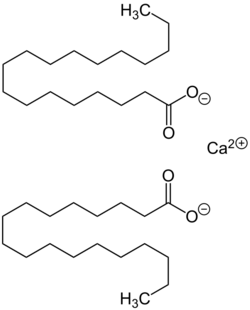
Chemistry:Calcium stearate
| |
| Names | |
|---|---|
| Preferred IUPAC name
Calcium di(octadecanoate) | |
| Other names
E470
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C36H70CaO4 | |
| Molar mass | 607.030 g·mol−1 |
| Appearance | white to yellowish-white powder |
| Density | 1.08 g/cm3 |
| Melting point | 155 °C (311 °F; 428 K) |
| 0.004 g/100 mL (15 °C) | |
| Solubility | soluble in hot pyridine slightly soluble in oil insoluble in alcohol, ether |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Calcium stearate is a carboxylate salt of calcium, classified as a calcium soap. The salt is a component of some lubricants, surfactants, as well as many foodstuffs. It is a white waxy powder.[1]
Production and occurrence
Calcium stearate is produced by heating stearic acid and calcium oxide:
- 2 C17H35COOH + CaO → (C17H35COO)2Ca + H2O
It is also the main component of soap scum, a white solid that forms when soap is mixed with hard water. Unlike soaps containing sodium and potassium, calcium stearate is insoluble in water and does not lather well.[2] Commercially it is sold as a 50% dispersion in water or as a spray dried powder. As a food additive it is known by the generic E number E470.
Applications
Calcium stearate is a waxy material with low solubility in water, unlike traditional sodium and potassium soaps. It is also easy and cheap to produce, and exhibits low toxicity. These attributes are the basis of many of its applications. Related applications exist for the magnesium stearate.[1]
- Calcium stearate is used as a flow agent and surface conditioner in some candies such as Smarties, jawbreakers and Sprees.
- It is a waterproofing agent for fabrics.
- A lubricant in pencils and crayons.
- A lubricant in the dry drawing method of wire production
- The concrete industry uses calcium stearate for efflorescence control of cementitious products used in the production of concrete masonry units i.e. paver and block, as well as waterproofing.[3]
- In paper production, calcium stearate is used as a lubricant to provide good gloss, preventing dusting and fold cracking in paper and paperboard making.[4]
- In plastics, it can act as an acid scavenger or neutralizer at concentrations up to 1000ppm, a lubricant and a release agent. It may be used in plastic colorant concentrates to improve pigment wetting. In rigid PVC, it can accelerate fusion, improve flow, and reduce die swell.
- Applications in the personal care and pharmaceutical industry include tablet mold release, anti-tack agent, and gelling agent.
- Calcium stearate is a component in some types of defoamers.
- Anticaking agent for drugs etc.[5]
References
- ↑ 1.0 1.1 "Metallic Soaps". Ullmann's Encyclopedia of Industrial Chemistry. 2001. doi:10.1002/14356007.a16_361. ISBN 3527306730.
- ↑ "Water". Ullmann's Encyclopedia of Industrial Chemistry. 2000. doi:10.1002/14356007.a28_001. ISBN 3527306730.
- ↑ Preventing Efflorescence, Portland Cement Association
- ↑ Flynn, Gregory J.; John M. Krasniewski & Joseph Wilson Jr., "Lubricant additives for paper coating compositions", US patent 5527383, published 1996-06-18
- ↑ "Foods, 3. Food Additives". Ullmann's Encyclopedia of Industrial Chemistry. 2000. doi:10.1002/14356007.a11_561. ISBN 3527306730.
 |

