Biology:Nitric oxide dioxygenase
| nitric oxide dioxygenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| EC number | 1.14.12.17 | ||||||||
| CAS number | 214466-78-1 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Nitric oxide dioxygenase (EC 1.14.12.17) is an enzyme that catalyzes the conversion of nitric oxide (NO) to nitrate (NO−3) .[2] The net reaction for the reaction catalyzed by nitric oxide dioxygenase is shown below:
- 2NO + 2O2 + NAD(P)H → 2NO3− + NAD(P)+ + H+
Nitric oxide is a ubiquitous small molecule that is integrated in a wide variety of physiological processes including smooth muscle vasodilation, platelet disaggregation, neurotransmission, and immune response to bacterial infection.[3][4] Overproduction of this signaling molecule can be lethal to cells by poisoning cellular energy production. The most sensitive targets of NO are aconitase, an enzyme that catalyzes the isomerization of citrate to isocitrate in the citric acid cycle, and cytochrome oxidase, the last enzyme in the respiratory electron transport chain of mitochondria.[5] Additionally NO, with its lone radical on the nitrogen atom, is implicated in a number of secondary mechanisms of toxicity, including catalase inhibition (resulting in hydrogen peroxide toxicity), Fe-S center iron liberation, and the formation of dinitosyl-iron complexes.
Due to the potential lethality of NO, cells benefitted greatly from the evolution of an enzyme capable of catalyzing the conversion of toxic NO to nitrate. A 'nitric oxide dioxygenase' is an enzyme that is capable of carrying out this reaction. NO dioxygenase belongs to the family of oxidoreductases, more specifically those acting on paired donors, with O2 as oxidant and with incorporation of two atoms of oxygen into the other donor.
Reaction mechanism
The mechanism of action has still not been entirely deduced, however, the leading theory suggests that the conversion is carried out through a series of redox reactions involving iron centers as shown in the series of half reactions below:[6]
| Step | Reaction |
|---|---|
| FAD reduction | NAD(P)H + FAD + H+ → NAD(P)+ + FADH2 |
| Iron reduction 1 | FADH2 + Fe3+ → Fe2+ + FADH + H+ |
| Iron Reduction 2 | FADH + Fe3+ → FAD + Fe2+ + H+ |
| O2 Binding | Fe2+ + O2 → Fe3+(O2−) |
| NO dioxygenation | Fe3+(O2−) + NO → Fe3+ + NO3− |
Another theory developed more recently (2009) suggests that a NO dioxygenase activity could also proceed through phenolic nitration via a putative heme-peroxynitrite intermediate.[7]
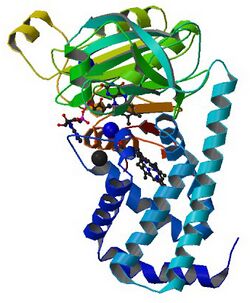
The most well studied NO dioxygenase is flavohemoglobin (flavoHb), shown to the right: Studies have shown that flavohemoglobins are induced by NO, nitrite, nitrate, and NO-releasing agents in various bacteria and fungi.[6][8] Additionally, flavoHbs have been shown to protect bacteria, yeast, and Dictyostelium discoideum against growth inhibition and damage mediated via NO.[8][9][10]
Discovery
Nitric oxide dioxygenase was discovered, and first reported in 1998, as an inducible O2-dependent enzymatic activity that protected bacteria against nitric oxide toxicity.[11] The enzyme was identified with the E. coli flavohemoglobin.[12]
More recently, another protein has been identified as a NO dioxygenase - rhodobacter sphaeroides haem protein (SHP), a novel cytochrome with NO dioxygenase activity.[13][14] Although the biological function of SHP has yet to be identified, SHP has been shown, that with oxygen bound, it can react rapidly with nitric oxide to form nitrate.[13]
Structure and molecular function
The flavohemoglobin protein contains two domains: an oxidoreductase FAD-binding domain, and a b-type heme-containing "globin" domain and optionally an oxidoreductase NAD-binding domain. The reductase domain supplies an electron to the heme iron to achieve a high rate of catalytic NO dioxygenation. In addition to numerous flavohemoglobins, many distantly related members of the hemoglobin superfamily including the muscle myoglobin, the non-symbiotic plant hemoglobin and symbiotic plant leghemoglobin, the neuronal neuroglobin, and the mammalian cytoplasmic cytoglobin[15][16] appear to function as nitric oxide dioxygenases (NODs), although the cellular electron donor(s) for many globins have yet to be defined. Electron donors may include ascorbate, cytochrome b5 or ferredoxin reductase.[17] The catalytic NO dioxygenation can be written in its simplest form:
- NO + O2 + e− NO3−
Catalysis is very efficient. The reported bimolecular NO dioxygenation rate constants range from 2 x 107 M−1s−1 for cytoglobin to 3 x 109 M−1s−1 for flavohemoglobin, and turnover rates range from 1 to 700 s−1. Structure, O2 binding, and reduction of globins appear optimized for a NO dioxygenase function.
