| [WCl6]2− | (t2g)2 | 
|
octahedral | W-Cl distances range from 2.34 to 2.37 Å (PPh4+ salt)[57] | |
| [WCl6]− | (t2g)1 | 
|
octahedral | W-Cl distance = 2.32 Å (Et4N+ salt)[58] | |
| W2Cl10 | black[59] | (t2g1)2 | 
|
bioctahedral | W-W distance = 3.814 Å[60] |
| [W2Cl8]4− | blue | 2(d4) | 
|
W-W quadruple bond | dW-W = 2.259 Å [Na(tmeda)+]4 salt[61] |
| [W2Cl9]2− | d3d2 | face-sharing bioctahedral | W-W distance = 2.54 Å W-Cl(terminal) = 2.36 Å, W-Cl(bridge) = 2.45 Å ((PPN+)2 salt)[62] | ||
| [W2Cl9]3− | d3d3 | octahedral | W-Cl distance = 2.32 Å (Et4N+ salt)[62] | ||
| [W3Cl13]3− | d3,d3,d4 | 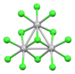
|
[W3(μ3-Cl)(μ-Cl)3Cl9]3- | W-W distances = 2.84 Å[63] | |
| [W3Cl13]2− | d3,d4,d4 | 
|
[W3(μ3-Cl)(μ-Cl)3Cl9]2-[63] | W-W distances = 2.78 Å[63] | |
| [W6Cl14]2- | yellow[64] | (d4)6 | 
|
see Mo6Cl12 | |
| [ReCl6]− | red-brown | (t2g)2 | 
|
octahedral | Re-Cl distance = 2.24-2.31 Å (PPh4+ salt)[65] |
| [ReCl6] | (t2g)1 | 
|
octahedral | Re-Cl distance = 226.3(6) Å[8] | |
| [ReCl6]2− | green | (t2g)3 | 
|
octahedral | Re-Cl distance = 2.35-2.38 Å ((PPN+)2 salt)[66] |
| [Re2Cl9]2− | (t2g)3(t2g)4 | face-sharing bioctahedral | Re-Re distance = 2.48 Å Re-Cl distances = 2.42 Å (bridge), 2.33 Å (terminal) ((Et4N+)2 salt)[67] | ||
| [Re2Cl9]− | ((t2g)3)2 | face-sharing bioctahedral | Re-Re distance = 2.70 Å Re-Cl distances = 2.41 (bridge), 2.28 Å (terminal) (Bu4N+ salt)[67] | ||
| [OsCl6]− | dark green | (t2g)3 | 
|
octahedral | dOs-Cl = 2.30 Å for Et4N+[68] and Ph4P+[69] salts |
| [OsCl6]2− | yellow-orange | (t2g)4 | 
|
octahedral[69] | Os-Cl distance 2.33 Å |
| [Os2Cl8]2− | green | (d5)2 | 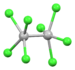
|
square antiprism | dOs-Os = 2.182 Å, dOs-Cl = 2.32 Å (Bu4N+)2 salt[70] |
| [Os2Cl10]2− | green | (d4)2 | 
|
octahedral | dOs-Cl(terminal) = 2.30 Å dOs-Cl(bridging) = 2.42 Å (Et4N+)2 salt[68] |
| [IrCl6]3− | red | (t2g)6 | 
|
octahedral | Ir-Cl = 2.36 Å[71] |
| [IrCl6]2− | brown | (t2g)5 | 
|
octahedral | Ir-Cl = 2.33 Å[72] |
| [Ir2Cl9]3− | - | ((t2g)6)2 | bi-octahedral[73] | ||
| [PtCl4]2− | pink | d8 | square planar | ||
| [PtCl6]2− | yellow | d6 | 
|
octahedral | Pt-Cl distance = 2.32 Å Et4N+ salt, ((Me4N+)2 salt)[29] |
| [Pt2Cl9]− | red (Bu4N+ salt) | ((t2g)6)2 | octahedral | Pt-Clt and Pt-Clbridge = 2.25, 2.38 Å[74] | |
| [Pt2Cl10]2− | yellow-brown (PPN+ salt) | ((t2g)6)2 | 
|
edge-shared bioctahedral | Pt-Clt and Pt-Clbridge = 2.27, 2.37 Å[74] |
| [Pt6Cl12] | yellow-brown | (d8)6 | 
|
square planar | Pt-Cl = 2.31[75] |
| [AuCl2]− | white/colorless | d10 | linear | Au-Cl distances of 2.28 Å NEt4+ salt[76] | |
| Au 4Cl 8 |
black | (d10)2(d8)2 | 
|
linear and square planar | rare example of mixed valence, molecular chloride[77] |
| [AuCl4]− | yellow | d8 | square planar | Au-Cl distances of 2.26 Å NBu4+ salt[78] | |
| [HgCl4]2− | white/colorless | d10 | tetrahedral | Hg-Cl distance is 2.46 Å[28] Et4N+ salt | |
| [Hg2Cl6]2− | white/colorless | d10 | 
|
edge-shared bitetrahedral | Hg-Cl distance is 2.46 Å[79] Bu4N+ salt |
Heteroleptic complexes
Heteroleptic complexes containing chloride are numerous. Most hydrated metal halides are members of this class. Hexamminecobalt(III) chloride and Cisplatin (cis-Pt(NH3)2Cl2) are prominent examples of metal-ammine-chlorides.
