Medicine:Cervical cancer
| Cervical cancer | |
|---|---|
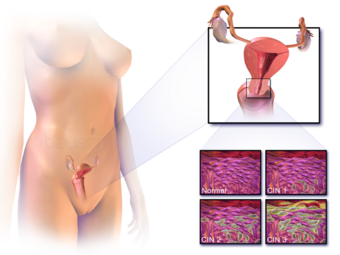 | |
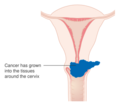
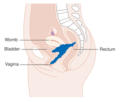
| Location of cervical cancer and an example of normal and abnormal cells | |
| Pronunciation | |
| Specialty | Gynecologic oncology |
| Symptoms | Early: none[2] Later: vaginal bleeding, pelvic pain, pain during sexual intercourse[2] |
| Usual onset | Over 10 to 20 years[3] |
| Types | Squamous cell carcinoma, adenocarcinoma, others[4] |
| Causes | Human papillomavirus infection (HPV)[5][6] |
| Risk factors | Smoking, weak immune system, birth control pills, starting sex at a young age, many sexual partners or a partner with many sexual partners[2][4][7] |
| Diagnostic method | Cervical screening followed by a biopsy[2] |
| Prevention | Regular cervical screening, HPV vaccine, sexual intercourse with condoms,[8][9] sexual abstinence |
| Treatment | Surgery, chemotherapy, radiation therapy, immunotherapy[2] |
| Prognosis | Five-year survival rate: 68% (US) 46% (India)[10] |
| Frequency | 604,127 new cases (2020)[11] |
| Deaths | 341,831 (2020)[11] |
Cervical cancer is a type of cancer that develops in the cervix or in any layer of the wall of the cervix.[2] It is due to the abnormal growth of cells that can invade or spread to other parts of the body.[12] Early on, typically no symptoms are seen.[2] Later symptoms may include abnormal vaginal bleeding, pelvic pain or pain during sexual intercourse.[2] While bleeding after sex may not be serious, it may also indicate the presence of cervical cancer.[13]
Virtually all cervical cancer cases (99%) are linked to genital human papillomavirus infection (HPV);[14][5][6] most who have had HPV infections, however, do not develop cervical cancer.[3][15] HPV 16 and 18 strains are responsible for approximately 70% of cervical cancer cases globally and nearly 50% of high-grade cervical pre-cancers.[16][17] Minor risk factors include smoking, a weak immune system, birth control pills, starting sex at a young age, and having many sexual partners.[2][4] Genetic factors also contribute to cervical cancer risk.[18] Cervical cancer typically develops from precancerous changes called cervical intraepithelial neoplasia over 10 to 20 years.[3] About 75% of cervical cancers are squamous cell carcinomas, 20-25% are adenocarcinoma, 3% are adenosquamous carcinomas, and less than 1% are small cell neuroendocrine tumors of the cervix.[19] Diagnosis is typically by cervical screening followed by a biopsy.[2] Medical imaging is then done to determine whether or not the cancer has spread beyond the cervix.[2]
HPV vaccination is the most cost-effective public health measure against cervical cancer.[14] There are six licensed HPV vaccines.[16] They protect against two to seven high-risk strains of this family of viruses. They may prevent up to 90% of cervical cancers.[9][20][21] By the end of 2023, 143 countries (74% of WHO member states) provided the HPV vaccine in their national immunization schedule for girls.[22] As of 2022, 47 countries (24% of WHO member states) also did it for boys.[23]: 654 As a risk of cancer still exists, guidelines recommend continuing regular Pap tests.[9] Other methods of prevention include having few or no sexual partners and the use of condoms.[8] Cervical cancer screening using the Pap test or acetic acid can identify precancerous changes, which when treated, can prevent the development of cancer.[24] Treatment may consist of some combination of surgery, chemotherapy, and radiation therapy.[2] Five-year survival rates in the United States are 68%.[25] Outcomes, however, depend very much on how early the cancer is detected.[4]
Worldwide, cervical cancer is both the fourth-most common type of cancer and the fourth-most common cause of death from cancer in women, with over 660,000 new cases and around 350,000 deaths in 2022.[3][26] This is about 8% of the total cases and total deaths from cancer.[27] 88% (2020 figure) of cervical cancers and 90% of deaths occur in low- and middle-income countries and 2% (2020 figure) in high-income countries.[23]: 650 [28][3][29] Of the 20 hardest hit countries by cervical cancer, 19 are in Africa.[30] In low-income countries, it is one of the most common causes of cancer death with an incidence rate of 47.3 per 100,000 women.[31][24] In developed countries, the widespread use of cervical screening programs has dramatically reduced rates of cervical cancer.[32] Expected scenarios for the reduction of mortality due to cervical cancer worldwide (and specially in low-income countries) have been reviewed, given assumptions with respect to the achievement of recommended prevention targets using triple-intervention strategies defined by WHO.[33] In medical research, the most famous immortalized cell line, known as HeLa, was developed from cervical cancer cells of a woman named Henrietta Lacks.[34]
17 November is the Cervical Cancer Elimination Day of Action.[35] The date marks the day in 2020 when WHO launched the Global strategy to accelerate the elimination of cervical cancer as a public health problem, with a resolution passed by 194 countries.[35] To eliminate cervical cancer, all countries must reach and maintain an incidence rate of below 4 per 100 000 women.[36][37]
Signs and symptoms
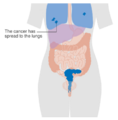
The early stages of cervical cancer may be completely free of symptoms.[5][32] Vaginal bleeding, contact bleeding (one most common form being bleeding after sexual intercourse), or (rarely) a vaginal mass may indicate the presence of cervical cancer. Also, moderate pain during sexual intercourse and vaginal discharge are symptoms of cervical cancer.[38] Bleeding after douching or after a pelvic exam is a common symptom of cervical cancer.[39] In advanced disease, metastases may be present in the abdomen, lungs, or elsewhere.[40]
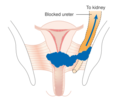
Symptoms of advanced cervical cancer may include loss of appetite, weight loss, fatigue, pelvic pain, back pain, leg pain, swollen legs, heavy vaginal bleeding, bone fractures, and (rarely) leakage of urine or faeces from the vagina.[41] Other signs of locally advanced disease (as the cancer invades organs in the pelvis) include hydronephrosis with flank pain as the ureters directing urine from the kidneys to bladder are blocked, leg swelling and blood clots in the legs as pelvic veins are blocked, rectal bleeding, and bleeding in the urine.[19]
Causes
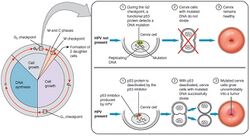
Infection with some types of HPV is the greatest risk factor for cervical cancer, followed by smoking.[42] HIV infection is also a risk factor.[42] Not all of the causes of cervical cancer are known, however, and several other contributing factors have been implicated.[43][44]
Human papillomavirus
Infection with HPV is thought to be required for cervical cancer to occur.[45] HPV types 16 and 18 are the cause of 75% of cervical cancer cases globally, while 31 and 45 are the causes of another 10%.[46]
Women who have multiple sexual partners, or have partners who have multiple sexual partners, regardless of sex, are at higher risk of cervical cancer.[47][48]
Over 200 types of HPV known,[49] 12 are classified as high-risk types (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59),[49] three as probable high-risk (26, 53, and 66), and 12 as low-risk (6, 11, 40, 42, 43, 44, 54, 61, 70, 72, 81, and CP6108).[50] Most cases of squamous cell carcinomas of the cervix are due to HPV type 16 and most cases of adenocarcinoma are due to HPV type 18.[19] High risk HPV viral subtypes can integrate their DNA into the host genome and induce transcription of the viral cancer causing proteins E6 and E7.[19][51] E6 degrades the tumor suppressing protein p53 and E7 degrades and inactivates the tumor suppressing protein pRb. The loss of p53 and pRb leads to increased blood vessel growth feeding tumors (via vascular endothelial growth factor(VEGF) over-expression), loss of tumor cell suppression and cell cycle regulation disruptions, all of which can lead to cervical cancer.[19]
Genital warts, which are a form of benign tumor of epithelial cells, are also caused by various strains of HPV. However, these serotypes are usually not related to cervical cancer. Having multiple strains at the same time is common, including those that can cause cervical cancer along with those that cause warts.
Smoking

Cigarette smoking, both active and passive, increases the risk of cervical cancer. Among HPV-infected women, current and former smokers have roughly two to three times the incidence of invasive cancer. Passive smoking is also associated with increased risk, but to a lesser extent.[52]
Smoking has also been linked to the development of cervical cancer.[53][54][42] Smoking can increase the risk in women a few different ways, which can be by direct and indirect methods of inducing cervical cancer.[53][42][55] A direct way of contracting this cancer is that someone who smokes has a higher chance of cervical intraepithelial neoplasia (CIN3) occurring, which has the potential of forming cervical cancer.[53] When CIN3 lesions lead to cancer, most of them have the assistance of the HPV virus, but that is not always the case, which is why it can be considered a direct link to cervical cancer.[55] Heavy smoking and long-term smoking seem to have a higher risk of getting the CIN3 lesions than lighter smoking or not smoking at all.[56] Although smoking has been linked to cervical cancer, it aids in the development of HPV, which is the leading cause of this type of cancer.[42] Also, not only does it aid in the development of HPV, but also if the woman is already HPV-positive, she is at an even greater likelihood of contracting cervical cancer.[56]
Oral contraceptives
Long-term use of oral contraceptives is associated with increased risk of cervical cancer in women who have had HPV. Women who have used oral contraceptives for 5 to 9 years have about three times the incidence of invasive cancer, and those who used them for 10 years or longer have about four times the risk.[52][57]
Multiple pregnancies
Having many pregnancies is associated with an increased risk of cervical cancer. Among HPV-infected women, those who have had seven or more full-term pregnancies have around four times the risk of cancer compared with women with no pregnancies, and two to three times the risk of women who have had one or two full-term pregnancies.[52]
Diagnosis
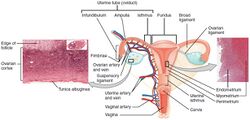
Biopsy
The Pap test can be used as a screening test, but it produces a false negative in up to 50% of cases of cervical cancer.[58][59] Another concern is the cost of doing Pap tests, which makes them unaffordable in many areas of the world.[60]
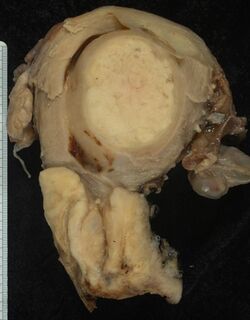
Confirmation of the diagnosis of cervical cancer or precancer requires a biopsy of the cervix. This is often done through colposcopy, a magnified visual inspection of the cervix aided by using a dilute acetic acid (e.g. vinegar) solution to highlight abnormal cells on the surface of the cervix,[5] with visual contrast provided by staining the normal tissues a mahogany brown with Lugol's iodine.[61] Medical devices used for biopsy of the cervix include punch forceps. Colposcopic impression, the estimate of disease severity based on the visual inspection, forms part of the diagnosis. Further diagnostic and treatment procedures are loop electrical excision procedure and cervical conization, in which the inner lining of the cervix is removed to be examined pathologically. These are carried out if the biopsy confirms severe cervical intraepithelial neoplasia.[62][63]
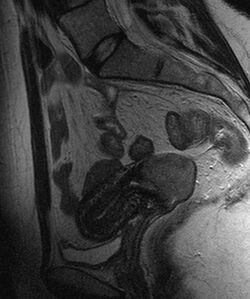
Often, before the biopsy, the doctor asks for medical imaging to rule out other causes of a woman's symptoms. Imaging modalities such as ultrasound, CT scan, and MRI have been used to look for alternative disease, spread of the tumor, and effect on adjacent structures. Typically, they appear as heterogeneous masses on the cervix.[64]
Interventions such as playing music during the procedure and viewing the procedure on a monitor can reduce the anxiety associated with the examination.[65]
Precancerous lesions

Cervical intraepithelial neoplasia (CIN) means the development of abnormal cells on the surface of the cervix. HPV infections cause CIN, but in most cases, it is resolved by the immune system. However, a small percentage of people might develop a more serious CIN, which, if left untreated, can develop into cervical cancer.[66][67] CIN is often diagnosed during routine Pap smear examination or colposcopy.[67]
The naming and histologic classification of cervical carcinoma precursor lesions has changed many times over the 20th century. The World Health Organization classification system was descriptive of the lesions, naming them mild, moderate, or severe dysplasia or carcinoma in situ (CIS).[68][69] The term cervical intraepithelial neoplasia (CIN) was developed to place emphasis on the spectrum of abnormality in these lesions and to help standardize treatment.[69] For premalignant dysplastic changes, cervical intraepithelial neoplasia grading (CIN 1–3) is used. It classifies mild dysplasia as CIN1, moderate dysplasia as CIN2, and severe dysplasia and CIS as CIN3.[70] More recently, CIN2 and CIN3 have been combined into CIN2/3. These results are what a pathologist might report from a biopsy. These should not be confused with the Bethesda system terms for Pap test (cytopathology) results. Among the Bethesda results: Low-grade squamous intraepithelial lesion (LSIL) and high-grade squamous intraepithelial lesion (HSIL). An LSIL Pap may correspond to CIN1, and HSIL may correspond to CIN2 and CIN3,[69] but they are results of different tests, and the Pap test results need not match the histologic findings.