Physiological function
Historically, nitric oxide dioxygenase (around 1.8 billion years ago) served to provide the modern day analogue of hemoglobin/myoglobin function for oxygen storage and transport. Gardner et al. (1998) suggested that the first hemoglobin/myoglobin probably functioned as an enzyme utilizing bound ‘activated’ oxygen gas to dioxygenate NO in microbes.[6]
The wide diversity of multicellular organisms benefitting from the oxygen storage and transport functions of myoglobin/hemoglobin appeared much later (approximately 0.5 billion years ago).
NODs are now known to serve two important physiological functions in diverse life forms: they prevent NO toxicity (otherwise known as "nitrosative stress") and regulate NO signalling.[2] NODs belong to the larger family of well-established free radical and reactive oxygen detoxifying enzymes that includes superoxide dismutase, catalase, and peroxidase.
Distribution in nature
NODs, as well as many hemoglobins that function as NODs, are distributed to most life forms including bacteria, fungi, protists, worms, plants and animals. In fact, nitric oxide dioxygenation appears to be a primal function for members of the hemoglobin superfamily. Moreover, it is becoming increasingly evident that the NOD function of globins is much more common[18] than the paradigmatic O2 transport-storage function of red cell hemoglobin[19] which was first investigated and reported over a century earlier by Felix Hoppe-Seyler and others.[20] Other proteins that may act as NODs include mammalian microsomal cytochrome P450(s)[21] and a novel O2-binding cytochrome b from Rhodobacter sphaeroides.[13]
Technologies
Inhibitors of the NODs are being developed for application as microbial antibiotics,[22][23] anti-tumor agents and modulators of NO signalling. The most prominent class of inhibitor of NO dioxygenase to date is imidazole antibiotics. Imidazoles have been shown to coordinate with the heme iron atom of microbial flavohemoglobin, impair ferric heme reduction, produce uncompetitive inhibition with respect to O2 and NO, and inhibit NO metabolism by yeasts and bacteria.[22] Specifically, imidazoles bearing bulky aromatic substituents have been shown to have potential for selective and high-affinity inhibition of NO dioxygenase function by coordinating the catalytic heme iron and "fitting" within the large hydrophobic distal heme pocket.[22][24][25] As a result, imidazole engineering has been suggested as a means to specifically inhibit NO dioxygenases.
In addition, genetically modified plants with heterologous flavohemoglobin-NODs are being developed to limit NO toxicity created by metabolism of nitrogen fertilizers by soil microbes and as a means towards plant self-fertilization through absorption of environmental NO.
Recently a lentiviral vector that allows for expression of E. coli flavoHb in mammalian cells has been described. This approach demonstrated that flavoHb is indeed enzymatically active within human and murine cells and potently blocks exogenous and endogenous sources of nitrosative stress.[26] This technology was then extended to interrogate the role of NO synthesis in the highly tumorigenic cancer stem cells (CSCs) from human glioblastoma (brain tumor) samples. Expression of flavoHb within xenografted tumors led to depletion of NO generated by iNOS/NOS2. The phenotypic result was loss of tumorigenicity of the CSCs and improved mouse survival.[27] These experiments demonstrate that flavoHb can be employed for in vivo studies of nitric oxide biology and suggest that therapeutic NO-depletion may be achieved via heterologous expression of bacterial flavoHbs.
References
- ↑ PDB: 1gvh; "The X-ray structure of ferric Escherichia coli flavohemoglobin reveals an unexpected geometry of the distal heme pocket". J. Biol. Chem. 277 (26): 23725–32. June 2002. doi:10.1074/jbc.M202228200. PMID 11964402.
- ↑ 2.0 2.1 "Protection from nitrosative stress: a central role for microbial flavohemoglobin". Free Radic. Biol. Med. 52 (9 =): 1620–33. May 2012. doi:10.1016/j.freeradbiomed.2012.01.028. PMID 22343413.
- ↑ "Nitric oxide: physiology, pathophysiology, and pharmacology". Pharmacol. Rev. 43 (2): 109–42. June 1991. PMID 1852778.
- ↑ Fang FC (October 2004). "Antimicrobial reactive oxygen and nitrogen species: concepts and controversies". Nat. Rev. Microbiol. 2 (10): 820–32. doi:10.1038/nrmicro1004. PMID 15378046.
- ↑ "Nitric oxide sensitivity of the aconitases". J. Biol. Chem. 272 (40): 25071–6. October 1997. doi:10.1074/jbc.272.40.25071. PMID 9312115.
- ↑ 6.0 6.1 6.2 "Nitric oxide dioxygenase: an enzymic function for flavohemoglobin". Proc. Natl. Acad. Sci. U.S.A. 95 (18): 10378–83. September 1998. doi:10.1073/pnas.95.18.10378. PMID 9724711.
- ↑ "Heme/O2/*NO nitric oxide dioxygenase (NOD) reactivity: phenolic nitration via a putative heme-peroxynitrite intermediate". J. Am. Chem. Soc. 131 (32): 11304–5. August 2009. doi:10.1021/ja904832j. PMID 19627146.
