Biology:PEGylation
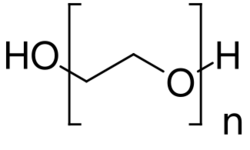
PEGylation (or pegylation) is the process of both covalent and non-covalent attachment or amalgamation of polyethylene glycol (PEG, in pharmacy called macrogol) polymer chains to molecules and macrostructures, such as a drug, therapeutic protein or vesicle, which is then described as PEGylated.[1][2][3][4] PEGylation affects the resulting derivatives or aggregates interactions, which typically slows down their coalescence and degradation as well as elimination in vivo.[5][6]
PEGylation is routinely achieved by the incubation of a reactive derivative of PEG with the target molecule. The covalent attachment of PEG to a drug or therapeutic protein can "mask" the agent from the host's immune system (reducing immunogenicity and antigenicity), and increase its hydrodynamic size (size in solution), which prolongs its circulatory time by reducing renal clearance. PEGylation can also provide water solubility to hydrophobic drugs and proteins. Having proven its pharmacological advantages and acceptability, PEGylation technology is the foundation of a growing multibillion-dollar industry.[7]
Methodology
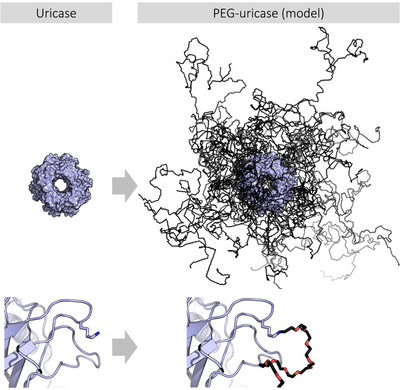
PEGylation is the process of attaching the strands of the polymer PEG to molecules, most typically peptides, proteins, and antibody fragments, that can improve the safety and efficiency of many therapeutics.[9][10] It produces alterations in the physiochemical properties including changes in conformation, electrostatic binding, hydrophobicity etc. These physical and chemical changes increase systemic retention of the therapeutic agent. Also, it can influence the binding affinity of the therapeutic moiety to the cell receptors and can alter the absorption and distribution patterns.
PEGylation, by increasing the molecular weight of a molecule, can impart several significant pharmacological advantages over the unmodified form, such as improved drug solubility, reduced dosage frequency with potentially reduced toxicity and without diminished efficacy, extended circulating life, increased drug stability, and enhanced protection from proteolytic degradation; PEGylated forms may also be eligible for patent protection.[11]
PEGylated drugs
The attachment of an inert and hydrophilic polymer was first reported around 1970 to extend blood life and control immunogenicity of proteins.[12] Polyethylene glycol was chosen as the polymer.[13][14] In 1981 Davis and Abuchowski founded Enzon, Inc., which brought three PEGylated drugs to market. Abuchowski later founded and is CEO of Prolong Pharmaceuticals.[15]
The clinical value of PEGylation is now well established. ADAGEN (pegademase bovine) manufactured by Enzon Pharmaceuticals, Inc., US was the first PEGylated protein approved by the U.S. Food and Drug Administration (FDA) in March 1990, to enter the market. It is used to treat a form of severe combined immunodeficiency syndrome (ADA-SCID), as an alternative to bone marrow transplantation and enzyme replacement by gene therapy. Since the introduction of ADAGEN, a large number of PEGylated protein and peptide pharmaceuticals have followed and many others are under clinical trial or under development stages. Sales of the two most successful products, Pegasys and Neulasta, exceeded $5 billion in 2011.[16][17] All commercially available PEGylated pharmaceuticals contain methoxypoly(ethylene glycol) or mPEG. PEGylated pharmaceuticals on the market (in reverse chronology by FDA approval year) have included:[18]
- A PEGylated lipid is used as an excipient in both the Moderna vaccine and the Pfizer–BioNTech COVID-19 vaccine. Both RNA vaccines consist of Messenger RNA, or mRNA, encased in a bubble of oily molecules called lipids. Proprietary lipid technology is used for each. In both vaccines, the bubbles are coated with a stabilizing molecule of polyethylene glycol. As of December 2020, there is some concern that PEG could trigger an allergic reaction,[19][20] as appears to have occurred by 19 December, in at least three "Alaska health care worker" people who were administered the Pfizer–BioNTech COVID-19 vaccine.[21] The particular PEGylated molecule in the Moderna vaccine is known as DMG-PEG 2000.