Cancer subtypes
Histologic subtypes of invasive cervical carcinoma include:[71][72]
- Squamous cell carcinoma (about 80–85%[73][74])
- adenocarcinoma (about 15% of cervical cancers in the UK[68])
- Adenosquamous carcinoma
- Small cell carcinoma
- Neuroendocrine tumour
- Glassy cell carcinoma
- Villoglandular adenocarcinoma
-
Invasive squamous cell carcinoma of the cervix is characterized by infiltration as irregular anastomosing nests or single cells.[75] This case is poorly differentiated. H&E stain.
-
Cervical squamous cell carcinoma generally shows diffuse staining of both nuclei and cytoplasm on p16 immuno-
histochemistry (except verrucous variant).[76] -
Invasive cervical squamous cell carcinoma on H&E histopathology and Ki-67 immunohistochemistry. The latter correlates well with the degree and level of dysplasia.[77]
-
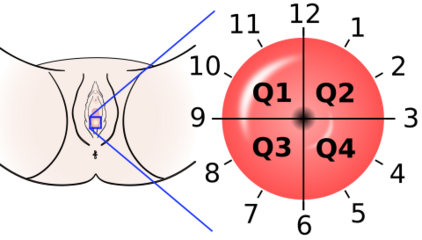
The location of cervical cancer can be described in terms of quadrants, or corresponding to a clock face when the subject is in supine position.
Though squamous cell carcinoma is the cervical cancer with the highest incidence, the incidence of adenocarcinoma of the cervix has been increasing in recent decades.[5] Endocervical adenocarcinoma represents 20–25% of the histological types of cervical carcinoma. Gastric-type mucinous adenocarcinoma of the cervix is a rare type of cancer with aggressive behavior. This type of malignancy is not related to high-risk human papillomavirus (HPV).[78]
Staging
Cervical cancer is staged by the FIGO system, which is based on clinical examination rather than surgical findings. Before the 2018 revisions to FIGO staging, the system allowed only these diagnostic tests to be used in determining the stage: palpation, inspection, colposcopy, endocervical curettage, hysteroscopy, cystoscopy, proctoscopy, intravenous urography, and X-ray examination of the lungs and skeleton, and cervical conization. However, the system allows the use of any imaging or pathological methods for staging.[79]
-
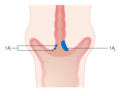
Stage 1A cervical cancer
-
Stage 1B cervical cancer
-
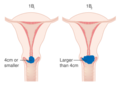
Stage 2A cervical cancer
-
Stage 2B cervical cancer
-
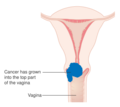
Stage 3B cervical cancer
-
Stage 4A cervical cancer
-
Stage 4B cervical cancer
Prevention
Screening

Checking cervical cells with the Papanicolaou test (Pap test) for cervical pre-cancer has dramatically reduced the number of cases of, and mortality from, cervical cancer.[32] Liquid-based cytology may reduce the number of inadequate samples.[80][81][82] Pap test screening every three to five years with appropriate follow-up can reduce cervical cancer incidence up to 80%.[83]
Pap test screening can reveal abnormal cells on the surface of the cervix called cervical intraepithelial neoplasia (CIN), which in a small percentage can develop into cervical cancer. These precancerous changes can be confirmed with further examination known as colposcopy.
Personal invitations encouraging women to get screened are effective at increasing the likelihood that they will do so. Educational materials also help increase the likelihood that women will go for screening, but they are not as effective as invitations.[84]
According to the 2010 European guidelines, the age at which to start screening ranges between 20 and 30 years of age, but preferentially not before age 25 or 30 years, and depends on the burden of the disease in the population and the available resources.[83]
In the United States, screening is recommended to begin at age 21, regardless of the age at which a woman began having sex or other risk factors.[85] Pap tests should be done every three years between the ages of 21 and 65.[85] In women over the age of 65, screening may be discontinued if no abnormal screening results were seen within the previous 10 years and no history of CIN2 or higher exists.[85][86][87] HPV vaccination status does not change screening rates.[86]
A number of recommended options exist for screening those 30 to 65.[88] This includes cervical cytology every 3 years, HPV testing every 5 years, or HPV testing together with cytology every 5 years.[88][86] Screening is not beneficial before age 25, as the rate of disease is low. Screening is not beneficial in women older than 60 years if they have a history of negative results.[52] The American Society of Clinical Oncology guideline has recommend for different levels of resource availability.[89]
Pap tests have not been as effective in developing countries.[90] This is in part because many of these countries have an impoverished health care infrastructure, too few trained and skilled professionals to obtain and interpret Pap tests, uninformed women who get lost to follow-up, and a lengthy turn-around time to get results.[90] Visual inspection with acetic acid and HPV DNA testing has been tried, though with mixed success.[90]
Vaccination
There are six licensed HPV vaccines: three bivalent (protect against two types of HPV), two quadrivalent (against four), and one nonavalent vaccine (against nine)[16] Three HPV vaccines (Gardasil, Gardasil 9, and Cervarix) reduce the risk of cancerous or precancerous changes of the cervix and perineum by about 93% and 62%, respectively.[91] All have excellent safety profiles and are highly efficacious, or have met immunobridging standards.[23]: 668 The vaccines are between 92% and 100% effective against HPV 16 and 18 up to at least 8 years.[52]
HPV vaccines are typically given to people ages 9 to 26, as the vaccine is most effective if given before infection occurs. The primary target group in most of the countries recommending HPV vaccination is young adolescent girls, aged 9-14.[16] As of 2022, 125 countries include HPV vaccines in their routine vaccinations for girls, and 47 countries recommend them for boys, as well,[23]: 654 including Japan.[92]
The duration of effectiveness and whether a booster will be needed is unknown. The high cost of this vaccine has been a cause for concern. Several countries have considered (or are considering) programs to fund HPV vaccination. The American Society of Clinical Oncology guideline has recommendations for different levels of resource availability.[89]
Barrier protection
Barrier protection or spermicidal gel use during sexual intercourse decreases but does not eliminate the risk of transmitting the infection.[52] Condoms may protect against genital warts.[93] They also protect against other sexually transmitted infections, such as HIV and Chlamydia, which are associated with greater risks of developing cervical cancer.
Nutrition
Vitamin A is associated with a lower risk[94] as are vitamin B12, vitamin C, vitamin E, and beta-Carotene.[95]
Treatment
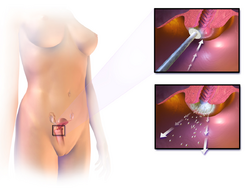
The treatment of cervical cancer varies worldwide, largely due to access to surgeons skilled in radical pelvic surgery and the emergence of fertility-sparing therapy in developed nations. Less advanced stages of cervical cancer typically have treatment options that allow fertility to be maintained if the patient desires.[96]
Because cervical cancers are radiosensitive, radiation may be used in all stages where surgical options do not exist. Surgical intervention may have better outcomes than radiological approaches.[97] In addition, chemotherapy can be used to treat cervical cancer and is more effective than radiation alone.[98] Chemoradiotherapy may increase overall survival and reduce the risk of disease recurrence compared to radiotherapy alone.[99]
For surgery to be curative, the entire cancer must be removed with no cancer found at the margins of the removed tissue on examination under a microscope.[100] This procedure is known as exenteration.[100]
Precancerous lesions
Precancerous cells (cervical intraepithelial neoplasia) that would lead to cancer and early-stage cervical cancer (IA1) can be treated effectively by various surgical techniques. Surgical treatment methods include excision, where a cone-shaped portion of the cervix is removed, and ablation, which removes only the parts with abnormal tissues. While these effectively reduce the risk of cancer developing or spreading, they cause an increased risk of premature birth in future pregnancies. Surgical techniques that remove more cervical tissue come with less risk of the cancer recurring but a higher chance of giving birth prematurely. Due to this risk, taking into account the age, childbearing plans of the woman, and the size and location of the cancer cells are crucial for choosing the right procedure.[62][63] There is low-certainty evidence that peri-operative care approaches, such as 'fast-track surgery' or 'enhanced recovery programmes' may lower surgical stress and improve recovery after gynaecological cancer surgery.[101]
Microinvasive cancer

Microinvasive cancer (stage IA) may also be treated by hysterectomy (removal of the whole uterus, including part of the vagina).[102] For stage IA2, the lymph nodes are removed as well. Alternatives include local surgical procedures such as a loop electrical excision procedure or cone biopsy.[103][104] A systematic review concluded that more evidence is needed to inform decisions about different surgical techniques for women with cervical cancer at stage IA2.[105]
If a cone biopsy does not produce clear margins[106] (findings on biopsy showing that the tumor is surrounded by cancer free tissue, suggesting all of the tumor is removed), one more possible treatment option for women who want to preserve their fertility is a trachelectomy.[107] This attempts to surgically remove the cancer while preserving the ovaries and uterus, providing for a more conservative operation than a hysterectomy. It is a viable option for those in stage I cervical cancer which has not spread; however, it is not yet considered a standard of care,[108] as few doctors are skilled in this procedure. Even the most experienced surgeon cannot promise that a trachelectomy can be performed until after surgical microscopic examination, as the extent of the spread of cancer is unknown. If the surgeon is not able to microscopically confirm clear margins of cervical tissue once the woman is under general anaesthesia in the operating room, a hysterectomy may still be needed. This can only be done during the same operation if the woman consents. Due to the possible risk of cancer spreading to the lymph nodes in stage 1B cancers and some stage 1A cancers, the surgeon may also need to remove some lymph nodes from around the uterus for pathologic evaluation. A radical trachelectomy can be performed abdominally[109] or vaginally[110] and opinions are conflicting as to which is better.[111] A radical abdominal trachelectomy with lymphadenectomy usually only requires a two- to three-day hospital stay, and most women recover very quickly (about six weeks). Complications are uncommon, although women who can conceive after surgery are susceptible to preterm labour and possible late miscarriage.[112] A wait of at least one year is generally recommended before attempting to become pregnant after surgery.[113] Recurrence in the residual cervix is rare if the trachelectomy has cleared the cancer.[108] Yet, women are recommended to practice vigilant prevention and follow-up care, including Pap screenings/colposcopy, with biopsies of the remaining lower uterine segment as needed (every 3–4 months for at least 5 years) to monitor for any recurrence in addition to minimizing any new exposures to HPV through safe sex practices until one is actively trying to conceive.