- ↑ 8.0 8.1 "Nitric-oxide dioxygenase activity and function of flavohemoglobins. sensitivity to nitric oxide and carbon monoxide inhibition". J. Biol. Chem. 275 (41): 31581–7. October 2000. doi:10.1074/jbc.M004141200. PMID 10922365.
- ↑ "Flavohemoglobin denitrosylase catalyzes the reaction of a nitroxyl equivalent with molecular oxygen". PNAS 98 (18): 10108–12. 2001. doi:10.1073/pnas.181199698. PMID 11517313.
- ↑ "Escherichia coli flavohaemoglobin (Hmp) with equistoichiometric FAD and haem contents has a low affinity for dioxygen in the absence or presence of nitric oxide". Biochem. J. 353 (Pt 2): 207–13. January 2001. doi:10.1042/0264-6021:3530207. PMID 11139382.
- ↑ "Constitutive and adaptive detoxification of nitric oxide in Escherichia coli. Role of nitric-oxide dioxygenase in the protection of aconitase". J. Biol. Chem. 273 (41): 26528–33. 1998. doi:10.1074/jbc.273.41.26528. PMID 9756889.
- ↑ "Nitric oxide dioxygenase: an enzymic function for flavohemoglobin". Proc. Natl. Acad. Sci. U.S.A. 95 (18): 10378–83. 1998. doi:10.1073/pnas.95.18.10378. PMID 9724711.
- ↑ 13.0 13.1 13.2 "Rhodobacter sphaeroides haem protein: a novel cytochrome with nitric oxide dioxygenase activity". Biochem. Soc. Trans. 36 (Pt 5): 992–5. October 2008. doi:10.1042/BST0360992. PMID 18793176.
- ↑ "Characterization of the nitric oxide reductase-encoding region in Rhodobacter sphaeroides 2.4.3". J. Bacteriol. 179 (11): 3534–40. June 1997. doi:10.1128/jb.179.11.3534-3540.1997. PMID 9171397.
- ↑ "Nitric-oxide dioxygenase function of human cytoglobin with cellular reductants and in rat hepatocytes". J. Biol. Chem. 285 (31): 23850–7. July 2010. doi:10.1074/jbc.M110.132340. PMID 20511233.
- ↑ "Cytoglobin is expressed in the vasculature and regulates cell respiration and proliferation via nitric oxide dioxygenation". J. Biol. Chem. 284 (13): 8539–47. March 2009. doi:10.1074/jbc.M808231200. PMID 19147491.
- ↑ Gardner PR (January 2005). "Nitric oxide dioxygenase function and mechanism of flavohemoglobin, hemoglobin, myoglobin and their associated reductases". J. Inorg. Biochem. 99 (1): 247–66. doi:10.1016/j.jinorgbio.2004.10.003. PMID 15598505.
- ↑ Gardner PR (October 2012). "Hemoglobin: A Nitric-Oxide Dioxygenase". Scientifica 2012: 34. doi:10.6064/2012/683729. PMID 24278729.
- ↑ "Diversity of globin function: enzymatic, transport, storage, and sensing". J. Biol. Chem. 283 (14): 8773–7. April 2008. doi:10.1074/jbc.R700029200. PMID 18211906.
- ↑ Hoppe-Seyler F (1866). "Über die Oxydation in lebenden Blute". Med.-Chem. Untersuch Lab. 1: 133–40.
- ↑ "Nitric oxide metabolism in mammalian cells: substrate and inhibitor profiles of a NADPH-cytochrome P450 oxidoreductase-coupled microsomal nitric oxide dioxygenase". Free Radic. Biol. Med. 37 (2): 216–28. July 2004. doi:10.1016/j.freeradbiomed.2004.04.031. PMID 15203193.
- ↑ 22.0 22.1 22.2 "Imidazole antibiotics inhibit the nitric oxide dioxygenase function of microbial flavohemoglobin". Antimicrob. Agents Chemother. 49 (5): 1837–43. May 2005. doi:10.1128/AAC.49.5.1837-1843.2005. PMID 15855504.
- ↑ "Structure of Ralstonia eutropha flavohemoglobin in complex with three antibiotic azole compounds". Biochemistry 50 (7): 1255–64. February 2011. doi:10.1021/bi101650q. PMID 21210640.
- ↑ "Crystal structure of the flavohemoglobin from Alcaligenes eutrophus at 1.75 Å resolution". EMBO J. 14 (24): 6067–77. December 1995. doi:10.1002/j.1460-2075.1995.tb00297.x. PMID 8557026.
- ↑ "Phospholipid bound to the flavohemoprotein from Alcaligenes eutrophus". Eur. J. Biochem. 262 (2): 396–405. June 1999. doi:10.1046/j.1432-1327.1999.00381.x. PMID 10336624.
- ↑ "Bacterial flavohemoglobin: a molecular tool to probe mammalian nitric oxide biology". BioTechniques 50 (1 =): 41–45. January 2011. doi:10.2144/000113586. PMID 21231921.
- ↑ "Glioma stem cell proliferation and tumor growth are promoted by nitric oxide synthase-2". Cell 146 (1 =): 53–66. July 2011. doi:10.1016/j.cell.2011.06.006. PMID 21729780.
 |

