Chemistry:Triphenylene
| |
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Triphenylene[1] | |
| Other names
Benzo[l]phenanthrene
9,10-Benzophenanthrene 1,2,3,4-Dibenzonaphthalene Isochrysene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
| KEGG | |
| MeSH | C009590 |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C18H12 | |
| Molar mass | 228.294 g·mol−1 |
| Appearance | white |
| Density | 1.308 g/cm3[2] |
| Melting point | 198 °C; 388 °F; 471 K |
| Boiling point | 438 °C; 820 °F; 711 K |
| -156.6·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
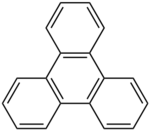
Triphenylene is an organic compound with the formula (C6H4)3. A flat polycyclic aromatic hydrocarbon (PAH), it consists of four fused benzene rings. Triphenylene has delocalized 18-π-electron systems based on a planar structure, corresponding to the symmetry group D3h. It is a white or colorless solid.
Preparation
Triphenylene can be isolated from coal tar. It can also be synthesized in various ways. One method is trimerization of benzyne.[3] Another method involves trapping benzyne with a biphenyl derivative.[4]
Properties
Triphenylene is more resonance stable than its isomers chrysene, benz[a]anthracene, benzo[c]phenanthrene, and tetracene. For this reason triphenylene resists hydrogenation.[5]
As a disc-shaped, planar molecule, triphenylene has attracted attention as the core of discotic mesogen in liquid crystalline materials.[6]
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 209. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ Ahmed, F. R.; Trotter, J. (1963). "The crystal structure of triphenylene". Acta Crystallographica 16 (6): 503–508. doi:10.1107/S0365110X63001365.
- ↑ Heaney, H.; Millar, I. T. (1960). "Triphenylene". Organic Syntheses 40: 105. doi:10.15227/orgsyn.040.0105.
- ↑ Katie A. Spence, Milauni M. Mehta, Neil K. Garg (2022). "Synthesis of Triphenylene via the Palladium–Catalyzed Annulation of Benzyne". Organic Syntheses 99: 174–189. doi:10.15227/orgsyn.099.0174.
- ↑ Kofman, V.; Sarre, P.J.; Hibbins, R.E.; ten Kate, I.L.; Linnartz, H. (2017). "Laboratory spectroscopy and astronomical significance of the fully-benzenoid PAH triphenylene and its cation" (in en). Molecular Astrophysics 7: 19–26. doi:10.1016/j.molap.2017.04.002. Bibcode: 2017MolAs...7...19K. https://linkinghub.elsevier.com/retrieve/pii/S2405675817300039.
- ↑ Janietz, Dietmar (2001), "Liquid Crystals at Interfaces" (in en), Handbook of Surfaces and Interfaces of Materials (Elsevier): pp. 436–437, doi:10.1016/b978-012513910-6/50014-1, ISBN 978-0-12-513910-6, https://linkinghub.elsevier.com/retrieve/pii/B9780125139106500141, retrieved 2020-08-23
External links
 |