- Pegvaliase (Biomarin) – PEGylated recombinant phenylalanine ammonia-lyase for the treatment of Phenylketonuria, approved by the FDA for the US in May 2018.[22][23]
- Adynovate – PEGylated Antihemophilic Factor VIII for the treatment of patients with hemophilia A. (Baxalta, 2015)[24]
- Irinotecan liposome (Onivyde) – PEGylated liposomal irinotecan hydrochloride trihydrate for the treatment of metastatic pancreatic cancer in adults proceeding treatment with gemcitabine-based therapy. (Ipsen, 2015)
- Plegridy – PEGylated Interferon Beta-1a for the treatment of patients with relapsing forms of multiple sclerosis. (Biogen, 2014)
- Naloxegol (Movantik) – PEGylated naloxol for the treatment of opioid-induced constipation in adults patients with chronic non-cancer pain (un-pegylated methadone can cause adverse gastrointestinal reactions). (AstraZeneca, 2014)
- Peginesatide (Omontys) – once-monthly medication to treat anemia associated with chronic kidney disease in adult patients on dialysis (Affymax/Takeda Pharmaceuticals, 2012)
- Pegloticase (Krystexxa) – PEGylated uricase for the treatment of gout (Savient, 2010)
- Certolizumab pegol (Cimzia) – monoclonal antibody for treatment of moderate to severe rheumatoid arthritis and Crohn's disease, an inflammatory gastrointestinal disorder (Nektar/UCB Pharma, 2008)
- Methoxy polyethylene glycol-epoetin beta (Mircera) – PEGylated form of erythropoietin to combat anemia associated with chronic kidney disease (Roche, 2007)
- Pegaptanib (Macugen) – used to treat neovascular age-related macular degeneration (Pfizer, 2004)
- Pegfilgrastim (Neulasta) – PEGylated recombinant methionyl human granulocyte colony-stimulating factor for severe cancer chemotherapy-induced neutropenia (Amgen, 2002)
- Pegvisomant (Somavert) – PEG-human growth hormone mutein receptor antagonist for treatment of Acromegaly (Pfizer, 2002)
- Peginterferon alfa-2a (Pegasys) – PEGylated interferon alpha for use in the treatment of chronic hepatitis C and hepatitis B (Hoffmann-La Roche, 2002)
- Peginterferon alfa-2b (PegIntron) – PEGylated interferon alpha for use in the treatment of chronic hepatitis C and hepatitis B (Schering-Plough/Enzon, 2000)
- Doxorubicin HCl liposome (Doxil/Caelyx) – PEGylated liposome containing doxorubicin for the treatment of cancer (Alza, 1995)
- Pegaspargase (Oncaspar) – PEGylated L-asparaginase for the treatment of acute lymphoblastic leukemia in patients who are hypersensitive to the native unmodified form of L-asparaginase (Enzon, 1994). This drug was recently approved for front line use.