Hydrates

As indicated in the table below, many hydrates of metal chlorides are molecular complexes.[80][81] These compounds are often important commercial sources of transition metal chlorides. Several hydrated metal chlorides are not molecular and thus are not included in this tabulation. For example the dihydrates of manganese(II) chloride, nickel(II) chloride, copper(II) chloride, iron(II) chloride, and cobalt(II) chloride are coordination polymers.
| Formula of hydrated metal halides |
Coordination sphere of the metal |
|---|---|
| TiCl3(H2O)6 | trans-[TiCl2(H2O)4]+[82] |
| VCl3(H2O)6 | trans-[VCl2(H2O)4]+[82] |
| CrCl3(H2O)6 | trans-[CrCl2(H2O)4]+ |
| CrCl3(H2O)6 | [CrCl(H2O)5]2+ |
| CrCl2(H2O)4 | trans-[CrCl2(H2O)4] |
| CrCl3(H2O)6 | [Cr(H2O)6]3+[83] |
| MnCl2(H2O)6 | trans-[MnCl2(H2O)4] |
| MnCl2(H2O)4 | cis-[MnCl2(H2O)4][84] |
| FeCl2(H2O)6 | trans-[FeCl2(H2O)4] |
| FeCl2(H2O)4 | trans-[FeCl2(H2O)4] |
| FeCl3(H2O)6 | one of four hydrates of ferric chloride,[85] |
| FeCl3(H2O)2.5 | cis-[FeCl2(H2O)4]+[86] |
| CoCl2(H2O)6 | trans-[CoCl2(H2O)4] |
| CoCl2(H2O)4 | cis-[CoCl2(H2O)4] |
| NiCl2(H2O)6 | trans-[NiCl2(H2O)4] |
| NiCl2(H2O)4 | cis-[NiCl2(H2O)4] |
Adducts
Metal chlorides form adducts with ethers to give transition metal ether complexes.
References
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ J. F. Hartwig (2009). "4: Covalent (X-Type) Ligands Bound Through Metal-Heteroatom Bonds". Organotransition Metal Chemistry. University Science Books. ISBN 978-1-891389-53-5.
- ↑ Hatfield, William E.; Fay, Robert C.; Pfluger, C. E.; Piper, T. S. (1963). "Hexachlorometallates of Trivalent Chromium, Manganese and Iron". Journal of the American Chemical Society 85 (3): 265–269. doi:10.1021/ja00886a003.
- ↑ 4.0 4.1 4.2 4.3 Rivard, Eric; McWilliams, Andrew R.; Lough, Alan J.; Manners, Ian (2002). "Bis(trichlorophosphine)iminium salts, [Cl3P=N=PCl3]+, with transition metal halide counter-ions". Acta Crystallographica Section C 58 (9): i114–i118. doi:10.1107/S0108270102012532. PMID 12205363.
- ↑ Castro, Stephanie L.; Streib, William E.; Huffmann, John C.; Christou, George (1996). "A mixed-valence (TiIIITiIV) Carboxylate Complex: Crystal Structures and Properties of [Ti2OCl3(O2CPh)2(THF)3] and [NEt4]3[Ti2Cl9]". Chemical Communications (18): 2177. doi:10.1039/CC9960002177.
- ↑ Chen, Linfeng; Cotton, F. Albert (1998). "Synthesis, Reactivity, and X-ray Structures of Face-Sharing Ti(III) Complexes; the New Trinuclear Ion, [Ti3Cl12]3−". Polyhedron 17 (21): 3727–3734. doi:10.1016/S0277-5387(98)00171-5.
- ↑ Chen, Linfeng; Cotton, F. A. (1998). "Partial Hydrolysis of Ti(III) and Ti(IV) Chlorides in the Presence of [PPh4]Cl". Inorganica Chimica Acta 267 (2): 271–279. doi:10.1016/S0020-1693(97)05766-6.
- ↑ 8.0 8.1 8.2 Tamadon, Farhad; Seppelt, K. (2012). "The Elusive Halides VCl5, MoCl6, and ReCl6". Angewandte Chemie International Edition 52 (2): 767–769. doi:10.1002/anie.201207552. PMID 23172658.
