Chemistry:Tungsten hexachloride
 α-Tungsten hexachloride
| |||
| Error creating thumbnail: Unable to save thumbnail to destination β-Tungsten hexachloride
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Tungsten hexachloride
Tungsten(VI) chloride | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| |||
| |||
| Properties | |||
| WCl 6 | |||
| Molar mass | 396.54 g·mol−1 | ||
| Appearance | dark blue crystals, moisture sensitive | ||
| Density | 3.52 g/cm3 | ||
| Melting point | 275 °C (527 °F; 548 K) | ||
| Boiling point | 346.7 °C (656.1 °F; 619.8 K) | ||
| Hydrolyzes | |||
| Solubility in chlorocarbons | soluble | ||
| −71.0·10−6 cm3/mol | |||
| Structure | |||
| α:rhombohedral, β: hexagonal | |||
| Octahedral | |||
| 0 D | |||
| Hazards | |||
| Main hazards | oxidizer; hydrolysis releases HCl | ||
| Related compounds | |||
Other anions
|
|||
Other cations
|
|||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tungsten hexachloride is an inorganic chemical compound of tungsten and chlorine with the chemical formula WCl
6. This dark violet-blue compound exists as volatile crystals under standard conditions. It is an important starting reagent in the preparation of tungsten compounds.[1] Other examples of charge-neutral hexachlorides are rhenium(VI) chloride and molybdenum(VI) chloride. The highly volatile tungsten hexafluoride is also known.
As a d0 atom, tungsten hexachloride is diamagnetic.
Preparation and structure
Tungsten hexachloride can be prepared by chlorinating tungsten metal in a sealed tube at 600 °C:[2]
- W + 3 Cl
2 → WCl
6
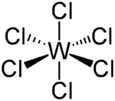
Tungsten hexachloride exists in both blue and red polymorphs, referred to respectively as α and β. The wine-red β can be obtained by rapid cooling, whereas the blue α form is more stable at room temperature. Although these polymorphs are distinctly colored, their molecular structures are very similar. Both polymorphs feature WCl
6 molecules that have octahedral geometry, in which all six W–Cl bonds are equivalent, and their length is equal to 224–226 pm. The densities are very similar: 3.68 g/cm3 for α and 3.62 g/cm3 for β. The low-temperature form is slightly more dense, as expected.[3]
Reactions
Tungsten hexachloride is readily hydrolyzed, even by moist air, giving the orange oxychlorides WOCl
4 and WO
2Cl
2, and subsequently, tungsten trioxide. WCl
6 is soluble in carbon disulfide, carbon tetrachloride, and phosphorus oxychloride.[2]
Methylation with trimethylaluminium affords hexamethyl tungsten:
- WCl
6 + 3 Al
2(CH
3)
6 → W(CH
3)
6 + 3 Al
2(CH
3)
4Cl
2
Treatment with butyl lithium affords a reagent that is useful for deoxygenation of epoxides.[4]
The chloride ligands in WCl
6 can be replaced by many anionic ligands including: bromide, thiocyanate, alkoxide, alkyl and aryl.
Reduction of WCl
6 can be effected with a mixture of tetrachloroethylene and tetraphenylarsonium chloride:[5]
- 2 WCl
6 + Cl
2C=CCl
2 + 2 (C
6H
5)
4AsCl → 2 (C
6H
5)
4As[WCl
6] + Cl
3C–CCl
3
The W(V) hexachloride is a derivative of tungsten(V) chloride.
It reacts with arsenic or hydrogen arsenide to form tungsten diarsenide.[6][7]
Safety considerations
WCl
6 is an aggressively corrosive oxidant, and hydrolyzes to release hydrogen chloride.
References
- ↑ J. W. Herndon; M. E. Jung (2007). "Encyclopedia of Reagents for Organic Synthesis". Encyclopedia of Reagents for Organic Synthesis. Wiley. doi:10.1002/9780470842898.rt430.pub2. ISBN 978-0-471-93623-7..
- ↑ 2.0 2.1 M. H. Lietzke; M. L. Holt (1950). "Tungsten(VI) Chloride (Tungsten Hexachloride)". Inorganic Syntheses. 3. p. 163. doi:10.1002/9780470132340.ch44. ISBN 978-0-470-13162-6.
- ↑ J. C. Taylor; P. W. Wilson (1974). "The Structure of β-Tungsten Hexachloride by Powder Neutron and X-ray Diffraction". Acta Crystallographica B30 (5): 1216–1220. doi:10.1107/S0567740874004572. Bibcode: 1974AcCrB..30.1216T..
- ↑ M. A. Umbreit, K. B. Sharpless (1990). "Deoxygenation of Epoxides with Lower Valent Tungsten Halides: trans-Cyclododecene". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=CV7P0121.; Collective Volume, 7, pp. 121
- ↑ Uhl, G.; Hey, E.; Becker, G.; Weller, F.; Dehnicke, K. (1983). "Über die Reaktion von 2,2-Dimethylpropylidinphosphan mit Wolframhexachlorid; die Kristallstrukturen von [(Cl3PO)WCL4(H9C4CCC4H9)] und [(H5C6)4As][WCL6]". Zeitschrift für Anorganische und Allgemeine Chemie 497 (2): 213–223. doi:10.1002/zaac.19834970221. http://nbn-resolving.de/urn:nbn:de:bsz:93-opus-57717.
- ↑ Lassner, Erik; Schubert, Wolf-Dieter (2012-12-06). Tungsten. Springer Science & Business Media. p. 145. ISBN 978-1-4615-4907-9.
- ↑ Meyer, R. J. (2013-09-03) (in de). Wolfram. Springer-Verlag. p. 207. ISBN 978-3-662-13401-6.
 |