Radical hysterectomy and chemoradiation
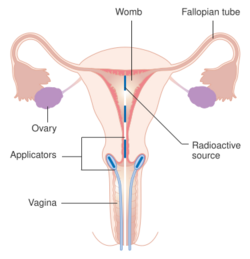
Early stages (IB1 and IIA less than 4 cm) can be treated with radical hysterectomy with removal of the lymph nodes or radiation therapy. Radiation therapy is given as external beam radiotherapy to the pelvis and brachytherapy (internal radiation). Women treated with surgery who have high-risk features found on pathologic examination are given radiation therapy with or without chemotherapy to reduce the risk of relapse. A Cochrane review has found moderate-certainty evidence that radiation decreases the risk of disease progression in people with stage IB cervical cancer when compared to no further treatment.[114] However, little evidence was found on its effects on overall survival.[114]
Larger early-stage tumors (IB2 and IIA more than 4 cm) may be treated with radiation therapy and cisplatin-based chemotherapy, hysterectomy (which then usually requires adjuvant radiation therapy), or cisplatin chemotherapy followed by hysterectomy. When cisplatin is present, it is thought to be the most active single agent in periodic diseases.[115] Such addition of platinum-based chemotherapy to chemoradiation seems not only to improve survival but also reduces risk of recurrence in women with early-stage cervical cancer (IA2–IIA).[116] A Cochrane review found a lack of evidence on the benefits and harms of primary hysterectomy compared to primary chemoradiotherapy for cervical cancer in stage IB2.[117]
Advanced-stage tumors (IIB-IVA) are treated with radiation therapy and cisplatin-based chemotherapy. On 15 June 2006, the US Food and Drug Administration approved the use of a combination of two chemotherapy drugs, hycamtin and cisplatin, for women with late-stage (IVB) cervical cancer treatment.[118] Combination treatment has significant risk of neutropenia, anemia, and thrombocytopenia side effects.[119]
There is insufficient evidence on whether anticancer drugs after standard care help women with locally advanced cervical cancer live longer.[120]
No evidence is available to suggest that any form of follow-up approach is better or worse in terms of prolonging survival, improving quality of life, or guiding the management of problems that can arise because of the treatment, and that in the case of radiotherapy treatment, worsen with time.[121] A 2019 review found no controlled trials regarding the efficacy and safety of interventions for vaginal bleeding in women with advanced cervical cancer.[122]
-
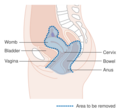
Diagram showing the area removed with a posterior surgery
-
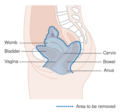
Diagram showing the area removed with a total operation
-
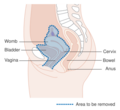
Diagram showing the area removed with an anterior operation
Immunotherapy
Immunotherapy with immune checkpoint inhibitors, such as pembrolizumab (Keytruda), has also been approved by the U.S. Food and Drug Administration (FDA) for certain patients with recurrent or metastatic cervical cancer, demonstrating promising results in ongoing clinical trials.[123][124] In October 2021, the FDA expanded this approval to include pembrolizumab in combination with chemotherapy, with or without bevacizumab, for people with persistent, recurrent, or metastatic cervical cancer, underscoring the potential of immunotherapeutic approaches in this setting.[125] Additional immunotherapy agents, including other PD-1 and PD-L1 inhibitors, are under investigation and have similarly shown encouraging outcomes in clinical studies.
Another immune checkpoint inhibitor, cemiplimab-rwlc (Libtayo), received FDA approval in September 2022 for patients with recurrent or metastatic cervical cancer that has progressed on or after chemotherapy, further highlighting the expanding role of immunotherapeutic strategies in advanced disease.[126]
Tisotumab vedotin (Tivdak) was approved for medical use in the United States in September 2021.[127][128]
Prognosis
Stage
The prognosis depends on the stage of the cancer. The prognosis for squamous cell carcinoma and adenocarcinoma of the cervix is the same for each given stage.[19] For intraepithelial cervical neoplasms, the prognosis is good.[129] With treatment, the five-year survival rate for FIGO stage 1 (cancer confined to the cervix) cervical cancer is 85%, and the overall (all stages combined) five-year survival rate is about 66%.[19][130] Five-year survival in Stage 2 disease (cancer invading beyond upper two-thirds of uterus) is 65%.[19] Stage 3 disease (in which the lower one-third of vagina, pelvic wall is involved or presence of hydronephrosis, pelvic or peri-aortic lymph node involvement) is 35%. Stage 4 disease, in which cancer extends beyond the pelvis, or involves the bladder or rectum, has a 5-year survival rate of 7%.[19]
About 35% of women with invasive cervical cancer have persistent or recurrent disease after treatment.[131]
By country
There is an ethnic disparity in five-year survival in the United States. Average survival rates of the dominant squamous cell carcinoma are 72% for Hispanic and Asian-Pacific women, 68% for White women and 61% for Black women.[132]
Regular screening has meant that precancerous changes and early-stage cervical cancers have been detected and treated early. Figures suggest that cervical screening is saving 5,000 lives each year in the UK by preventing cervical cancer.[133] About 1,000 women per year die of cervical cancer in the UK. All of the Nordic countries have cervical cancer screening programs in place.[134] The Pap test was integrated into clinical practice in the Nordic countries in the 1960s.[134]
In Africa, outcomes are often worse as diagnosis is frequently at a later stage of the disease.[135] In a scoping review of publicly-available cervical cancer prevention and control plans from African countries, plans tended to emphasize survivorship rather than early HPV diagnosis and prevention.[136]
Adverse effects
Chemotherapy works by attacking cells that rapidly divide. This kills cancer cells, but can also impact normal cells, leading to adverse side effects. Common chemotherapy side effects include: hair loss, mouth sores, loss of appetite, diarrhea, nausea and vomiting, premature menopause, infertility, and damage to the blood-forming cells within bone marrow. Most acute side effects are temporary, dissipating when treatment ceases, but some can be long-lasting or permanent. Long-term chemotherapy side effects include changes in the menstrual cycle, neuropathy, and nephrotoxicity.[137]
Radiation therapy (RT) adverse effects; for a complete side effect list see
Curative cervical radiation therapy may affect unintended tissues located within the delivery pathway(s) or adjacent to the target lesion, each tissue with a unique sensitivity and response to radiation injury. Common acute RT effects involve the gastrointestinal system, e.g., diarrhea and constipation; urinary tract, e.g., frequent urination; and may cause cervicitis. Common late RT complications include: infertility or premature ovarian failure; vaginal stenosis; lower motor neuron syndrome; telangiectasias, and subsequent hemorrhage; and progressive myelopathy, which may result in irreversible neurologic deficits ranging from minor sensory symptoms to complete paraplegia.[138] Radiotherapy late effects (with occurrence rates) include osteonecrosis (8-20%), bladder ulceration (<3%), vaginal stenosis (>2.5%)[139] and chronic pelvic radiation disease (1-10%), e.g., irreversible lumbosacral plexopathy.[140]
Pelvic radiation also induces secondary malignancies such as leukemia, lymphoma, bladder cancer, pelvic malignancy, colorectal cancer, bone, and soft-tissue sarcoma with occurrence rates between 0.2 and 1.0% per year for each.[138]
Epidemiology
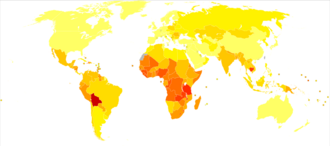
| No data <2.4 2.4–4.8 4.8–7.2 7.2–9.6 9.6–12 12–14.4 | 14.4–16.8 16.8–19.2 19.2–21.6 21.6–24 24–26.4 >26.4 |
Worldwide, cervical cancer is both the fourth-most common type of cancer and the fourth-most common cause of death from cancer in women, with over 660,000 new cases and around 350,000 deaths in 2022.[3][26] It is the second-most common cause of female-specific cancer after breast cancer, accounting for around 8% of both total cancer cases and total cancer deaths in women.[27] 88% (2020 figure) of cervical cancers and 90% of deaths occur in low- and middle-income countries (LMICs) and 2% (2020 figure) in high-income countries (HICs).[23]: 650 [28][142] It is the most frequently detected cancer during pregnancy, with an occurrence of 1.5 to 12 for every 100,000 pregnancies.[143]
The large majority of cervical cancer cases in 2020 (88%) occurred in LMICs, where they account for 17% of all cancers in women, compared with only 2% in high-income countries (HICs). In sub-Saharan Africa, the region with the highest rates of young women living with HIV (WLWH), approximately 20% of cervical cancer cases occur in WLWH. HPV infection is more likely to persist and to progress to cancer in WLWH. Mortality rates vary 50-fold between countries, ranging from <2 per 100,000 women in some HICs to >40 per 100,000 in some countries of sub-Saharan Africa.[23]: 650
Africa
Of the 20 hardest hit countries by cervical cancer, 19 are in Africa.[30]
Australia
Australia is on target to eliminate cervical cancer.[36] It anticipates achieving this in the next 10 years.[36]
In 2022, it is estimated that 942 new cases of cervical cancer will be diagnosed in Australia. In 2022, it is estimated that a female has a 1 in 180 (or 0.56%) risk of being diagnosed with cervical cancer by the age of 85.[144]
In 2020, there were 165 women aged 25–74 who died from cervical cancer, which is a mortality rate of 2 deaths per 100,000 women in the population. Over the 5 years 2016–2020, there were 62 Aboriginal and Torres Strait Islander women aged 25–74 who died from cervical cancer, which is a mortality rate of 7 deaths per 100,000 Indigenous women in the population. Over the 5 years 2016–2020, the age-standardised mortality rate among Aboriginal and Torres Strait Islander women was 3.8 times the rate of non-Indigenous Australians.[145]
The number of women diagnosed with cervical cancer has dropped on average by 4.5% each year since organised screening began in 1991 (1991–2005).[146] Regular twice-yearly Pap tests [this is out of date] can reduce the incidence of cervical cancer up to 90% in Australia, and save 1,200 Australian women from dying from the disease each year.[147] It is predicted that because of the success of the primary HPV testing programme there will be fewer than four new cases per 100 000 women annually by 2028.[148]
Nepal
Hundreds of women in Nepal are diagnosed with cervical cancer each year, primarily caused by human papillomavirus (HPV) infection. In February 2025, Nepal launched a nationwide HPV vaccination campaign targeting over 1.6 million girls aged 10–15, supported by WHO, UNICEF, and Gavi, alongside educational programs to raise awareness and prevent cervical cancer [149]
Canada
An estimated 1,450 Canadians will be diagnosed with cervical cancer in 2022. An estimated 380 will die from it.[150]
India
In India, the number of people with cervical cancer is rising, but overall, the age-adjusted rates are decreasing.[151][152] Usage of condoms in the female population has improved the survival of women with cancers of the cervix.[153]
European Union
As of 2022, the World Health Organization announced that "each year in the WHO European Region, more than 66,000 women are newly diagnosed with cervical cancer and more than 30,000 die from this preventable disease."[154]
United Kingdom
| Parts of this medicine (those related to section) need to be updated. Please update this medicine to reflect recent events or newly available information. (September 2021) |
Cervical cancer is the 12th-most common cancer in women in the UK (around 3,100 women were diagnosed with the disease in 2011) and accounts for 1% of cancer deaths (around 920 died in 2012).[155] With a 42% reduction from 1988 to 1997, the NHS-implemented screening programme has been highly successful, screening the highest-risk age group (25–49 years) every 3 years, and those ages 50–64 every 5 years.