- ↑ Hayton, Trevor W.; Patrick, Brian O.; Legzdins, Peter (2004). "New Details Concerning the Reactions of Nitric Oxide with Vanadium Tetrachloride". Inorganic Chemistry 43 (22): 7227–7233. doi:10.1021/ic0491534. PMID 15500362.
- ↑ 10.0 10.1 10.2 10.3 10.4 Hatfield, William E.; Fay, Robert C.; Pfluger, C. E.; Piper, T. S. (1963). "Hexachlorometallates of Trivalent Chromium, Manganese and Iron". Journal of the American Chemical Society 85 (3): 265–269. doi:10.1021/ja00886a003.
- ↑ O. S. Filipenko, D. D. Makitova, O. N. Krasochka, V. I. Ponomarev, L. O. Atovmyan (1987). Koord. Khim. 13: 669.
- ↑ Dyer, Philip W.; Gibson, Vernon C.; Jeffery, John C. (1995). "Unexpected Synthesis of a Binuclear Chromium(III) salt Exhibiting NH…Cl Hydrogen-Bonding Interactions". Polyhedron 14 (20–21): 3095–3098. doi:10.1016/0277-5387(95)00089-B.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 13.6 13.7 Gill, N. S.; Taylor, F. B. (1967). Tetrahalo Complexes of Dipositive Metals in the First Transition Series. Inorganic Syntheses. 9. pp. 136–142. doi:10.1002/9780470132401.ch37. ISBN 978-0-470-13240-1.
- ↑ Chang, Jui-Cheng; Ho, Wen-Yueh; Sun, I-Wen; Chou, Yu-Kai; Hsieh, Hsin-Hsiu; Wu, Tzi-Yi (2011). "Synthesis and Properties of New Tetrachlorocobaltate (II) and Tetrachloromanganate (II) Anion Salts with Dicationic Counterions". Polyhedron 30 (3): 497–507. doi:10.1016/j.poly.2010.11.009.
- ↑ Moews, P. C. (1966). "The Crystal Structure, Visible, and Ultraviolet Spectra of Potassium Hexachloromanganate(IV)". Inorganic Chemistry 5: 5–8. doi:10.1021/ic50035a002.
- ↑ O. S. Filipenko, D. D. Makitova, O. N. Krasochka, V. I. Ponomarev, L. O. Atovmyan (1987). Koord. Khim. 13: 669.
- ↑ 17.0 17.1 17.2 17.3 Sun, Jui-Sui; Zhao, Hanhua; Ouyang, Xiang; Clérac, Rodolphe; Smith, Jennifer A.; Clemente-Juan, Juan M.; Gómez-Garcia, Carlos; Coronado, Eugenio et al. (1999). "Structures, Magnetic Properties, and Reactivity Studies of Salts Containing the Dinuclear Anion [M2Cl6]2-(M = Mn, Fe, Co)". Inorganic Chemistry 38 (25): 5841–5855. doi:10.1021/ic990525w.
- ↑ Sen, Abhijit; Swain, Diptikanta; Guru Row, Tayur N.; Sundaresan, A. (2019). "Unprecedented 30 K Hysteresis Across Switchable Dielectric and Magnetic Properties in a Bright Luminescent Organic–Inorganic Halide (CH6N3)2MnCl4". Journal of Materials Chemistry C 7 (16): 4838–4845. doi:10.1039/C9TC00663J. http://eprints.iisc.ac.in/62693/1/Jou_Mat_Che_7-16_4838-4845.pdf.
- ↑ Lutz, Martin; Huang, Yuxing; Moret, Marc-Etienne; Klein Gebbink, Robertus J. M. (2014). "Phase Transitions and Twinned Low-Temperature Structures of Tetraethylammonium Tetrachloridoferrate(III)". Acta Crystallographica Section C 70 (5): 470–476. doi:10.1107/S2053229614007955. PMID 24816016.
- ↑ Stucky, G. D.; Folkers, J. B.; Kistenmacher, T. J. (1967). "The Crystal and Molecular Structure of Tetraethylammonium Tetrachloronickelate(II)". Acta Crystallographica 23 (6): 1064–1070. doi:10.1107/S0365110X67004268.
- ↑ 21.0 21.1 Gerdes, Allison; Bond, Marcus R. (2009). "Octakis(dimethylammonium) Hexa-μ2-chlorido-Hexachloridotrinickelate(II) Dichloride: A Linear Trinickel Complex with Asymmetric Bridging". Acta Crystallographica Section C 65 (10): m398–m400. doi:10.1107/S0108270109036853. PMID 19805875.
- ↑ Mahoui, A.; Lapasset, J.; Moret, J.; Saint Grégoire, P. (1996). "Tetraethylammonium Tetramethylammonium Tetrachlorocuprate(II), [(C2H5)4N] [(CH3)4N] [CuCl4]". Acta Crystallographica Section C 52 (11): 2674–2676. doi:10.1107/S0108270196009031.
