Chemistry:Tungsten(V) chloride
| |
| Names | |
|---|---|
| Other names
tungsten pentachloride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| W2Cl10 | |
| Molar mass | 361.1 g/mol |
| Appearance | black crystals hygroscopic |
| Density | 3.86 g/cm3 |
| Melting point | 248 °C (478 °F; 521 K) |
| Boiling point | 275.6 °C (528.1 °F; 548.8 K) |
| +387.0·10−6 cm3/mol | |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | Danger |
| H302, H314 | |
| Related compounds | |
Related compounds
|
Tungsten(IV) chloride Tungsten hexachloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tungsten(V) chloride is an inorganic compound with the formula W2Cl10. This compound is analogous in many ways to the more familiar molybdenum pentachloride.
Synthesis
The material is prepared by reduction of tungsten hexachloride. One method involves the use of tetrachloroethylene as the reductant[2]
- 2 WCl6 + C2Cl4 → W2Cl10 + C2Cl6
The blue green solid is volatile under vacuum and slightly soluble in nonpolar solvents. The compound is oxophilic and is highly reactive toward Lewis bases.
When the same reduction is conducted in the presence of tetraphenylarsonium chloride, one obtains instead the hexachlorotungstate(V) salt:[3]
- 2 WCl
6 + Cl
2C=CCl
2 + 2 (C
6H
5)
4AsCl → 2 (C
6H
5)
4As[WCl
6] + Cl
3C–CCl
3
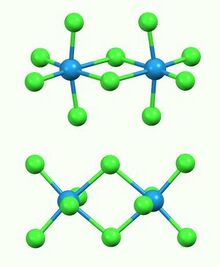
Structure
The compound exists as a dimer, with a pair of octahedral tungsten(V) centres bridged by two chloride ligands. The W---W separation is 3.814 Å, which is non-bonding. The compound is isostructural with Nb2Cl10 and Mo2Cl10. The compound evaporates to give trigonal bipyramidal WCl5 monomers.[4]
References
- ↑ "C&L Inventory". https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/244169.
- ↑ McCann, III, E. L.; Brown, T. M. (1972). Tungsten(V) Chloride. Inorganic Syntheses. XIII. pp. 150–154. doi:10.1002/9780470132449.ch29.
- ↑ Uhl, G.; Hey, E.; Becker, G.; Weller, F.; Dehnicke, K. (1983). "Über die Reaktion von 2,2-Dimethylpropylidinphosphan mit Wolframhexachlorid; die Kristallstrukturen von [(Cl3PO)WCL4(H9C4CCC4H9)] und [(H5C6)4As][WCL6]". Zeitschrift für anorganische und allgemeine Chemie 497 (2): 213–223. doi:10.1002/zaac.19834970221. http://nbn-resolving.de/urn:nbn:de:bsz:93-opus-57717.
- ↑ Cotton, F. A.; Rice, C. E. (1978). "Tungsten Pentachloride". Acta Crystallogr. B34 (9): 2833–2834. doi:10.1107/S0567740878009322.
 |