- Pegademase bovine (Adagen) – PEG-adenosine deaminase for the treatment of severe combined immunodeficiency disease (SCID) (Enzon, 1990)
Patent litigation
The PEGylated lipid nanoparticle drug delivery (LNP) system of the mRNA vaccine known as mRNA-1273 has been the subject of ongoing patent litigation with Arbutus Biopharma, from whom Moderna had previously licensed LNP technology.[25][26] On 4 September 2020, Nature Biotechnology reported that Moderna had lost a key challenge in the ongoing case.[27]
Use in research
PEGylation has practical uses in biotechnology for protein delivery,[28] cell transfection, and gene editing in non-human cells.[29]
Process
The first step of the PEGylation is the suitable functionalization of the PEG polymer at one or both ends. PEGs that are activated at each end with the same reactive moiety are known as "homobifunctional", whereas if the functional groups present are different, then the PEG derivative is referred as "heterobifunctional" or "heterofunctional". The chemically active or activated derivatives of the PEG polymer are prepared to attach the PEG to the desired molecule.[30]
The overall PEGylation processes used to date for protein conjugation can be broadly classified into two types, namely a solution phase batch process and an on-column fed-batch process.[31] The simple and commonly adopted batch process involves the mixing of reagents together in a suitable buffer solution, preferably at a temperature between 4 and 6 °C, followed by the separation and purification of the desired product using a suitable technique based on its physicochemical properties, including size exclusion chromatography (SEC), ion exchange chromatography (IEX), hydrophobic interaction chromatography (HIC) and membranes or aqueous two-phase systems (ATPS).[32][33]
The choice of the suitable functional group for the PEG derivative is based on the type of available reactive group on the molecule that will be coupled to the PEG. For proteins, typical reactive amino acids include lysine, cysteine, histidine, arginine, aspartic acid, glutamic acid, serine, threonine and tyrosine. The N-terminal amino group and the C-terminal carboxylic acid can also be used as a site specific site by conjugation with aldehyde functional polymers.[34]
The techniques used to form first generation PEG derivatives are generally reacting the PEG polymer with a group that is reactive with hydroxyl groups, typically anhydrides, acid chlorides, chloroformates and carbonates. In the second generation PEGylation chemistry more efficient functional groups such as aldehyde, esters, amides etc. are made available for conjugation.
As applications of PEGylation have become more and more advanced and sophisticated, there has been an increase in need for heterobifunctional PEGs for conjugation. These heterobifunctional PEGs are very useful in linking two entities, where a hydrophilic, flexible and biocompatible spacer is needed. Preferred end groups for heterobifunctional PEGs are maleimide, vinyl sulfones, pyridyl disulfide, amine, carboxylic acids and NHS esters.[35][36][37]
Third-generation pegylation agents, where the polymer has been branched, Y-shaped or comb-shaped are available and show reduced viscosity and lack of organ accumulation.[38] Recently also enzymatic approaches of PEGylation have been developed, thus further expanding the conjugation tools.[39][40] PEG-protein conjugates obtained by enzymatic methods are already in clinical use, for example: Lipegfilgrastim, Rebinyn, Esperoct.
Limitations
Unpredictability in clearance times for PEGylated compounds may lead to the accumulation of large-molecular-weight compounds in the liver leading to inclusion bodies with no known toxicologic consequences.[41] Furthermore, alteration in the chain length may lead to unexpected clearance times in vivo.[42] Moreover, the experimental conditions of PEGylation reaction (i.e. pH, temperature, reaction time, overall cost of the process and molar ratio between PEG derivative and peptide) also have an impact on the stability of the final PEGylated products.[43] To overcome the above-mentioned limitations different strategies such as changing the size (Mw), the number, the location and the type of linkage of PEG molecule were offered by several researchers.[44][45] Conjugation to biodegradable polysaccharides, which is a promising alternative to PEGylation, is another way to solve the biodegradability issue of PEG.[46]
See also
- Cytochrome c
- Interferon
- Matrix-assisted laser desorption/ionization
- Proteomics
- Solid lipid nanoparticles
References
- ↑ Jokerst, Jesse V; Lobovkina, Tatsiana; Zare, Richard N; Gambhir, Sanjiv S (June 2011). "Nanoparticle PEGylation for imaging and therapy". Nanomedicine 6 (4): 715–728. doi:10.2217/nnm.11.19. PMID 21718180.
- ↑ Knop, Katrin; Hoogenboom, Richard; Fischer, Dagmar; Schubert, Ulrich S. (23 August 2010). "Poly(ethylene glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives". Angewandte Chemie International Edition 49 (36): 6288–6308. doi:10.1002/anie.200902672. PMID 20648499.
