Chemistry:Uranium compounds
Uranium compounds are compounds formed by the element uranium (U). Although uranium is a radioactive actinide, its compounds are well studied due to its long half-life and its applications. It usually forms in the +4 and +6 oxidation states, although it can also form in other oxidation states.
Oxidation states and oxides
Oxides
Calcined uranium yellowcake, as produced in many large mills, contains a distribution of uranium oxidation species in various forms ranging from most oxidized to least oxidized. Particles with short residence times in a calciner will generally be less oxidized than those with long retention times or particles recovered in the stack scrubber. Uranium content is usually referenced to U3O8, which dates to the days of the Manhattan Project when U3O8 was used as an analytical chemistry reporting standard.[1]
Phase relationships in the uranium-oxygen system are complex. The most important oxidation states of uranium are uranium(IV) and uranium(VI), and their two corresponding oxides are, respectively, uranium dioxide (UO2) and uranium trioxide (UO3).[2] Other uranium oxides such as uranium monoxide (UO), diuranium pentoxide (U2O5), and uranium peroxide (UO4·2H2O) also exist.
The most common forms of uranium oxide are triuranium octoxide (U3O8) and UO2.[3] Both oxide forms are solids that have low solubility in water and are relatively stable over a wide range of environmental conditions. Triuranium octoxide is (depending on conditions) the most stable compound of uranium and is the form most commonly found in nature. Uranium dioxide is the form in which uranium is most commonly used as a nuclear reactor fuel.[3] At ambient temperatures, UO2 will gradually convert to U3O8. Because of their stability, uranium oxides are generally considered the preferred chemical form for storage or disposal.[3]
Aqueous chemistry

Salts of many oxidation states of uranium are water-soluble and may be studied in aqueous solutions. The most common ionic forms are U3+ (brown-red), U4+ (green), UO+2 (unstable), and UO2+2 (yellow), for U(III), U(IV), U(V), and U(VI), respectively.[4] A few solid and semi-metallic compounds such as UO and US exist for the formal oxidation state uranium(II), but no simple ions are known to exist in solution for that state. Ions of U3+ liberate hydrogen from water and are therefore considered to be highly unstable. The UO2+2 ion represents the uranium(VI) state and is known to form compounds such as uranyl carbonate, uranyl chloride and uranyl sulfate. UO2+2 also forms complexes with various organic chelating agents, the most commonly encountered of which is uranyl acetate.[4]
Unlike the uranyl salts of uranium and polyatomic ion uranium-oxide cationic forms, the uranates, salts containing a polyatomic uranium-oxide anion, are generally not water-soluble.
Carbonates
The interactions of carbonate anions with uranium(VI) cause the Pourbaix diagram to change greatly when the medium is changed from water to a carbonate containing solution. While the vast majority of carbonates are insoluble in water (students are often taught that all carbonates other than those of alkali metals are insoluble in water), uranium carbonates are often soluble in water. This is because a U(VI) cation is able to bind two terminal oxides and three or more carbonates to form anionic complexes.
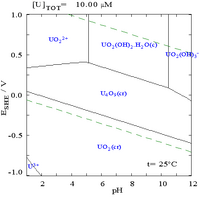 |
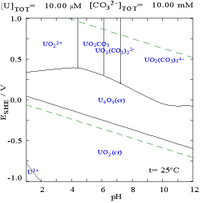 |
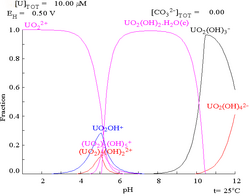
| Uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[5] | Uranium in carbonate solution |
 |
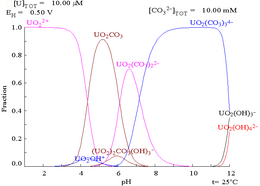 |
| Relative concentrations of the different chemical forms of uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[5] | Relative concentrations of the different chemical forms of uranium in an aqueous carbonate solution.[5] |
Effects of pH
The uranium fraction diagrams in the presence of carbonate illustrate this further: when the pH of a uranium(VI) solution increases, the uranium is converted to a hydrated uranium oxide hydroxide and at high pHs it becomes an anionic hydroxide complex.
When carbonate is added, uranium is converted to a series of carbonate complexes if the pH is increased. One effect of these reactions is increased solubility of uranium in the pH range 6 to 8, a fact that has a direct bearing on the long term stability of spent uranium dioxide nuclear fuels.