- ↑ Guillermo Mínguez Espallargas; Lee Brammer; Jacco van de Streek; Kenneth Shankland; Alastair J. Florence; Harry Adams (2006). "Reversible Extrusion and Uptake of HCl Molecules by Crystalline Solids Involving Coordination Bond Cleavage and Formation". J. Am. Chem. Soc. 128 (30): 9584–9585. doi:10.1021/ja0625733. PMID 16866484.
- ↑ Kelley, A.; Nalla, S.; Bond, M. R. (2015). "The Square-Planar to Flattened-Tetrahedral CuX42- (X = Cl, Br) Structural Phase Transition in 1,2,6-Trimethylpyridinium salts". Acta Crystallographica Section B 71 (Pt 1): 48–60. doi:10.1107/S205252061402664X. PMID 25643715.
- ↑ Halcrow, Malcolm A. (2013). "Jahn–Teller Distortions in Transition Metal Compounds, and Their Importance in Functional Molecular and Inorganic Materials". Chemical Society Reviews 42 (4): 1784–1795. doi:10.1039/C2CS35253B. PMID 22968285. http://eprints.whiterose.ac.uk/77270/7/halcrow1.pdf.
- ↑ Reinen, Dirk (2014). "A New Approach to Treating Vibronic Coupling Under Stress—The Strain-Induced Enhancement or Suppression of Jahn–Teller Distortions in Tetrahedral CuIICl4-Complexes, and the Transition to Octahedral Structures". Coordination Chemistry Reviews 272: 30–47. doi:10.1016/j.ccr.2014.03.004.
- ↑ Willett, Roger D.; Butcher, Robert E.; Landee, Christopher P.; Twamley, Brendan (2006). "Two Halide Exchange in Copper(II) Halide Dimers: (4,4′-Bipyridinium)Cu2Cl6−x BRX". Polyhedron 25 (10): 2093–2100. doi:10.1016/j.poly.2006.01.005.
- ↑ 28.0 28.1 28.2 Mahoui, A.; Lapasset, J.; Moret, J.; Saint Grégoire, P. (1996). "Bis(tetraethylammonium) Tetrachlorometallates, [(C2H5)4N]2[MCl4], where M = Hg, Cd, Zn". Acta Crystallographica Section C 52 (11): 2671–2674. doi:10.1107/S010827019600666X.
- ↑ 29.0 29.1 29.2 Autillo, Matthieu; Wilson, Richard E. (2017). "Phase Transitions in Tetramethylammonium Hexachlorometalate Compounds (TMA)2MCl6 (M = U, Np, Pt, Sn, Hf, Zr)". European Journal of Inorganic Chemistry 2017 (41): 4834–4839. doi:10.1002/ejic.201700764.
- ↑ Cotton, F.A., P. A. Kibala, M. Matusz and R. B. W. Sandor (1991). "Structure of the Second Polymorph of Niobium Pentachloride". Acta Crystallogr. C 47 (11): 2435–2437. doi:10.1107/S0108270191000239.
- ↑ Simon, Arndt; von Schnering, Hans-Georg; Schäfer, Harald (1968). "Beiträge zur Chemie der Elemente Niob und Tantal. LXIX K4Nb6Cl18 Darstellung, Eigenschaften und Struktur". Zeitschrift für anorganische und allgemeine Chemie 361 (5–6): 235–248. doi:10.1002/zaac.19683610503.
- ↑ Rabe, Susanne; Bubenheim, Wilfried; Müller, Ulrich (2004). "Crystal Structures of Acetonitrile Solvates of Bis(tetraphenylphosphonium) Tetrachlorooxovanadate(IV), Hexachlorostannate(IV) and -Molybdate(IV), [P(C6H5)4]2[VOCl4] · 4CH3CN, [P(C6H5)4]2[MCl6]·4CH3CN (M = Sn, Mo)". Zeitschrift für Kristallographie - New Crystal Structures 219 (2): 101–105. doi:10.1524/ncrs.2004.219.2.101.
- ↑ 33.0 33.1 Brignole, A. B.; Cotton, F. A.; Dori, Z. (1972). "Rhenium and Molybdenum Compounds Containing Quadruple Bonds". Inorganic Syntheses. 13. 81–89. doi:10.1002/9780470132449.ch15. ISBN 978-0-470-13244-9.
- ↑ 34.0 34.1 Cotton, F.A; Ucko, David A. (1972). "The Structure of Trimethylphenylammonium Nonachlorodirhodate(III) and a Survey of Metal-Metal Interactions in Confacial Bioctahedra". Inorganica Chimica Acta 6: 161–172. doi:10.1016/S0020-1693(00)91778-X.