United States
| Parts of this medicine (those related to section) need to be updated. Please update this medicine to reflect recent events or newly available information. (September 2021) |
An estimated 13,170 new cervical cancers and 4,250 cervical cancer deaths will occur in the United States in 2019.[156] The median age at diagnosis is 50. The rate of new cases in the United States was 7.3 per 100,000 women, based on rates from 2012 to 2016. Cervical cancer deaths decreased by approximately 74% in the last 50 years, largely due to widespread Pap test screening.[157] The annual direct medical cost of cervical cancer prevention and treatment before the introduction of the HPV vaccine was estimated at $6 billion.[157]
Nigeria
The Nigerian Institute of Medical Research (NIMR) reports that 28 Nigerian women lose their lives daily due to this disease. This alarming statistic underscores the pressing need for better awareness, prevention, and treatment efforts nationally. Numerous Nigerian women lack access to these preventive measures. In many regions of the country, screening tests such as Pap tests and HPV tests are not easily accessible or affordable[158][159]
History
- 400 BCE: Hippocrates noted that cervical cancer was incurable.
- 1925: Hinselmann invented the colposcope.
- 1928: Papanicolaou developed the Papanicolaou technique.
- 1941: Papanicolaou and Traut: Pap test screening began.
- 1946: Aylesbury spatula was developed to scrape the cervix, collecting the sample for the Pap test.
- 1951: First successful in-vitro cell line, HeLa, derived from biopsy of cervical cancer of Henrietta Lacks.
- 1976: Harald zur Hausen and Gisam found HPV DNA in cervical cancer and genital warts; Hausen later won the Nobel Prize for his work.[160]
- 1988: Bethesda System for reporting Pap results was developed.
- 2006: First HPV vaccine was approved by the FDA.
- 2015: HPV vaccine shown to protect against infection at multiple body sites.[161]
- 2018: Evidence for single-dose protection with HPV vaccine.[162]
Epidemiologists working in the early 20th century noted that cervical cancer behaved like a sexually transmitted disease. In summary:
- Cervical cancer was noted to be common in female sex workers.
- It was rare in nuns, except for those who had been sexually active before entering the convent (Rigoni in 1841).
- It was more common in the second wives of men whose first wives had died from cervical cancer.
- It was rare in Jewish women.[163]
- In 1935, Syverton and Berry discovered a relationship between RPV (Rabbit Papillomavirus) and skin cancer in rabbits.[164] (HPV is species-specific and therefore cannot be transmitted to rabbits).[165]
These historical observations suggested a sexually transmitted agent caused cervical cancer. Initial research in the 1940s and 1950s attributed cervical cancer to smegma.[166] During the 1960s and 1970s it was suspected that infection with herpes simplex virus (HSV) was the cause of the disease. In summary, HSV was seen as a likely cause because it is known to survive in the female reproductive tract, and to be transmitted sexually in a way compatible with known risk factors, such as promiscuity and low socioeconomic status.[167] Herpes viruses were also implicated in other malignant diseases, including Burkitt's lymphoma, Nasopharyngeal carcinoma, Marek's disease and the Lucké renal adenocarcinoma. HSV was recovered from cervical tumour cells. A description of human papillomavirus (HPV) by electron microscopy was given in 1949, and HPV-DNA was identified in 1963.[168] It was not until the 1980s that HPV was identified in cervical cancer tissue.[169] It has since been demonstrated that HPV is implicated in virtually all cervical cancers.[170] Specific viral subtypes implicated are HPV 16, 18, 31, 45 and others.
In work that was initiated in the mid-1980s, the HPV vaccine was developed, in parallel, by researchers at Georgetown University Medical Center, the University of Rochester, the University of Queensland in Australia, and the U.S. National Cancer Institute.[171] In 2006, the US Food and Drug Administration (FDA) approved the first preventive HPV vaccine, marketed by Merck & Co. under the trade name Gardasil. 17 November is the Cervical Cancer Elimination Day of Action.[35] The date marks the day in 2020 when WHO launched the Global strategy to accelerate the elimination of cervical cancer as a public health problem, with a resolution passed by 194 countries.[35] In November 2020, the World Health Organization (WHO), under backing from the World Health Assembly, set out a strategy to eliminate cervical cancer by 2050. The strategy involves vaccinating 90% of girls by the age of 15, screening 70% of women by the age of 35 and again by the age of 45, and treating 90% of women identified with cervical disease.[172] To eliminate cervical cancer, all countries must reach and maintain an incidence rate of below 4 per 100 000 women.[36][37]
Society and culture
Australia
In Australia, Aboriginal women are more than five times more likely to die from cervical cancer than non-Aboriginal women, suggesting that Aboriginal women are less likely to have regular Pap tests.[173] Several factors may limit indigenous women from engaging in regular cervical screening practices, including sensitivity in discussing the topic in Aboriginal communities, embarrassment, anxiety and fear about the procedure.[174] Difficulty in accessing screening services (for example, transport difficulties) and a lack of female GPs, trained Pap test providers and trained female Aboriginal Health Workers are also issues.[174]
The Australian Cervical Cancer Foundation (ACCF), founded in 2008, promotes 'women's health by eliminating cervical cancer and enabling treatment for women with cervical cancer and related health issues, in Australia and in developing countries.'[175] Ian Frazer, one of the developers of the Gardasil cervical cancer vaccine, is the scientific advisor to ACCF.[176] Janette Howard, the wife of the then-Prime Minister of Australia, John Howard, was diagnosed with cervical cancer in 1996, and first spoke publicly about the disease in 2006.[177]
United States

A 2007 survey of American women found 40% had heard of HPV infection, and less than half of those knew it causes cervical cancer.[178] Over a longitudinal study from 1975 to 2000, it was found that people of lower socioeconomic census brackets had higher rates of late-stage cancer diagnosis and higher morbidity rates. After controlling for stage, there still existed differences in survival rates.[179] Women in the US experience stigma around HPV infection, vaccination, and cervical cancer. This is predominantly driven by fear of social judgment and rejection, self-blame, and shame, with notable negative influences from gender and social norms, as both human papillomavirus infection and cervical cancer were stigmatized due to the perception that they arise from reckless behavior such as having multiple sexual partners or neglecting screening.[180]
LGBTQ populations
Transgender men and gender-diverse people who have a cervix (even if partially intact) or have a prior history of cervical cancer or precancerous conditions, and are age 21 or older, who have ever had sex with anyone, need to get screened for cervical cancer.[181][182] Transmasculine people are just as likely as cisgender women to have cervical cancer, but are less likely to undergo cervical screening, because of dysphoria, gender disaffirmation or disempowerment of the individual by healthcare providers,[183] or being misinformed of HPV and cervical cancer risks[184] as well as many healthcare providers perceiving transmasculine individuals to be at low risk of cervical cancer.[185]
Transgender women who have not had bottom surgery have no risk of cervical cancer, as they do not have a cervix. Trans women who have had bottom surgery to create a vagina (vaginoplasty) and possibly a cervix, are at low risk to develop cancer in the tissues of their neo-vagina or neo-cervix as these tissues are made up of different cells than a cervix in a cisgender woman[186][187] Cervical cancer screening is not necessary in trans women who have undergone vaginoplasty because they do not have a cervix.[188]
Intersex people with a cervix are also able to have cervical cancer.[189]
References
- ↑ "CERVICAL | meaning in the Cambridge English Dictionary". https://dictionary.cambridge.org/dictionary/english/cervical.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 "Cervical Cancer Treatment (PDQ®)". 14 March 2014. http://www.cancer.gov/cancertopics/pdq/treatment/cervical/Patient/page1/AllPages.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 5.12. ISBN 978-92-832-0429-9.
- ↑ 4.0 4.1 4.2 4.3 "Cervical Cancer Treatment (PDQ®)". 14 March 2014. http://www.cancer.gov/cancertopics/pdq/treatment/cervical/HealthProfessional/page1/AllPages.
- ↑ 5.0 5.1 5.2 5.3 5.4 Robbins Basic Pathology (8th ed.). Saunders Elsevier. 2007. pp. 718–721. ISBN 978-1-4160-2973-1.
- ↑ 6.0 6.1 Holland-Frei cancer medicine (8th ed.). New York: McGraw-Hill Medical. 2009. p. 1299. ISBN 978-1-60795-014-1. https://books.google.com/books?id=R0FbhLsWHBEC&pg=PA1299.
- ↑ "The epidemiology of human papillomavirus infection and cervical cancer". Disease Markers 23 (4): 213–227. 2007. doi:10.1155/2007/914823. PMID 17627057.
- ↑ 8.0 8.1 "Cervical Cancer Prevention (PDQ®)". 27 February 2014. http://www.cancer.gov/cancertopics/pdq/prevention/cervical/HealthProfessional/page1/AllPages.