- ↑ Veronese, Francesco M; Mero, Anna (2008). "The Impact of PEGylation on Biological Therapies". BioDrugs 22 (5): 315–329. doi:10.2165/00063030-200822050-00004. PMID 18778113.
- ↑ Veronese, Francesco M.; Pasut, Gianfranco (November 2005). "PEGylation, successful approach to drug delivery". Drug Discovery Today 10 (21): 1451–1458. doi:10.1016/S1359-6446(05)03575-0. PMID 16243265.
- ↑ "Liposomes for the sustained drug release in vivo". Biochimica et Biophysica Acta (BBA) - Biomembranes 1029 (1): 91–97. 13 April 1990. doi:10.1016/0005-2736(90)90440-y. PMID 2223816.
- ↑ "Amphipathic polyethyleneglycols effectively prolong the circulation time of liposomes". FEBS Lett 268 (1): 235–237. 30 July 1990. doi:10.1016/0014-5793(90)81016-h. PMID 2384160.
- ↑ Damodaran V. B. ; Fee C. J. (2010). "Protein PEGylation: An overview of chemistry and process considerations". European Pharmaceutical Review 15 (1): 18–26. http://www.europeanpharmaceuticalreview.com/494/european-pharmaceutical-review-magazine/past-issues/issue-1-2010/protein-pegylation-an-overview-of-chemistry-and-process-considerations/#.VBmc4Y7tC-8.
- ↑ Sherman, MR; Saifer, MG; Perez-Ruiz, F (3 January 2008). "PEG-uricase in the management of treatment-resistant gout and hyperuricemia.". Advanced Drug Delivery Reviews 60 (1): 59–68. doi:10.1016/j.addr.2007.06.011. PMID 17826865.
- ↑ Veronese, FM; Harris, JM (June 2002). "Introduction and overview of peptide and protein pegylation". Advanced Drug Delivery Reviews 54 (4): 453–456. doi:10.1016/s0169-409x(02)00020-0. PMID 12052707.
- ↑ Porfiryeva, N. N.; Moustafine, R. I.; Khutoryanskiy, V. V. (January 2020). "PEGylated Systems in Pharmaceutics". Polymer Science, Series C 62 (1): 62–74. doi:10.1134/S181123822001004X. http://centaur.reading.ac.uk/91652/9/19Aug2020PEGylation%20review%20-accepted.pdf.
- ↑ Milla, P; Dosio, F (13 January 2012). "PEGylation of proteins and liposomes: a powerful and flexible strategy to improve the drug delivery.". Current Drug Metabolism 13 (1): 105–119. doi:10.2174/138920012798356934. PMID 21892917.
- ↑ Davis, Frank F. (June 2002). "The origin of pegnology". Advanced Drug Delivery Reviews 54 (4): 457–458. doi:10.1016/s0169-409x(02)00021-2. PMID 12052708.
- ↑ Abuchowski, A; Van Es, T; Palczuk, N. C.; Davis, F. F. (1977). "Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol". The Journal of Biological Chemistry 252 (11): 3578–81. doi:10.1016/S0021-9258(17)40291-2. PMID 405385.
- ↑ Abuchowski, A; McCoy, J. R.; Palczuk, N. C.; Van Es, T; Davis, F. F. (1977). "Effect of covalent attachment of polyethylene glycol on immunogenicity and circulating life of bovine liver catalase". The Journal of Biological Chemistry 252 (11): 3582–6. doi:10.1016/S0021-9258(17)40292-4. PMID 16907.
- ↑ "Dr. Abraham Abuchowski, Ph.D. – Home". https://prolongpharma.com/members/abe-abuchowski/.
- ↑ Klauser, Alexander (Head), Roche Group Media Relations, "Roche in 2011: Strong results and positive outlook," www.roche.com/med-cor-2012-02-01-e.pdf, Feb 1, 2012, p.7
- ↑ "Amgen 2011 Annual Report and Financial Summary," [1] 2011 AnnualReport.pdf, Feb 23 2012, p. 71
- ↑ Zalipsky, Samuel; Pasut, Gianfranco (2020). "Evolution of polymer conjugation to proteins". Polymer-Protein Conjugates. pp. 3–22. doi:10.1016/b978-0-444-64081-9.00001-2. ISBN 9780444640819.