Hydrides, carbides and nitrides
Uranium metal heated to 250 to 300 °C (482 to 572 °F) reacts with hydrogen to form uranium hydride. Even higher temperatures will reversibly remove the hydrogen. This property makes uranium hydrides convenient starting materials to create reactive uranium powder along with various uranium carbide, nitride, and halide compounds.[6] Two crystal modifications of uranium hydride exist: an α form that is obtained at low temperatures and a β form that is created when the formation temperature is above 250 °C.[6]
Uranium carbides and uranium nitrides are both relatively inert semimetallic compounds that are minimally soluble in acids, react with water, and can ignite in air to form U3O8.[6] Carbides of uranium include uranium monocarbide (U C), uranium dicarbide (UC2), and diuranium tricarbide (U2C3). Both UC and UC2 are formed by adding carbon to molten uranium or by exposing the metal to carbon monoxide at high temperatures. Stable below 1800 °C, U2C3 is prepared by subjecting a heated mixture of UC and UC2 to mechanical stress.[7] Uranium nitrides obtained by direct exposure of the metal to nitrogen include uranium mononitride (UN), uranium dinitride (UN2), and diuranium trinitride (U2N3).[7]
Halides

All uranium fluorides are created using uranium tetrafluoride (UF4); UF4 itself is prepared by hydrofluorination of uranium dioxide.[6] Reduction of UF4 with hydrogen at 1000 °C produces uranium trifluoride (UF3). Under the right conditions of temperature and pressure, the reaction of solid UF4 with gaseous uranium hexafluoride (UF6) can form the intermediate fluorides of U2F9, U4F17, and UF5.[6]
At room temperatures, UF6 has a high vapor pressure, making it useful in the gaseous diffusion process to separate the rare uranium-235 from the common uranium-238 isotope. This compound can be prepared from uranium dioxide and uranium hydride by the following process:[6]
- UO2 + 4 HF → UF4 + 2 H2O (500 °C, endothermic)
- UF4 + F2 → UF6 (350 °C, endothermic)
The resulting UF6, a white solid, is highly reactive (by fluorination), easily sublimes (emitting a vapor that behaves as a nearly ideal gas), and is the most volatile compound of uranium known to exist.[6]
One method of preparing uranium tetrachloride (UCl4) is to directly combine chlorine with either uranium metal or uranium hydride. The reduction of UCl4 by hydrogen produces uranium trichloride (UCl3) while the higher chlorides of uranium are prepared by reaction with additional chlorine.[6] All uranium chlorides react with water and air.
Bromides and iodides of uranium are formed by direct reaction of, respectively, bromine and iodine with uranium or by adding UH3 to those element's acids.[6] Known examples include: UBr3, UBr4, UI3, and UI4. UI5 has never been prepared. Uranium oxyhalides are water-soluble and include UO2F2, UOCl2, UO2Cl2, and UO2Br2. Stability of the oxyhalides decrease as the atomic weight of the component halide increases.[6]
See also
Citations
- ↑ Kloprogge 2021, pp. 861–862.
- ↑ Seaborg 1968, p. 779.
- ↑ 3.0 3.1 3.2 Argonne Nat. Lab.
- ↑ 4.0 4.1 Seaborg 1968, p. 778.
- ↑ 5.0 5.1 5.2 5.3 Puigdomenech, Ignasi Hydra/Medusa Chemical Equilibrium Database and Plotting Software (2004) KTH Royal Institute of Technology, freely downloadable software at (Department of Chemistry {{{2}}})
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 Seaborg 1968, p. 782.
- ↑ 7.0 7.1 Seaborg 1968, p. 780.
Sources
- "Chemical Equilibrium Software". Department of Chemistry, KTH Royal Institute of Technology. http://www.kemi.kth.se/medusa/.
- "Chemical Forms of Uranium". Argonne National Laboratory. http://web.ead.anl.gov/uranium/guide/ucompound/forms/index.cfm.
- Kloprogge, J. Theo (2021). The periodic table: nature's building blocks: an introduction to the naturally occurring elements, their origins and their uses. Concepcion P. Ponce, Tom A. Loomis. Amsterdam: Elsevier. pp. 861–862. ISBN 978-0-12-821538-8. OCLC 1223058470. https://www.worldcat.org/oclc/1223058470.
- Seaborg, Glenn T. (1968). "Uranium". The Encyclopedia of the Chemical Elements. Skokie, Illinois: Reinhold Book Corporation. pp. 773–786.
 |