- ↑ 9.0 9.1 9.2 "Human Papillomavirus (HPV) Vaccines". 29 December 2011. http://www.cancer.gov/cancertopics/factsheet/prevention/HPV-vaccine.
- ↑ "Global Cancer Facts & Figures 3rd Edition". 2015. p. 9. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/global-cancer-facts-and-figures/global-cancer-facts-and-figures-3rd-edition.pdf.
- ↑ 11.0 11.1 "Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries". CA: A Cancer Journal for Clinicians 71 (3): 209–249. May 2021. doi:10.3322/caac.21660. PMID 33538338.
- ↑ "Defining Cancer". 17 September 2007. http://www.cancer.gov/cancertopics/cancerlibrary/what-is-cancer.
- ↑ "Postcoital bleeding: a review on etiology, diagnosis, and management". Obstetrics and Gynecology International 2014. 2014. doi:10.1155/2014/192087. PMID 25045355.
- ↑ 14.0 14.1 "Immunizing against HPV". https://www.who.int/activities/immunizing-against-hpv.
- ↑ "HPV and HPV-associated diseases". Infectious Disease Clinics of North America 27 (4): 765–778. December 2013. doi:10.1016/j.idc.2013.09.001. PMID 24275269.
- ↑ 16.0 16.1 16.2 16.3 "Human papillomavirus vaccines (HPV)". https://www.who.int/teams/immunization-vaccines-and-biologicals/diseases/human-papillomavirus-vaccines-(HPV).
- ↑ "Cervical cancer" (in en). https://www.who.int/news-room/fact-sheets/detail/cervical-cancer.
- ↑ "Genomic Risk Factors for Cervical Cancer". Cancers 13 (20): 5137. October 2021. doi:10.3390/cancers13205137. PMID 34680286.
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 19.7 19.8 Tewari, Krishnansu S. (2 January 2025). "Cervical Cancer". New England Journal of Medicine 392 (1): 56–71. doi:10.1056/NEJMra2404457. PMID 39752299.
- ↑ "FDA approves Gardasil 9 for prevention of certain cancers caused by five additional types of HPV". 10 December 2014. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm426485.htm.
- ↑ "Control of HPV Infection and Related Cancer Through Vaccination". Viruses and Human Cancer. Recent Results in Cancer Research. 193. 2014. pp. 149–171. doi:10.1007/978-3-642-38965-8_9. ISBN 978-3-642-38964-1.
- ↑ "Immunization coverage". 15 July 2024. https://www.who.int/news-room/fact-sheets/detail/immunization-coverage.
- ↑ 23.0 23.1 23.2 23.3 23.4 23.5 "Human papillomavirus vaccines: WHO position paper (2022 update)". Weekly Epidemiological Record 97 (50): 645–672. December 2022.
- ↑ 24.0 24.1 World Health Organization (February 2014). "Fact sheet No. 297: Cancer". https://www.who.int/mediacentre/factsheets/fs297/en/index.html.
- ↑ "SEER Stat Fact Sheets: Cervix Uteri Cancer". National Cancer Institute. 10 November 2014. http://seer.cancer.gov/statfacts/html/cervix.html.
- ↑ 26.0 26.1 WHO (5 March 2024). "Cervical cancer". https://www.who.int/news-room/fact-sheets/detail/cervical-cancer.
- ↑ 27.0 27.1 World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 1.1. ISBN 978-92-832-0429-9.
- ↑ 28.0 28.1 "WHO adds an HPV vaccine for single-dose use". 4 October 2024. https://www.who.int/news/item/04-10-2024-who-adds-an-hpv-vaccine-for-single-dose-use.
- ↑ "Cervical cancer prevention and control saves lives in the Republic of Korea". https://www.who.int/news-room/feature-stories/detail/cervical-cancer-prevention-and-control-saves-lives-in-the-republic-of-korea.
- ↑ 30.0 30.1 "WHO adds an HPV vaccine for single-dose use". 4 October 2024. https://www.who.int/news/item/04-10-2024-who-adds-an-hpv-vaccine-for-single-dose-use.
- ↑ "An update on uterine cervix pathologies related to infertility". Fertility and Sterility 113 (4): 683–684. April 2020. doi:10.1016/j.fertnstert.2020.02.107. PMID 32228872.
- ↑ 32.0 32.1 32.2 "Cervical cancer". American Family Physician 61 (5): 1369–1376. March 2000. PMID 10735343. http://www.aafp.org/afp/20000301/1369.html.
- ↑ "Mortality impact of achieving WHO cervical cancer elimination targets: a comparative modelling analysis in 78 low-income and lower-middle-income countries". Lancet 395 (10224): 591–603. February 2020. doi:10.1016/S0140-6736(20)30157-4. PMID 32007142.
- ↑ Carraher's polymer chemistry (9th ed.). Boca Raton: Taylor & Francis. 2014. p. 385. ISBN 978-1-4665-5203-6. https://books.google.com/books?id=YXo0AAAAQBAJ&pg=PA385.
- ↑ 35.0 35.1 35.2 35.3 "Cervical Cancer Elimination Day of Action". https://www.who.int/campaigns/cervical-cancer-elimination-day-of-action.
- ↑ 36.0 36.1 36.2 36.3 "Global partners cheer progress towards eliminating cervical cancer and underline challenges". 17 November 2023. https://www.who.int/news/item/17-11-2023-global-partners-cheer-progress-towards-eliminating-cervical-cancer-and-underline-challenges.
- ↑ 37.0 37.1 "Global strategy to accelerate the elimination of cervical cancer as a public health problem". 17 November 2020. https://www.who.int/publications/i/item/9789240014107.
- ↑ "Cervical Cancer Symptoms, Signs, Causes, Stages & Treatment". https://www.medicinenet.com/cervical_cancer/article.htm#what_is_cervical_cancer.
- ↑ "Cervical Cancer Prevention and Early Detection". Cancer. http://www.cancer.org/cancer/cervicalcancer/moreinformation/cervicalcancerpreventionandearlydetection/cervical-cancer-prevention-and-early-detection-cervical-cancer-signs-and-symptoms.
- ↑ "Advances in diagnosis and treatment of metastatic cervical cancer". Journal of Gynecologic Oncology 27 (4). July 2016. doi:10.3802/jgo.2016.27.e43. PMID 27171673.
- ↑ "Cervical cancer". MedlinePlus Medical Encyclopedia. National Institutes of Health. 9 June 2006. https://www.medlineplus.gov/ency/article/000893.htm. Retrieved 2 December 2007.
- ↑ 42.0 42.1 42.2 42.3 42.4 "Smoking habit, immune suppression, oral contraceptive use, and hormone replacement therapy use and cervical carcinogenesis: a review of the literature". Gynecological Endocrinology 27 (8): 597–604. August 2011. doi:10.3109/09513590.2011.558953. PMID 21438669.
- ↑ Gynaecology by Ten Teachers (18th ed.). Hodder Education. 2006. ISBN 978-0-340-81662-2. https://archive.org/details/gynaecology0000unse.
- ↑ "Cervical Cancer Symptoms, Signs, Causes, Stages & Treatment". https://www.medicinenet.com/cervical_cancer/article.htm#cervical_cancer_facts.
- ↑ "HPV-mediated cervical carcinogenesis: concepts and clinical implications". The Journal of Pathology 208 (2): 152–164. January 2006. doi:10.1002/path.1866. PMID 16362994.
- ↑ Principles of cancer biotherapy (5th ed.). Dordrecht: Springer. 2009. p. 149. ISBN 978-90-481-2289-9. https://books.google.com/books?id=emGC_fRJH_IC&pg=PA149.
- ↑ "What Causes Cancer of the Cervix?". American Cancer Society. 30 November 2006. http://www.cancer.org/docroot/CRI/content/CRI_2_2_2X_What_causes_cancer_of_the_cervix_Can_it_be_prevented_8.asp?sitearea=.
- ↑ "Papanicolaou test screening and prevalence of genital human papillomavirus among women who have sex with women". American Journal of Public Health 91 (6): 947–952. June 2001. doi:10.2105/AJPH.91.6.947. PMID 11392939.
- ↑ 49.0 49.1 "HPV and Cancer - NCI" (in en). 1 March 2019. https://www.cancer.gov/about-cancer/causes-prevention/risk/infectious-agents/hpv-and-cancer.
- ↑ "Epidemiologic classification of human papillomavirus types associated with cervical cancer". The New England Journal of Medicine 348 (6): 518–527. February 2003. doi:10.1056/NEJMoa021641. PMID 12571259. https://revistasojs.ucaldas.edu.co/index.php/hacialapromociondelasalud/article/view/2173.
- ↑ Peng, Qiu; Wang, Lujuan; Zuo, Liang; Gao, Shuichao; Jiang, Xianjie; Han, Yaqian; Lin, Jinguan; Peng, Mingjing et al. (January 2024). "HPV E6/E7: insights into their regulatory role and mechanism in signaling pathways in HPV-associated tumor". Cancer Gene Therapy 31 (1): 9–17. doi:10.1038/s41417-023-00682-3. PMID 38102462.
- ↑ 52.0 52.1 52.2 52.3 52.4 52.5 "Cervical Cancer Prevention". PDQ. Bethesda, MD: National Cancer Institute, National Institutes of Health. 26 December 2022. http://cancer.gov/cancertopics/pdq/prevention/cervical/HealthProfessional.
- ↑ 53.0 53.1 53.2 "The role of co-factors in the progression from human papillomavirus infection to cervical cancer". Gynecologic Oncology 128 (2): 265–270. February 2013. doi:10.1016/j.ygyno.2012.11.003. PMID 23146688.
- ↑ "Risk factors for cervical human papillomavirus infection and high-grade intraepithelial lesion in women aged 20 to 31 years in Germany". International Journal of Gynecological Cancer 23 (3): 519–526. March 2013. doi:10.1097/IGC.0b013e318285a4b2. PMID 23360813.
- ↑ 55.0 55.1 "Detection and typing of human papillomavirus DNA in uterine cervices with coexistent grade I and grade III intraepithelial neoplasia: biologic progression or independent lesions?". European Journal of Obstetrics, Gynecology, and Reproductive Biology 121 (1): 99–103. July 2005. doi:10.1016/j.ejogrb.2004.11.024. PMID 15949888.
- ↑ 56.0 56.1 "Risk for cervical intraepithelial neoplasia grade 3 or worse in relation to smoking among women with persistent human papillomavirus infection". Cancer Epidemiology, Biomarkers & Prevention 21 (11): 1949–1955. November 2012. doi:10.1158/1055-9965.EPI-12-0663. PMID 23019238.
- ↑ "Oral contraceptives use and risk of cervical cancer-A systematic review & meta-analysis". European Journal of Obstetrics, Gynecology, and Reproductive Biology 247: 163–175. April 2020. doi:10.1016/j.ejogrb.2020.02.014. PMID 32114321.
- ↑ Cecil Medicine: Expert Consult Premium Edition. ISBN 1-4377-3608-4, 9781437736083. Page 1317.