- ↑ R. A. D. Wentworth, R. Saillant, R. B. Jackson, W. E. Streib, K. Folting (1971). "Crystal structures of Cs3Cr2Br9, Cs3Mo2Cl9, and Cs3Mo2Br9". Inorg. Chem. 10 (7): 1453–1457. doi:10.1021/ic50101a027.
- ↑ Beck, J.; Wolf, F. (1997). "Three New Polymorphic Forms of Molybdenum Pentachloride". Acta Crystallogr. B53 (6): 895–903. doi:10.1107/S0108768197008331.
- ↑ Hey, E.; Weller, F.; Dehnicke, K. (1984). "Synthese und Kristallstruktur von (PPh4)2[Mo2Cl10]". Zeitschrift für anorganische und allgemeine Chemie 508: 86–92. doi:10.1002/zaac.19845080113.
- ↑ Ahmed, Ejaz; Ruck, Michael (2011). "Chemistry of Polynuclear Transition-Metal Complexes in Ionic Liquids". Dalton Transactions 40 (37): 9347–57. doi:10.1039/c1dt10829h. PMID 21743925. http://tud.qucosa.de/id/qucosa%3A27774.
- ↑ Kei Inumaru, Takashi Kikudome, Hiroshi Fukuoka, Shoji Yamanaka (2008). "Reversible Emergence of a Self-Assembled Layered Structure From Three-Dimensional Isotropic Ionic Crystal of a Cluster Compound (4-HNC5H4OH)2Mo6Cl14 Driven By Absorption of Water and Alcohols". J. Am. Chem. Soc. 130 (31): 10038–10039. doi:10.1021/ja802752y. PMID 18613684.
- ↑ Baldas, J.; Bonnyman, J.; Samuels, D. L.; Williams, G. A. (1984). "Structure Studies of Technetium Complexes. VII. Structure of Tetraphenylarsonium Hexachlorotechnetate(IV), [As(C6H5)4]2[TcCl6]". Acta Crystallographica Section C Crystal Structure Communications 40 (8): 1343–1346. doi:10.1107/S0108270184007903.
- ↑ Poineau, Frederic; Johnstone, Erik V.; Forster, Paul M.; Ma, Longzou; Sattelberger, Alfred P.; Czerwinski, Kenneth R. (2012). "Probing the Presence of Multiple Metal–Metal Bonds in Technetium Chlorides by X-ray Absorption Spectroscopy: Implications for Synthetic Chemistry". Inorganic Chemistry 51 (17): 9563–9570. doi:10.1021/ic3014859. PMID 22906536.
- ↑ Sharutin, V. V.; Sharutina, O. K.; Senchurin, V. S.; Andreev, P. V. (2018). "Synthesis and Structure of Ruthenium Complexes [Ph3PR]+2[RuCl6]2− (R = C2H5, CH=CHCH3, CH2CH=CHCH3, CH2OCH3), and [Ph3PCH2CH CHCH2PPh3]2+2[Ru2Cl10O]4− · 4H2O". Russian Journal of Inorganic Chemistry 63 (9): 1178–1185. doi:10.1134/S0036023618090188.
- ↑ I. A. Efimenko, T. A. Balakaeva, A. P. Kurbakova, A. S. Kanishcheva, A. V. Chuvaev, V. M. Stepanovich, Yu. N. Mikhailov (1992). Zh. Neorg. Khim. (Russ. J. Inorg. Chem.) 37: 1312.
- ↑ Bino, Avi; Cotton, F. Albert (1980). "A Linear, Trinuclear, Mixed-Valence Chloro Complex of Ruthenium, [Ru3Cl12]4-". Journal of the American Chemical Society 102 (2): 608–611. doi:10.1021/ja00522a027.
- ↑ Frank, Walter; Reiß, Guido J.; Kleinwächter, Ingo (1996). "Spezielle Alkylammoniumhexachlorometallate. I. Kristallisationsverhalten und Kristallstruktur von Diethylentriammoniumhexachlororhodat, [H3N(CH2)2NH2(CH2)2NH3] [RhCl6]". Zeitschrift für anorganische und allgemeine Chemie 622 (4): 729–733. doi:10.1002/zaac.19966220428.
- ↑ Schwarz, Simon; Strähle, Joachim; Weisser, Ulrike (2002). "Synthese und Struktur der Komplexe [(n-Bu)4N]2[{(THF)Cl4Re≡N}2PdCl2], [Ph4P]2[(THF)Cl4Re≡N-Pd Cl(μ-Cl)]2 und [(n-Bu)4N]2[Pd3Cl8]". Zeitschrift für anorganische und allgemeine Chemie 628 (11): 2495–2499. doi:10.1002/1521-3749(200211)628:11<2495::AID-ZAAC2495>3.0.CO;2-G.
