Chemistry:Hexadecane
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Hexadecane[1] | |
| Other names
Cetane
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1736592 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| 103739 | |
| MeSH | n-hexadecane |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C16H34 | |
| Molar mass | 226.448 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Gasoline-like to odorless |
| Density | 0.77 g/cm3[2][3] |
| Melting point | 18.18 °C (64.72 °F; 291.33 K)[2] |
| Boiling point | 286.9 °C (548.4 °F; 560.0 K)[2] |
| log P | 8.859 |
| Vapor pressure | < 0.1 mbar (20 °C) |
Henry's law
constant (kH) |
43 nmol Pa−1 kg−1 |
| -187.6·10−6 cm3/mol[4] | |
| Thermal conductivity | 0.140 W/(m·K)[5] |
Refractive index (nD)
|
1.4329[2] |
| Viscosity | 3.03 mPa·s[6] |
| Thermochemistry[7] | |
Heat capacity (C)
|
501.6 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−456.1 kJ mol−1 |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | WARNING |
| H315 | |
| Flash point | 136 °C (277 °F; 409 K)[8] |
| 202 °C (396 °F; 475 K)[8] | |
| Related compounds | |
Related alkanes
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
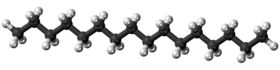
Hexadecane (also called cetane) is an alkane hydrocarbon with the chemical formula C16H34. Hexadecane consists of a chain of 16 carbon atoms, with three hydrogen atoms bonded to the two end carbon atoms, and two hydrogens bonded to each of the 14 other carbon atoms.
Cetane number
Cetane is often used as a shorthand for cetane number, a measure of the combustion of diesel fuel.[9] Cetane ignites very easily under compression; for this reason, it is assigned a cetane number of 100, and serves as a reference for other fuel mixtures.[10]
Hexadecyl radical
Hexadecyl is an alkyl radical of carbon and hydrogen derived from hexadecane, with formula C16H33 and with mass 225.433,[11] occurring especially in cetyl alcohol.[12] It confers strong hydrophobicity on molecules containing it.[13] Carboplatin modified with hexadecyl and polyethylene glycol has increased liposolubility and PEGylation, proposed to useful in chemotherapy, specifically non-small-cell lung cancer.[14]
Hexadecyl was used from 1982 for radiolabelling,[15] and this continues to be useful,[16] for example for radiolabelling exosomes and hydrogels,[17] and for positron emission tomography.[18]
Hexadecyl platelet-activating factor has profound effects on the lung,[19] and hexadecyl glyceryl ether participates in the biosynthesis of plasmalogens.[20]
See also
- Cetane index
- Isocetane
- Higher alkanes
References
- ↑ CID 11006 from PubChem
- ↑ 2.0 2.1 2.2 2.3 Haynes, p. 3.294
- ↑ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ↑ Haynes, p. 3.578
- ↑ Haynes, p. 6.256
- ↑ Haynes, p. 6.245
- ↑ Haynes, p. 5.21
- ↑ 8.0 8.1 Haynes, p. 16.25
- ↑ "Cetane" (in en). http://www.mckinseyenergyinsights.com/resources/refinery-reference-desk/cetane/.
- ↑ Speight, James G. (2015). Handbook of Petroleum Product Analysis.. Hoboken, NJ: Wiley. pp. 158–159. ISBN 978-1-322-95015-0. OCLC 903318141. https://www.worldcat.org/oclc/903318141.
- ↑ "Hexadecyl". ChemSpider. Royal Society of Chemistry. http://www.chemspider.com/Chemical-Structure.150674.html.
- ↑ "hexadecyl noun". Merriam-Webster. https://www.merriam-webster.com/dictionary/hexadecyl.
- ↑ Hatanaka, K (2011). Horváth, István T.. ed. Fluorous Chemistry. Springer Science & Business Media. p. 294. ISBN 9783642252334. https://books.google.com/books?id=7KdIV-iPA5cC&pg=PA294.
- ↑ Lang, Tingting; Li, Nuannuan; Zhang, Jing; Li, Yi; Rong, Rong; Fu, Yuanlei (2021). "Prodrug-based nano-delivery strategy to improve the antitumor ability of carboplatin in vivo and in vitro". Drug Delivery 28 (1): 1272–1280. doi:10.1080/10717544.2021.1938754. PMID 34176381.
- ↑ Pool, G. L.; French, M. E.; Edwards, R. A.; Huang, L.; Lumb, R. H. (1982). "Use of radiolabeled hexadecyl cholesteryl ether as a liposome marker". Lipids 17 (6): 448–452. doi:10.1007/BF02535225. PMID 7050582.
- ↑ Manual Kollareth, Denny Joseph; Chang, Chuchun L.; Hansen, Inge H.; Deckelbaum, Richard J. (2018). "Radiolabeled cholesteryl ethers: A need to analyze for biological stability before use". Biochemistry and Biophysics Reports 13: 1–6. doi:10.1016/j.bbrep.2017.10.007. PMID 29188234.
- ↑ Lee, Yanick (July 2017) (Master's thesis). Radiosynthesis of hexadecyl-4-[ 18Ffluorobenzoate for labeling exosomes and chitosan hydrogels]. Université de Montréal. https://papyrus.bib.umontreal.ca/xmlui/bitstream/handle/1866/19446/Lee_Yanick_2017_memoire.pdf?sequence=2&isAllowed=y.
- ↑ "Radioactive cell labeling agent". KR101130737B1. 2012. https://patents.google.com/patent/KR101130737B1/en.
- ↑ Haroldsen, P. E.; Voelkel, N. F.; Henson, J. E.; Henson, P. M.; Murphy, R. C. (1987). "Metabolism of platelet-activating factor in isolated perfused rat lung". Journal of Clinical Investigation 79 (6): 1860–1867. doi:10.1172/JCI113028. PMID 3108322.
- ↑ Wood, Randall; Healy, Kathleen (1970). "Tumor lipids: Biosynthesis of plasmalogens". Biochemical and Biophysical Research Communications 38 (2): 205–211. doi:10.1016/0006-291x(70)90697-2. PMID 5418699.
Cited sources
- Haynes, William M., ed (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. ISBN 9781498754293.
External links
- Vapor pressure and liquid density calculation
- Technique to determine hexadecane transfer
 |