- ↑ Berek and Hacker's Gynecologic Oncology. ISBN 0-7817-9512-5, 9780781795128. Page 342
- ↑ "Screening for cervical cancer in developing countries". International Journal of Gynaecology and Obstetrics 84 (2): 101–108. February 2004. doi:10.1016/j.ijgo.2003.09.009. PMID 14871510.
- ↑ "Chapter 4: An introduction to colposcopy: indications for colposcopy, instrumentation, principles and documentation of results". Colposcopy and treatment of cervical intraepithelial neoplasia: a beginners' manual. International Agency for Research on Cancer. 2003. ISBN 978-92-832-0412-1. http://screening.iarc.fr/colpochap.php?lang=1&chap=4.
- ↑ 62.0 62.1 "Comparative effectiveness and risk of preterm birth of local treatments for cervical intraepithelial neoplasia and stage IA1 cervical cancer: a systematic review and network meta-analysis". The Lancet. Oncology 23 (8): 1097–1108. August 2022. doi:10.1016/S1470-2045(22)00334-5. PMID 35835138.
- ↑ 63.0 63.1 "Prevention of cervical cancer: what are the risks and benefits of different treatments?". NIHR Evidence (National Institute for Health and Care Research). 10 November 2023. doi:10.3310/nihrevidence_60599. https://evidence.nihr.ac.uk/alert/prevention-of-cervical-cancer-what-are-the-risks-and-benefits-of-different-treatments/.
- ↑ "CT evaluation of cervical cancer: spectrum of disease". Radiographics 21 (5): 1155–1168. September–October 2001. doi:10.1148/radiographics.21.5.g01se311155. PMID 11553823.
- ↑ "Interventions for reducing anxiety in women undergoing colposcopy". The Cochrane Database of Systematic Reviews 2022 (12). December 2011. doi:10.1002/14651858.cd006013.pub3. PMID 22161395.
- ↑ "Cervical Intraepithelial Neoplasia". StatPearls. Treasure Island (FL): StatPearls Publishing. 2023. https://www.ncbi.nlm.nih.gov/books/NBK544371/. Retrieved 16 November 2023.
- ↑ 67.0 67.1 "Cervical Dysplasia: Causes, Symptoms, Diagnosis & Treatment" (in en). https://my.clevelandclinic.org/health/diseases/15678-cervical-intraepithelial-neoplasia-cin.
- ↑ 68.0 68.1 "Cancer Research UK website". http://info.cancerresearchuk.org/cancerstats/types/cervix/incidence/.
- ↑ 69.0 69.1 69.2 Practical principles of cytopathology. Revised edition.. Chicago, IL: American Society for Clinical Pathology Press. 2007. ISBN 978-0-89189-549-7.
- ↑ "Intraepithelial neoplasia of the lower genital tract (cervix, vagina, vulva)" (in en). Comprehensive Gynecology. Elsevier. 2022. pp. 637–647.e2. doi:10.1016/b978-0-323-65399-2.00038-3. ISBN 978-0-323-65399-2.
- ↑ "Cervical Cancer". eMedicine. WebMD. 6 July 2006. http://www.emedicine.com/med/topic324.htm#section~workup.
- ↑ "Cervical Cancer: The Basics". OncoLink (Abramson Cancer Center of the University of Pennsylvania). 17 July 2006. http://www.oncolink.upenn.edu/types/article.cfm?c=6&s=17&ss=129&id=8226.
- ↑ "What Is Cervical Cancer?". American Cancer Society. https://www.cancer.org/cancer/cervical-cancer/about/what-is-cervical-cancer.html.
- ↑ "Cervical cancer – Types and grades". Cancer Research UK. http://www.cancerresearchuk.org/about-cancer/cervical-cancer/stages-types-grades/types-and-grades.
- ↑ "Squamous cell carcinoma and variants". https://www.pathologyoutlines.com/topic/cervixscc.html.
- ↑ Image by Mikael Häggström, MD. Source for caption: "Cervix - Squamous cell carcinoma and variants". https://www.pathologyoutlines.com/topic/cervixscc.html. Last author update: 24 September 2020
- ↑ Image by Mikael Häggström, MD. Source for caption: "Assessment of p16 and Ki67 Immunohistochemistry Expression in Squamous Intraepithelial Lesion with Cytohistomorphological Correlation.". Iran J Pathol 15 (4): 268–273. 2020. doi:10.30699/ijp.2020.112421.2208. PMID 32944038.
- ↑ "A rare case of gastric-type mucinous endocervical adenocarcinoma in a 59-year-old woman". Przeglad Menopauzalny = Menopause Review 19 (3): 147–150. September 2020. doi:10.5114/pm.2020.99563. PMID 33100952.
- ↑ "Revised FIGO staging for carcinoma of the cervix uteri". International Journal of Gynaecology and Obstetrics 145 (1): 129–135. April 2019. doi:10.1002/ijgo.12749. PMID 30656645.
- ↑ "Liquid-based cytology in cervical screening: a rapid and systematic review". Health Technology Assessment 4 (18): 1–73. 2000. doi:10.3310/hta4180. PMID 10932023.
- ↑ "Liquid-based cytology in cervical screening: an updated rapid and systematic review and economic analysis". Health Technology Assessment 8 (20): iii, 1-iii, 78. May 2004. doi:10.3310/hta8200. PMID 15147611.
- ↑ "Liquid Based Cytology (LBC): NHS Cervical Screening Programme". http://www.cancerscreening.nhs.uk/cervical/lbc.html.
- ↑ 83.0 83.1 "European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition--summary document". Annals of Oncology 21 (3): 448–458. March 2010. doi:10.1093/annonc/mdp471. PMID 20176693.
- ↑ "Interventions targeted at women to encourage the uptake of cervical screening". The Cochrane Database of Systematic Reviews 2021 (9). September 2021. doi:10.1002/14651858.CD002834.pub3. PMID 34694000.
- ↑ 85.0 85.1 85.2 "Cervical Cancer Screening Guidelines for Average-Risk Women". https://www.cdc.gov/cancer/cervical/pdf/guidelines.pdf.
- ↑ 86.0 86.1 86.2 Committee on Practice Bulletins—Gynecology (November 2012). "ACOG Practice Bulletin Number 131: Screening for cervical cancer". Obstetrics and Gynecology 120 (5): 1222–1238. doi:10.1097/AOG.0b013e318277c92a. PMID 23090560.
- ↑ "New cervical cancer screening guidelines, again". Obstetrics and Gynecology Clinics of North America 40 (2): 211–223. June 2013. doi:10.1016/j.ogc.2013.03.001. PMID 23732026.
- ↑ 88.0 88.1 "Screening for Cervical Cancer: US Preventive Services Task Force Recommendation Statement". JAMA 320 (7): 674–686. August 2018. doi:10.1001/jama.2018.10897. PMID 30140884.
- ↑ 89.0 89.1 "Primary Prevention of Cervical Cancer: American Society of Clinical Oncology Resource-Stratified Guideline". Journal of Global Oncology 3 (5): 611–634. October 2017. doi:10.1200/JGO.2016.008151. PMID 29094100.
- ↑ 90.0 90.1 90.2 Comprehensive cervical cancer control. A guide to essential practice (Second ed.). World Health Organization. 2014. ISBN 978-92-4-154895-3. https://www.who.int/reproductivehealth/publications/cancers/cervical-cancer-guide/en/.
- ↑ "Efficacy of human papillomavirus vaccines: a systematic quantitative review". International Journal of Gynecological Cancer 19 (7): 1166–1176. October 2009. doi:10.1111/IGC.0b013e3181a3d100. PMID 19823051.
- ↑ Kitano, Taito (24 March 2022). "Risk-Benefit Analysis of the 9-Valent HPV Vaccination for Adolescent Boys from an Individual Perspective". Japanese Journal of Infectious Diseases 75 (2): 114–120. doi:10.7883/yoken.JJID.2021.367. PMID 34334535.
- ↑ "Do condoms prevent genital HPV infection, external genital warts, or cervical neoplasia? A meta-analysis". Sexually Transmitted Diseases 29 (11): 725–735. November 2002. doi:10.1097/00007435-200211000-00018. PMID 12438912.
- ↑ "Vitamin A and risk of cervical cancer: a meta-analysis". Gynecologic Oncology 124 (2): 366–373. February 2012. doi:10.1016/j.ygyno.2011.10.012. PMID 22005522.
- ↑ "Vitamin or antioxidant intake (or serum level) and risk of cervical neoplasm: a meta-analysis". BJOG 118 (11): 1285–1291. October 2011. doi:10.1111/j.1471-0528.2011.03032.x. PMID 21749626.
- ↑ "Cervical Cancer Treatment Options | Treatment Choices by Stage". https://www.cancer.org/cancer/cervical-cancer/treating/by-stage.html.
- ↑ "Primary surgery versus primary radiotherapy with or without chemotherapy for early adenocarcinoma of the uterine cervix". The Cochrane Database of Systematic Reviews 2021 (1). January 2013. doi:10.1002/14651858.CD006248.pub3. PMID 23440805.
- ↑ "A systematic overview of radiation therapy effects in cervical cancer (cervix uteri)". Acta Oncologica 42 (5–6): 546–556. 2003. doi:10.1080/02841860310014660. PMID 14596512.
- ↑ Chemoradiotherapy for Cervical Cancer Meta-analysis Collaboration (CCCMAC) (January 2010). "Reducing uncertainties about the effects of chemoradiotherapy for cervical cancer: individual patient data meta-analysis". The Cochrane Database of Systematic Reviews 2010 (1). doi:10.1002/14651858.cd008285. PMID 20091664.
- ↑ 100.0 100.1 "Curative pelvic exenteration for recurrent cervical carcinoma in the era of concurrent chemotherapy and radiation therapy. A systematic review". European Journal of Surgical Oncology 41 (8): 975–985. August 2015. doi:10.1016/j.ejso.2015.03.235. PMID 25922209. https://hal-univ-rennes1.archives-ouvertes.fr/hal-01147372/file/Curative%20Pelvic%20Exenteration%20For%20Recurrent%20Cervical.pdf.
- ↑ "Perioperative enhanced recovery programmes for women with gynaecological cancers". The Cochrane Database of Systematic Reviews 2022 (3). March 2022. doi:10.1002/14651858.CD008239.pub5. PMID 35289396.
- ↑ "Microinvasive carcinoma of the cervix". American Journal of Obstetrics and Gynecology 145 (8): 981–991. April 1983. doi:10.1016/0002-9378(83)90852-9. PMID 6837683.
- ↑ "Cone biopsy (conization) for abnormal cervical cell changes". 12 January 2007. http://www.webmd.com/cancer/cervical-cancer/cone-biopsy-conization-for-abnormal-cervical-cell-changes.
- ↑ "Vaginectomy and vaginoplasty for isolated vaginal recurrence 8 years after cervical cancer radical hysterectomy: A case report and literature review". The Journal of Obstetrics and Gynaecology Research 43 (9): 1493–1497. September 2017. doi:10.1111/jog.13375. PMID 28691384.