- ↑ Cabanillas, Beatriz; Akdis, Cezmi; Novak, Natalija (2020). "Allergic reactions to the first COVID‐19 vaccine: A potential role of Polyethylene glycol?". Allergy 76 (6): 1617–1618. doi:10.1111/all.14711. PMID 33320974.
- ↑ Weiland, Noah; LaFraniere, Sharon; Baker, Mike; Thomas, Katie (17 December 2020). "2 Alaska Health Workers Got Emergency Treatment After Receiving Pfizer's Vaccine". New York Times. https://www.nytimes.com/2020/12/16/health/covid-pfizer-vaccine-allergic-reaction.html?action=click&module=Top%20Stories&pgtype=Homepage.
- ↑ Firger, Jessica; Caldwell, Travis (19 December 2020). "Third Alaskan health care worker has allergic reaction to Covid-19 vaccine". Cable News Network. https://edition.cnn.com/2020/12/18/health/alaska-third-allergic-reaction-vaccine/index.html.
- ↑ Powers, Marie (May 29, 2018). "Biomarin aces final exam: Palynziq gains FDA approval to treat PKU in adults" (in en). BioWorld. https://www.bioworld.com/articles/331980-biomarin-aces-final-exam-palynziq-gains-full-fda-approval-to-treat-pku-in-adults.
- ↑ Levy, Harvey L.; Sarkissian, Christineh N.; Stevens, Raymond C.; Scriver, Charles R. (June 2018). "Phenylalanine ammonia lyase (PAL): From discovery to enzyme substitution therapy for phenylketonuria". Molecular Genetics and Metabolism 124 (4): 223–229. doi:10.1016/j.ymgme.2018.06.002. PMID 29941359.
- ↑ "FDA approves modified antihemophilic factor for hemophilia A". https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm472643.htm.
- ↑ Auth, Dorothy R.; Powell, Michael B. (14 September 2020). "Patent Issues Highlight Risks of Moderna's COVID-19 Vaccine". New York Law Journal. https://www.law.com/newyorklawjournal/2020/09/14/patent-issues-highlight-risks-of-modernas-covid-19-vaccine/?slreturn=20201101060230. Retrieved 1 December 2020.
- ↑ Vardi, Nathan (29 June 2020). "Moderna's Mysterious Coronavirus Vaccine Delivery System". Forbes. https://www.forbes.com/sites/nathanvardi/2020/07/29/modernas-mysterious-coronavirus-vaccine-delivery-system/?sh=6e8879fb62d9. Retrieved 1 December 2020.
- ↑ "Moderna loses key patent challenge". Nature Biotechnology 38 (9): 1009. September 2020. doi:10.1038/s41587-020-0674-1. PMID 32887970.
- ↑ Pasut, Gianfranco; Zalipsky, Samuel (2020). Polymer-Protein Conjugates: From Pegylation and Beyond. Elsevier. ISBN 978-0-444-64082-6. OCLC 1127111107.[page needed]
- ↑ Balazs, Daniel A.; Godbey, WT (15 December 2011). "Liposomes for Use in Gene Delivery". Journal of Drug Delivery 2011: 326497. doi:10.1155/2011/326497. PMID 21490748.
- ↑ Pasut, Gianfranco; Veronese, Francesco M. (July 2012). "State of the art in PEGylation: The great versatility achieved after forty years of research". Journal of Controlled Release 161 (2): 461–472. doi:10.1016/j.jconrel.2011.10.037. PMID 22094104.
- ↑ Fee, Conan J.; Van Alstine, James M. (2006). "PEG-proteins: Reaction engineering and separation issues". Chemical Engineering Science 61 (3): 924. doi:10.1016/j.ces.2005.04.040.
- ↑ "Protein conjugates purification and characterization". PEGylated protein drugs basic science and clinical applications (Online-Ausg. ed.). Basel: Birkhäuser. 2009. pp. 113–125. ISBN 978-3-7643-8679-5.
