Chemistry:Threitol
From HandWiki
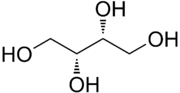
| |
| Names | |
|---|---|
| IUPAC name
D-Threitol[2]
| |
| Systematic IUPAC name
(2R,3R)-Butane-1,2,3,4-tetrol | |
| Identifiers | |
3D model (JSmol)
|
|
| 1719752 | |
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| 1782960 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C4H10O4 | |
| Molar mass | 122.12 |
| Appearance | Solid |
| Melting point | 88 to 90 °C (190 to 194 °F; 361 to 363 K) |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P332+313, P337+313, P362, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Threitol is the chiral four-carbon sugar alcohol with the molecular formula C4H10O4. It is primarily used as an intermediate in the chemical synthesis of other compounds. It exists in the enantiomorphic forms D-threitol and L-threitol, the reduced forms of D- and L-threose. It is the diastereomer of erythritol, which is used as a sugar substitute.
In living organisms, threitol is found in the edible fungus Armillaria mellea.[4] It serves as a cryoprotectant (antifreeze agent) in the Alaskan beetle Upis ceramboides.[5]
See also
- Antifreeze protein
- Dithiothreitol, a thiol derivative of threitol
References
- ↑ Threitol at Sigma-Alrich
- ↑ "2-Carb-19". https://iupac.qmul.ac.uk/2carb/19.html.
- ↑ "D-Threitol" (in en). https://pubchem.ncbi.nlm.nih.gov/compound/169019#section=Safety-and-Hazards.
- ↑ Elks, J.; Ganellin, C. R. (1990). Dictionary of Drugs. doi:10.1007/978-1-4757-2085-3. ISBN 978-1-4757-2087-7.
- ↑ Walters, K. R. Jr; Pan, Q.; Serianni, A. S.; Duman, J. G. (2009). "Cryoprotectant biosynthesis and the selective accumulation of threitol in the freeze-tolerant Alaskan beetle, Upis ceramboides". Journal of Biological Chemistry 284 (25): 16822–16831. doi:10.1074/jbc.M109.013870. PMID 19403530.
External links
 |