- ↑ "Surgical treatment of stage IA2 cervical cancer". The Cochrane Database of Systematic Reviews 2018 (5). May 2014. doi:10.1002/14651858.cd010870.pub2. PMID 24874726.
- ↑ "Early invasive carcinoma of the cervix". Gynecologic Oncology 51 (1): 26–32. October 1993. doi:10.1006/gyno.1993.1241. PMID 8244170.
- ↑ "Trachelectomy". 2001. http://www.baymoon.com/~gyncancer/library/glossary/bldeftrachelect.htm.
- ↑ 108.0 108.1 "Radical trachelectomy with laparoscopic lymphadenectomy: review of oncologic and obstetrical outcomes". Current Opinion in Obstetrics & Gynecology 18 (1): 8–13. February 2006. doi:10.1097/01.gco.0000192968.75190.dc. PMID 16493253.
- ↑ "[Abdominal radical trachelectomy--technique and experience]" (in cs). Ceska Gynekologie 70 (2): 117–122. March 2005. PMID 15918265.
- ↑ "Vaginal radical trachelectomy: a valuable fertility-preserving option in the management of early-stage cervical cancer. A series of 50 pregnancies and review of the literature". Gynecologic Oncology 98 (1): 3–10. July 2005. doi:10.1016/j.ygyno.2005.04.014. PMID 15936061.
- ↑ "Vaginal radical hysterectomy versus abdominal radical hysterectomy in the treatment of early-stage cervical cancer". Gynecologic Oncology 62 (3): 336–339. September 1996. doi:10.1006/gyno.1996.0245. PMID 8812529.
- ↑ "Laparoscopic vaginal radical trachelectomy: a treatment to preserve the fertility of cervical carcinoma patients". Cancer 88 (8): 1877–1882. April 2000. doi:10.1002/(SICI)1097-0142(20000415)88:8<1877::AID-CNCR17>3.0.CO;2-W. PMID 10760765.
- ↑ "Radical trachelectomy and pelvic lymphadenectomy with uterine preservation in the treatment of cervical cancer". American Journal of Obstetrics and Gynecology 188 (1): 29–34. January 2003. doi:10.1067/mob.2003.124. PMID 12548192.
- ↑ 114.0 114.1 "Radiotherapy and chemoradiation after surgery for early cervical cancer". The Cochrane Database of Systematic Reviews 5 (5). May 2012. doi:10.1002/14651858.cd007583.pub3. PMID 22592722.
- ↑ "Cervical cancer". Lancet 361 (9376): 2217–2225. June 2003. doi:10.1016/S0140-6736(03)13778-6. PMID 12842378.
- ↑ "Adjuvant platinum-based chemotherapy for early stage cervical cancer". The Cochrane Database of Systematic Reviews 11 (11). November 2016. doi:10.1002/14651858.CD005342.pub4. PMID 27873308.
- ↑ "Type II or type III radical hysterectomy compared to chemoradiotherapy as a primary intervention for stage IB2 cervical cancer". The Cochrane Database of Systematic Reviews 2018 (10). October 2018. doi:10.1002/14651858.cd011478.pub2. PMID 30311942.
- ↑ "FDA Approves First Drug Treatment for Late-Stage Cervical Cancer". U.S. Food and Drug Administration. 15 June 2006. https://www.fda.gov/bbs/topics/NEWS/2006/NEW01391.html.
- ↑ "The combination of cisplatin and topotecan as a second-line treatment for patients with advanced/recurrent uterine cervix cancer". Medicine 97 (14). April 2018. doi:10.1097/MD.0000000000010340. PMID 29620661.
- ↑ "Adjuvant chemotherapy after concurrent chemoradiation for locally advanced cervical cancer". The Cochrane Database of Systematic Reviews 2019 (12). December 2014. doi:10.1002/14651858.cd010401.pub2. PMID 25470408.
- ↑ "Follow-up protocols for women with cervical cancer after primary treatment". The Cochrane Database of Systematic Reviews 2014 (11). November 2013. doi:10.1002/14651858.CD008767.pub2. PMID 24277645.
- ↑ "Palliative interventions for controlling vaginal bleeding in advanced cervical cancer". The Cochrane Database of Systematic Reviews 3 (3). March 2019. doi:10.1002/14651858.cd011000.pub3. PMID 30888060.
- ↑ "FDA Approves Pembrolizumab for Advanced Cervical Cancer". U.S. Food and Drug Administration. 12 June 2018. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pembrolizumab-advanced-cervical-cancer.
- ↑ "Immunotherapy in Cervical Cancer: Advances and Perspectives". Cancers (Basel) 14 (15): 3803. July 2022. doi:10.3390/cancers14153803. PMID 35955074.
- ↑ "FDA Approves Pembrolizumab for Cervical Cancer with a New Dosing Regimen". U.S. Food and Drug Administration. 13 October 2021. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pembrolizumab-cervical-cancer-new-dosing-regimen.
- ↑ "FDA Approves Cemiplimab-rwlc for Recurrent or Metastatic Cervical Cancer with Disease Progression on or after Chemotherapy". U.S. Food and Drug Administration. 29 September 2022. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-cemiplimab-rwlc-recurrent-or-metastatic-cervical-cancer-disease-progression-or-after-chemotherapy.
- ↑ "Enforcement Reports". https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761208s000lbl.pdf.
- ↑ "Seagen and Genmab Announce FDA Accelerated Approval for Tivdak (tisotumab vedotin-tftv) in Previously Treated Recurrent or Metastatic Cervical Cancer". Seagen. 20 September 2021. https://www.businesswire.com/news/home/20210920005921/en/.
- ↑ "Cervical Intraepithelial Neoplasia.". StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. January 2022. https://www.ncbi.nlm.nih.gov/books/NBK544371/.
- ↑ "Survival Rates for Cervical Cancer". American Cancer Society. 3 January 2020. https://www.cancer.org/cancer/cervical-cancer/detection-diagnosis-staging/survival.html.
- ↑ "Cervical Cancer". Cervical Cancer: Pathology, Symptoms and Signs, Diagnosis, Prognosis and Treatment. Armenian Health Network, Health.am. http://www.health.am/cr/cervical-cancer/.
- ↑ Cohen, Camryn M.; Wentzensen, Nicolas; Castle, Philip E.; Schiffman, Mark; Zuna, Rosemary; Arend, Rebecca C.; Clarke, Megan A. (1 December 2022). "Racial and Ethnic Disparities in Cervical Cancer Incidence, Survival, and Mortality by Histologic Subtype". Journal of Clinical Oncology (ASCO Publications) 41 (5): 1059–1068. doi:10.1200/JCO.22.01424. PMID 36455190.
- ↑ "Cervical cancer statistics and prognosis". Cancer Research UK. http://www.cancerhelp.org.uk/help/default.asp?page=9260.
- ↑ 134.0 134.1 "Screening for cervical cancer: when theory meets reality". BMC Cancer 11. June 2011. doi:10.1186/1471-2407-11-240. PMID 21668947.
- ↑ "Quality of Life among Female Cancer Survivors in Africa: An Integrative Literature Review". Asia-Pacific Journal of Oncology Nursing 4 (1): 6–17. 2016. doi:10.4103/2347-5625.199078. PMID 28217724.
- ↑ "African cervical cancer prevention and control plans: A scoping review". Journal of Cancer Policy 16: 73–81. 2018. doi:10.1016/j.jcpo.2018.05.002.
- ↑ "Chemotherapy for Cervical Cancer". American Cancer Society. 28 June 2024. https://www.cancer.org/cancer/types/cervical-cancer/treating/chemotherapy.html.
- ↑ 138.0 138.1 Majeed, Hafsa; Gupta, Vikas (14 August 2023). Adverse Effects of Radiation Therapy. StatPearls Publishing LLC.. NBK563259. https://www.ncbi.nlm.nih.gov/books/NBK563259/. Retrieved 24 October 2024.
- ↑ Schmitt, Luiza G.; Amarnath, Sudha R. (1 September 2023). "Late Effects of Pelvic Radiation Therapy in the Female Patient: A Comprehensive Review". Applied Radiation Oncology 2023 (3): 13–24. doi:10.37549/ARO-D-23-00016. https://www.appliedradiationoncology.com/articles/late-effects-of-pelvic-radiation-therapy-in-the-female-patient-a-comprehensive-review. Retrieved 16 October 2024.
- ↑ Huh, Jung Wook; Tanksley, Jarred; Chino, Junzo; Willett, Christopher G.; Dewhirst, Mark W. (1 July 2020). "Long-term Consequences of Pelvic Irradiation: Toxicities, Challenges, and Therapeutic Opportunities with Pharmacologic Mitigators". Clinical Cancer Research 26 (13): 3079–3090. doi:10.1158/1078-0432.CCR-19-2744. ISSN 1078-0432. PMID 32098770. https://aacrjournals.org/clincancerres/article/26/13/3079/82622/Long-term-Consequences-of-Pelvic-Irradiation. Retrieved 24 October 2024.
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2009. https://www.who.int/healthinfo/global_burden_disease/estimates_country/en/index.html.
- ↑ "HPV Vaccination and Testing". Reviews in Obstetrics & Gynecology 3 (1): 33–34. Winter 2010. PMID 20508781.
- ↑ "Gynecologic Malignancies in Pregnancy: Balancing Fetal Risks With Oncologic Safety". Obstetrical & Gynecological Survey 72 (3): 184–193. March 2017. doi:10.1097/OGX.0000000000000407. PMID 28304416.
- ↑ "Cervical cancer in Australia statistics". 18 August 2022. https://www.canceraustralia.gov.au/cancer-types/cervical-cancer/statistics.
- ↑ "National Cervical Screening Program monitoring report 2022, Summary" (in en-AU). December 2022. https://www.aihw.gov.au/reports/cancer-screening/ncsp-monitoring-2022/summary.
- ↑ "Incidence and mortality rates". January 1900. http://www.papscreen.org.au/browse.asp?ContainerID=c15.
- ↑ "Papscreen Victoria". http://www.papscreen.org.au/.
- ↑ "The projected timeframe until cervical cancer elimination in Australia: a modelling study". The Lancet. Public Health 4 (1): e19–e27. January 2019. doi:10.1016/S2468-2667(18)30183-X. PMID 30291040.
- ↑ G C, Binod; G C, Alisha (2025). "Other countries should copy Nepal's approach to tackling cervical cancer". Nature 638 (8052): 891. doi:10.1038/d41586-025-00589-z. ISSN 1476-4687. PMID 40000865. Bibcode: 2025Natur.638..891G.
- ↑ "Cervical Cancer statistics". May 2022. https://cancer.ca/en/cancer-information/cancer-types/cervical/statistics.
- ↑ National Cancer Registry Programme under Indian Council of Medical Research Reports
- ↑ "Plunes HealthCare - Surgery Experience Made Easy | Upto 50% Off". https://www.plunes.com/.