- ↑ Fábry, Jan; Dušek, Michal; Fejfarová, Karla; Krupková, Radmila; Vaněk, Přemysl; Němec, Ivan (2004). "Two Phases of Bis(tetraethylammonium) Di-μ-chloro-bis[dichloropalladium(II)]". Acta Crystallographica Section C Crystal Structure Communications 60 (9): m426–m430. doi:10.1107/S0108270104016725. PMID 15345822.
- ↑ Dell'Amico, Daniela Belli; Calderazzo, Fausto; Marchetti, Fabio; Ramello, Stefano (1996). "Molecular Structure of[Pd6Cl12] in Single Crystals Chemically Grown at Room Temperature". Angewandte Chemie International Edition in English 35 (12): 1331–1333. doi:10.1002/anie.199613311.
- ↑ Helgesson, Goeran; Jagner, Susan (1991). "Halogenoargentate(I) with unusual coordination geometries. Synthesis and Structure of Potassium-crypt Salts of Chloro-, Bromo- and Iodoargentates(I), Including the First Example of a Two-Coordinated Chloroargentate(I) in the Solid State". Inorganic Chemistry 30 (11): 2574–2577. doi:10.1021/ic00011a024.
- ↑ 50.0 50.1 Hao, Pengfei; Guo, Chunyu; Shen, Junju; Fu, Yunlong (2019). "A Novel Photochromic Hybrid Containing Trinuclear [Cd3Cl12]6− Clusters and Protonated Tripyridyl-Triazines". Dalton Transactions 48 (44): 16497–16501. doi:10.1039/C9DT03494C. PMID 31559400.
- ↑ Costin-Hogan, Christina E.; Chen, Chun-Long; Hughes, Emma; Pickett, Austin; Valencia, Richard; Rath, Nigam P.; Beatty, Alicia M. (2008). ""Reverse" engineering: Toward 0-D Cadmium Halide clusters". CrystEngComm 10 (12): 1910. doi:10.1039/b812504j.
- ↑ Chen, Chun-Long; Beatty, Alicia M. (2007). "From Crystal Engineering to Cluster Engineering: How to Transform Cadmium Chloride from 2-D to 0-D". Chemical Communications (1): 76–78. doi:10.1039/B613761J. PMID 17279266.
- ↑ Neumüller, Bernhard; Dehnicke, Kurt (2004). "Die Kristallstrukturen von (Ph4P)2[HfCl6]2CH2Cl2 und (Ph4P)2[Hf2Cl10]CH2Cl2". Zeitschrift für anorganische und allgemeine Chemie 630 (15): 2576–2578. doi:10.1002/zaac.200400370.
- ↑ Dötterl, Matthias; Haas, Isabelle; Alt, Helmut G. (2011). "Solubility Behaviour of TiCl4, ZrCl4, and HfCl4 in Chloroaluminate Ionic Liquids". Zeitschrift für anorganische und allgemeine Chemie 637 (11): 1502–1506. doi:10.1002/zaac.201100244.
- ↑ Jacobson, Robert A.; Thaxton, Charles B. (1971). "Crystal structure of H2[Ta6Cl18].6H2O". Inorganic Chemistry 10 (7): 1460–1463. doi:10.1021/ic50101a029.
- ↑ J. C. Taylor; P. W. Wilson (1974). "The Structure of β-Tungsten Hexachloride by Powder Neutron and X-ray Diffraction". Acta Crystallographica B30 (5): 1216–1220. doi:10.1107/S0567740874004572.
- ↑ Lau, C.; Dietrich, A.; Plate, M.; Dierkes, P.; Neumüller, B.; Wocadlo, S.; Massa, W.; Harms, K. et al. (2003). "Die Kristallstrukturen der Hexachlorometallate NH4[SbCl6], NH4[WCl6], [K(18-Krone-6)(CH2Cl2)]2[WCl6]·6CH2Cl2 und (PPh4)2[WCl6]·4CH3CN". Zeitschrift für anorganische und allgemeine Chemie 629 (3): 473–478. doi:10.1002/zaac.200390078.
- ↑ Eichler, W.; Seifert, H.-J. (1977). "Strukturelle und magnetische Untersuchungen an Hexachlorowolframaten(V)". Zeitschrift für anorganische und allgemeine Chemie 431: 123–133. doi:10.1002/zaac.19774310112.
- ↑ McCann, III, E. L.; Brown, T. M. (1972). Tungsten(V) Chloride. Inorganic Syntheses. XIII. p. 150-154. doi:10.1002/9780470132449.ch29.
- ↑ Cotton, F. A.; Rice, C. E. (1978). "Tungsten Pentachloride". Acta Crystallogr. B34 (9): 2833-2834. doi:10.1107/S0567740878009322.