- ↑ Fee, Conan J. (2003). "Size-exclusion reaction chromatography (SERC): A new technique for protein PEGylation". Biotechnology and Bioengineering 82 (2): 200–6. doi:10.1002/bit.10561. PMID 12584761.
- ↑ Fee, Conan J.; Damodaran, Vinod B. (2012). "Production of PEGylated Proteins". Biopharmaceutical Production Technology. pp. 199. doi:10.1002/9783527653096.ch7. ISBN 9783527653096.
- ↑ "Polypeptide therapeutics and uses thereof". Wipo (PCT) WO (138413A1). 2016.
- ↑ "Methods and pharmaceutical compositions for treating candida auris in blood". Wipo (PCT) WO (126695A2). 2019.
- ↑ "Arginase formulations and methods". Wipo (PCT) WO (8495A2). 2011.
- ↑ Ryan, Sinéad M; Mantovani, Giuseppe; Wang, Xuexuan; Haddleton, David M; Brayden, David J (2008). "Advances in PEGylation of important biotech molecules: Delivery aspects". Expert Opinion on Drug Delivery 5 (4): 371–83. doi:10.1517/17425247.5.4.371. PMID 18426380.
- ↑ Maso, Katia; Grigoletto, Antonella; Pasut, Gianfranco (2018). "Transglutaminase and Sialyltransferase Enzymatic Approaches for Polymer Conjugation to Proteins". Therapeutic Proteins and Peptides. Advances in Protein Chemistry and Structural Biology. 112. pp. 123–142. doi:10.1016/bs.apcsb.2018.01.003. ISBN 9780128143407.
- ↑ da Silva Freitas, Débora; Mero, Anna; Pasut, Gianfranco (20 March 2013). "Chemical and Enzymatic Site Specific PEGylation of hGH". Bioconjugate Chemistry 24 (3): 456–463. doi:10.1021/bc300594y. PMID 23432141.
- ↑ Kawai, F. (1 January 2002). "Microbial degradation of polyethers". Applied Microbiology and Biotechnology 58 (1): 30–38. doi:10.1007/s00253-001-0850-2. PMID 11831473.
- ↑ Veronese, Francesco M (March 2001). "Peptide and protein PEGylation". Biomaterials 22 (5): 405–417. doi:10.1016/s0142-9612(00)00193-9. PMID 11214751.
- ↑ González-Valdez, José; Rito-Palomares, Marco; Benavides, Jorge (June 2012). "Advances and trends in the design, analysis, and characterization of polymer–protein conjugates for 'PEGylaided' bioprocesses". Analytical and Bioanalytical Chemistry 403 (8): 2225–2235. doi:10.1007/s00216-012-5845-6. PMID 22367287.
- ↑ Zhang, Genghui; Han, Baozhong; Lin, Xiaoyan; Wu, Xin; Yan, Husheng (December 2008). "Modification of Antimicrobial Peptide with Low Molar Mass Poly(ethylene glycol)". The Journal of Biochemistry 144 (6): 781–788. doi:10.1093/jb/mvn134. PMID 18845567.
- ↑ Obuobi, Sybil; Wang, Ying; Khara, Jasmeet Singh; Riegger, Andreas; Kuan, Seah Ling; Ee, Pui Lai Rachel (October 2018). "Antimicrobial and Anti-Biofilm Activities of Surface Engineered Polycationic Albumin Nanoparticles with Reduced Hemolytic Activity". Macromolecular Bioscience 18 (10): 1800196. doi:10.1002/mabi.201800196. PMID 30066983.
- ↑ Zhou, Yang; Petrova, Stella P.; Edgar, Kevin J. (2021-11-15). "Chemical synthesis of polysaccharide–protein and polysaccharide–peptide conjugates: A review" (in en). Carbohydrate Polymers 274: 118662. doi:10.1016/j.carbpol.2021.118662. ISSN 0144-8617. PMID 34702481. https://www.sciencedirect.com/science/article/pii/S0144861721010493.
External links
 |