- ↑ "Association of educational levels with survival in Indian patients with cancer of the uterine cervix". Asian Pacific Journal of Cancer Prevention 16 (8): 3121–3123. 2015. doi:10.7314/apjcp.2015.16.8.3121. PMID 25921107.
- ↑ "The cancer we can eliminate – WHO/Europe urges Member States to consign cervical cancer to history" (in en). https://www.who.int/europe/news/item/12-09-2022-the-cancer-we-can-eliminate---who-europe-urges-member-states-to-consign-cervical-cancer-to-history.
- ↑ "Cervical cancer statistics". http://www.cancerresearchuk.org/cancer-info/cancerstats/types/cervix/.
- ↑ "Cancer Stat Facts: Cervical Cancer". https://seer.cancer.gov/statfacts/html/cervix.html.
- ↑ 157.0 157.1 "Prophylaxis of cervical cancer and related cervical disease: a review of the cost-effectiveness of vaccination against oncogenic HPV types". Journal of Managed Care Pharmacy 16 (3): 217–230. April 2010. doi:10.18553/jmcp.2010.16.3.217. PMID 20331326.
- ↑ "Eliminating Cervical Cancer in Nigeria: How Awareness, Infrastructure, and Government Support Can Make a Difference - My Daily News Usa" (in en-US). 4 April 2023. https://mydailynewsusa.com/eliminating-cervical-cancer-in-nigeria-how-awareness-infrastructure-and-government-support-can-make-a-difference/.
- ↑ "Eliminating Cervical Cancer in Nigeria: How Awareness, Infrastructure, and Government Support Can Make a Difference". 4 April 2023. https://mydailynewsusa.com/eliminating-cervical-cancer-in-nigeria-how-awareness-infrastructure-and-government-support-can-make-a-difference/.
- ↑ "Papillomaviruses and cancer: from basic studies to clinical application". Nature Reviews. Cancer 2 (5): 342–350. May 2002. doi:10.1038/nrc798. PMID 12044010.
- ↑ "Multisite HPV16/18 Vaccine Efficacy Against Cervical, Anal, and Oral HPV Infection". Journal of the National Cancer Institute 108 (1). January 2016. doi:10.1093/jnci/djv302. PMID 26467666.
- ↑ "Evidence for single-dose protection by the bivalent HPV vaccine-Review of the Costa Rica HPV vaccine trial and future research studies". Vaccine 36 (32 Pt A): 4774–4782. August 2018. doi:10.1016/j.vaccine.2017.12.078. PMID 29366703.
- ↑ "The low incidence of cervical cancer in Jewish women: has the puzzle finally been solved?". The Israel Medical Association Journal 5 (2): 120–123. February 2003. PMID 12674663. http://www.ima.org.il/IMAJ/ViewArticle.aspx?year=2003&month=02&page=120. Retrieved 28 November 2015.
- ↑ Syverton, J. T.; Berry, G. P. (1935). "Carcinoma in the Cottontail Rabbit Following Spontaneous Virus Papilloma (Shope)". Experimental Biology and Medicine 33 (3): 399–400. doi:10.3181/00379727-33-8386P.
- ↑ IARC Working Group on the Evaluation of Carcinogenic Risks to Humans (2007). "Studies of Animal Papillomaviruses". Human Papillomaviruses. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 90. Lyon, France: International Agency for Research on Cancer. https://www.ncbi.nlm.nih.gov/books/NBK321758/.
- ↑ "The possible role of smegma in carcinoma of the cervix". American Journal of Obstetrics and Gynecology 76 (4): 726–33; discussion 733–5. October 1958. doi:10.1016/0002-9378(58)90004-8. PMID 13583012.
- ↑ "Possible etiologies of cancer of the cervix other than herpesvirus". Cancer Research 33 (6): 1485–1490. June 1973. PMID 4352386.
- ↑ Human Papillomavirus: A Practical Guide for Urologists. Springer Science & Business Media. 6 August 2009. ISBN 978-3-540-70974-9. https://books.google.com/books?id=IS84In6aguYC&pg=PA6.
- ↑ "A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions". Proceedings of the National Academy of Sciences of the United States of America 80 (12): 3812–3815. June 1983. doi:10.1073/pnas.80.12.3812. PMID 6304740. Bibcode: 1983PNAS...80.3812D..
- ↑ "Prophylactic human papillomavirus vaccines". The Journal of Clinical Investigation 116 (5): 1167–1173. May 2006. doi:10.1172/JCI28607. PMID 16670757.
- ↑ "Who invented the VLP cervical cancer vaccines?". Journal of the National Cancer Institute 98 (7): 433. April 2006. doi:10.1093/jnci/djj144. PMID 16595773.
- ↑ "WHO rolls out plan to rid world of cervical cancer, saving millions of lives". 16 November 2020. https://news.un.org/en/story/2020/11/1077752.
- ↑ Cancer Institute NSW (2013). "Information about cervical screening for Aboriginal women". NSW Government. http://www.csp.nsw.gov.au/aboriginal.
- ↑ 174.0 174.1 "Aboriginal cervical cancer rates parallel health inequity". Science Network Western Australia. 17 October 2011. http://www.sciencewa.net.au/topics/health-a-medicine/item/995-aboriginal-cervical-cancer-rates-parallel-health-inequity.html.
- ↑ Australian Cervical Cancer Foundation. "Vision and Mission". Australian Cervical Cancer Foundation. http://accf.org.au/about/vision.html.
- ↑ Australian Cervical Cancer Foundation. "Our People". Australian Cervical Cancer Foundation. http://accf.org.au/about/ourboard.html.
- ↑ "Janette Howard speaks on her battle with cervical cancer". Australian Broadcasting Corporation. 16 October 2006. http://www.abc.net.au/pm/content/2006/s1766104.htm.
- ↑ "What do women in the U.S. know about human papillomavirus and cervical cancer?". Cancer Epidemiology, Biomarkers & Prevention 16 (2): 288–294. February 2007. doi:10.1158/1055-9965.EPI-06-0756. PMID 17267388.
- ↑ "Persistent area socioeconomic disparities in U.S. incidence of cervical cancer, mortality, stage, and survival, 1975–2000". Cancer 101 (5): 1051–1057. September 2004. doi:10.1002/cncr.20467. PMID 15329915.
- ↑ "Stigma and cervical cancer prevention: A scoping review of the U.S. literature". Prev Med 153. December 2021. doi:10.1016/j.ypmed.2021.106849. PMID 34662598. https://figshare.com/articles/journal_contribution/16926994.
- ↑ "As a trans man, do I need to get screened for cervical cancer?". https://cancer.ca/en/cancer-information/find-cancer-early/screening-in-lgbtq-communities/as-a-trans-man-do-i-need-to-get-screened-for-cervical-cancer.
- ↑ "Should trans men have cervical screening tests?" (in en). 27 June 2018. https://www.nhs.uk/common-health-questions/sexual-health/should-trans-men-have-cervical-screening-tests/.
- ↑ "Enacting power and constructing gender in cervical cancer screening encounters between transmasculine patients and health care providers". Culture, Health & Sexuality 22 (12): 1315–1332. December 2020. doi:10.1080/13691058.2019.1677942. PMID 31661659.
- ↑ "Cervical Cancer Screening for Patients on the Female-to-Male Spectrum: a Narrative Review and Guide for Clinicians". Journal of General Internal Medicine 30 (12): 1857–1864. December 2015. doi:10.1007/s11606-015-3462-8. PMID 26160483.
- ↑ "Perceptions of cervical cancer risk and screening among transmasculine individuals: patient and provider perspectives". Culture, Health & Sexuality 18 (10): 1192–1206. October 2016. doi:10.1080/13691058.2016.1177203. PMID 27142466.
- ↑ "I'm trans or non-binary, does this affect my cancer screening?" (in en). 10 October 2019. https://www.cancerresearchuk.org/about-cancer/cancer-symptoms/spot-cancer-early/screening/trans-and-non-binary-cancer-screening.
- ↑ "As a trans woman, do I need to get screened for cervical cancer?". https://cancer.ca/en/cancer-information/find-cancer-early/screening-in-lgbtq-communities/as-a-trans-woman-do-i-need-to-get-screened-for-cervical-cancer.
- ↑ "Transgender patients: considerations for routine gynecologic care and cancer screening". International Journal of Gynecological Cancer 30 (12): 1990–1996. December 2020. doi:10.1136/ijgc-2020-001860. PMID 33109526.
- ↑ "Causes of cervical cancer" (in en). 20 October 2017. https://www.nhs.uk/conditions/cervical-cancer/causes/.
Further reading
- "Worldwide burden of cervical cancer in 2008". Annals of Oncology 22 (12): 2675–2686. December 2011. doi:10.1093/annonc/mdr015. PMID 21471563.
- "Cancer of the cervix uteri". International Journal of Gynaecology and Obstetrics 143 (Suppl 2): 22–36. October 2018. doi:10.1002/ijgo.12611. PMID 30306584.
- "Management and Care of Women With Invasive Cervical Cancer: American Society of Clinical Oncology Resource-Stratified Clinical Practice Guideline". Journal of Global Oncology 2 (5): 311–340. October 2016. doi:10.1200/JGO.2016.003954. PMID 28717717.
- "The cervical cancer epidemic that screening has prevented in the UK". Lancet 364 (9430): 249–256. 2004. doi:10.1016/S0140-6736(04)16674-9. PMID 15262102.
- "Estimate of the global burden of cervical adenocarcinoma and potential impact of prophylactic human papillomavirus vaccination". BMC Cancer 13 (1). November 2013. doi:10.1186/1471-2407-13-553. PMID 24261839.
External links
- Fact sheet on cervical cancer by the WHO
- Cervical cancer at the NIH National Cancer Institute
- Jo's Cervical Cancer Trust, UK cervical cancer charity
| Classification | |
|---|---|
| External resources |
Template:Female genital neoplasia
 |
![Invasive squamous cell carcinoma of the cervix is characterized by infiltration as irregular anastomosing nests or single cells.[75] This case is poorly differentiated. H&E stain.](/wiki/images/thumb/4/43/Histopathology_of_squamous_cell_carcinoma_of_the_cervix.png/320px-Histopathology_of_squamous_cell_carcinoma_of_the_cervix.png)
![Cervical squamous cell carcinoma generally shows diffuse staining of both nuclei and cytoplasm on p16 immuno- histochemistry (except verrucous variant).[76]](/wiki/images/thumb/4/48/Immunohistochemistry_for_p16_in_cervical_squamous_cell_carcinoma.jpg/179px-Immunohistochemistry_for_p16_in_cervical_squamous_cell_carcinoma.jpg)
![Invasive cervical squamous cell carcinoma on H&E histopathology and Ki-67 immunohistochemistry. The latter correlates well with the degree and level of dysplasia.[77]](/wiki/images/thumb/4/4c/Invasive_cervical_squamous_cell_carcinoma_on_H%26E_histopathology_and_Ki-67_immunohistochemistry.jpg/361px-Invasive_cervical_squamous_cell_carcinoma_on_H%26E_histopathology_and_Ki-67_immunohistochemistry.jpg)