- ↑ Cotton, F. Albert; Mott, Graham N.; Schrock, Richard R.; Sturgeoff, Lynda G. (1982). "Preparation and Characterization of a Compound Containing the Octachloroditungstate (Tungsten-Tungsten Quadruple Bond) Ion, [W2Cl8]4-". Journal of the American Chemical Society 104 (24): 6781–6782. doi:10.1021/ja00388a050.
- ↑ 62.0 62.1 Cotton, F. Albert; Falvello, Larry R.; Mott, Graham N.; Schrock, Richard R.; Sturgeoff, Lynda G. (1983). "Structural Characterization of the Nonachloroditungsten(II, III) Ion". Inorganic Chemistry 22 (18): 2621–2623. doi:10.1021/ic00160a031.
- ↑ 63.0 63.1 63.2 Kolesnichenko, Vladimir; Luci, Jeffrey J.; Swenson, Dale C.; Messerle, Louis (1998). "W3(μ3-Cl)(μ-Cl)3Cl9n-(n= 2, 3), Discrete Monocapped Tritungsten Clusters Derived from a New Binary Tungsten Chloride, W3Cl10: Effect of Electron Count on Bonding in Isostructural Triangulo M3X13 Clusters1". Journal of the American Chemical Society 120 (50): 13260–13261. doi:10.1021/ja9831958.
- ↑ Kolesnichenko, Vladimir; Messerle, Louis (1998). "Facile Reduction of Tungsten Halides with Nonconventional, Mild Reductants. 2. Four Convenient, High-Yield Solid-State Syntheses of the Hexatungsten Dodecachloride Cluster W6Cl12 and Cluster Acid (H3O)2[W6(μ3-Cl)8Cl6](OH2)x, Including New Cation-Assisted Ternary Routes". Inorganic Chemistry 37 (15): 3660–3663. doi:10.1021/ic980232n. PMID 11670462.
- ↑ Arp, O.; Preetz, W. (1994). "Darstellung, Schwingungsspektren und Normalkoordinatenanalyse von Hexachlororhenat(V) sowie Kristallstruktur von [P(C6H5)4] [ReCl6]". Zeitschrift für anorganische und allgemeine Chemie 620 (8): 1391–1396. doi:10.1002/zaac.19946200811.
- ↑ Chau, C.-N.; Wardle, R. W. M.; Ibers, J. A. (1988). "Structure of Di[bis(triphenylphosphine)iminium] Hexachlororhenate(IV)". Acta Crystallographica Section C 44 (4): 751–753. doi:10.1107/S0108270187011910.
- ↑ 67.0 67.1 Heath, Graham A.; McGrady, John E.; Raptis, Raphael G.; Willis, Anthony C. (1996). "Valence-Dependent Metal−Metal Bonding and Optical Spectra in Confacial Bioctahedral [Re2Cl9]z- (z= 1, 2, 3). Crystallographic and Computational Characterization of [Re2Cl9]−and [Re2Cl9]2-". Inorganic Chemistry 35 (23): 6838–6843. doi:10.1021/ic951604k. PMID 11666851.
- ↑ 68.0 68.1 Krebs, B.; Henkel, G.; Dartmann, M.; Preetz, W.; Bruns, M. (1984). "Reaktionen und Strukturen von [(C2H5)4N] [OsCl6] und [(n-C4H9)4N]2[Os2Cl10]". Z. Naturforsch. 39 (7): 843. doi:10.1515/znb-1984-0701.
- ↑ 69.0 69.1 Kim, Eunice E.; Eriks, Klaas; Magnuson, Roy (1984). "Crystal Structures of the Tetraphenylphosphonium salts of Hexachloroosmate(V) and Hexachloroosmate(IV), [(C6H5)4P]OsCl6 and [(C6H5)4P]2OsCl6". Inorganic Chemistry 23 (4): 393–397. doi:10.1021/ic00172a003.
- ↑ Agaskar, Pradyot A.; Cotton, F. Albert.; Dunbar, Kim R.; Falvello, Larry R.; Tetrick, Stephen M.; Walton, Richard A. (1986). "The Multiply Bonded Octahalodiosmate(III) Anions. 2. Structure and Bonding". Journal of the American Chemical Society 108 (16): 4850–4855. doi:10.1021/ja00276a024.
- ↑ Rankin, DA; Penfold, BR; Fergusson, JE (1983). "The Chloro and Bromo Complexes of Iridium(III) and Iridium(IV). II. Structural Chemistry of IrIII Complexes". Australian Journal of Chemistry 36 (5): 871. doi:10.1071/CH9830871.
- ↑ Sanchis-Perucho, Adrián; Martínez-Lillo, José (2019). "Ferromagnetic Exchange Interaction in a New Ir(IV)–Cu(II) Chain Based on the Hexachloroiridate(IV) Anion". Dalton Transactions 48 (37): 13925–13930. doi:10.1039/C9DT02884F. PMID 31411207.
- ↑ Yellowlees, L.; Elliot, M.; Parsons, S.; Messenger, D.. "MASNEA". Cambridge Crystallographic Database CCDC 278284.
- ↑ 74.0 74.1 Belli Dell'Amico, Daniela; Calderazzo, Fausto; Marchetti, Fabio; Ramello, Stefano; Samaritani, Simona (2008). "Simple Preparations of Pd6Cl12, Pt6Cl12, and Qn[Pt2Cl8+n],n= 1, 2 (Q = TBA+, PPN+) and Structural Characterization of [TBA] [Pt2Cl9] and [PPN]2[Pt2Cl10]·C7H8". Inorganic Chemistry 47 (3): 1237–1242. doi:10.1021/ic701932u. PMID 18166044.
- ↑ von Schnering, Hans Georg; Chang, Jen-Hui; Peters, Karl; Peters, Eva-Maria; Wagner, Frank R.; Grin, Yuri; Thiele, Gerhard (2003). "Structure and Bonding of the Hexameric Platinum(II) Dichloride, Pt6Cl12 (β-PtCl2)". Zeitschrift für Anorganische und Allgemeine Chemie 629 (3): 516–522. doi:10.1002/zaac.200390084.
- ↑ Helgesson, Göran; Jagner, Susan; Vicentini, G.; Rodellas, C.; Niinistö, L. (1987). "Crystal Structures of Tetraethylammonium Dichloroaurate(I) and Tetraethylammonium Diiodoaurate(I)". Acta Chemica Scandinavica 41a: 556–561. doi:10.3891/acta.chem.scand.41a-0556.
- ↑ Dell'Amico, Daniela Belli; Calderazzo, Fausto; Marchetti, Fabio; Merlino, Stefano; Perego, Giovanni (1977). "X-Ray crystal and molecular structure of Au4Cl8, the product of the reduction of Au2Cl6 by Au(CO)Cl". Journal of the Chemical Society, Chemical Communications (1): 31. doi:10.1039/C39770000031.
- ↑ Buckley, Robbie W.; Healy, Peter C.; Loughlin, Wendy A. (1997). "Reduction of [NBu4] [AuCl4] to [NBu4] [AuCl2] with Sodium Acetylacetonate". Australian Journal of Chemistry 50 (7): 775. doi:10.1071/C97029.
- ↑ Goggin, Peter L.; King, Paul; McEwan, David M.; Taylor, Graham E.; Woodward, Peter; Sandström, Magnus (1982). "Vibrational Spectroscopic Studies of Tetra-n-butylammonium Trihalogenomercurates; Crystal Structures of [NBun4](HgCl3) and [NBun4(HgI3)". Journal of the Chemical Society, Dalton Transactions (5): 875–882. doi:10.1039/dt9820000875.
- ↑ Waizumi, K.; Masuda, H.; Ohtaki, H. (1992). "X-ray Structural Studies of FeBr2 • 4H2O, CoBr2 • 4H2O, NiCl2 • 4H2O, and CuBr2 • 4H2O. cis/trans Selectivity in Transition Metal(II) dihalide Tetrahydrate". Inorganica Chimica Acta 192: 173–181. doi:10.1016/S0020-1693(00)80756-2.
- ↑ Morosin, B. (1967). "An X-ray Diffraction Study on Nickel(II) Chloride Dihydrate". Acta Crystallographica 23 (4): 630–634. doi:10.1107/S0365110X67003305.
- ↑ 82.0 82.1 Donovan, William F.; Smith, Peter W. (1975). "Crystal and Molecular Structures of Aquahalogenovanadium(III) Complexes. Part I. X-Ray Crystal Structure of trans-Tetrakisaquadibromo-Vanadium(III) Bromide Dihydrate and the Isomorphous Chloro- Compound". Journal of the Chemical Society, Dalton Transactions (10): 894. doi:10.1039/DT9750000894.
- ↑ Andress, K.R.; Carpenter, C. "Kristallhydrate. II.Die Struktur von Chromchlorid- und Aluminiumchloridhexahydrat" Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie 1934, volume 87, p446-p463.
- ↑ Zalkin, Allan; Forrester, J. D.; Templeton, David H. (1964). "Crystal structure of manganese dichloride tetrahydrate". Inorganic Chemistry 3 (4): 529–33. doi:10.1021/ic50014a017. http://www.escholarship.org/uc/item/7vf7p79j.
- ↑ Lind, M. D. (1967). "Crystal Structure of Ferric Chloride Hexahydrate". The Journal of Chemical Physics 47 (3): 990–993. doi:10.1063/1.1712067. Bibcode: 1967JChPh..47..990L.
- ↑ Simon A. Cotton (2018). "Iron(III) chloride and its coordination chemistry". Journal of Coordination Chemistry 71 (21): 3415–3443. doi:10.1080/00958972.2018.1519188.
 |

