Chemistry:Anidulafungin
 | |
| Clinical data | |
|---|---|
| Pronunciation | /eɪˌnɪdjʊləˈfʌndʒɪn/ ay-NID-yuu-lə-FUN-jin |
| Trade names | Eraxis, Ecalta |
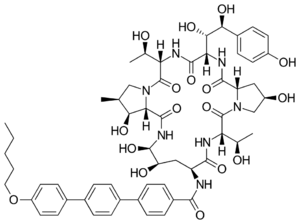
| Other names | (4R,5S)-4,5-Dihydroxy-N2-[[4''-(pentyloxy)-p-terphenyl-4-yl]carbonyl]-L-ornithyl-L-threonyl-trans-4-hydroxy-L-prolyl-(S)-4-hydroxy-4-(p-hydroxyphenyl)-L-threonyl-L-threonyl-(3S,4S)-3-hydroxy-4-methyl-L-proline cyclic (6→1)-peptide[1] 1-[(4R,5R)-4,5-Dihydroxy-N2-[[4''-(pentyloxy)[1',1':4',1''-terphenyl]-4-yl]carbonyl]-L-ornithine]echinocandin B[2] |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 100% (intravenous use only) |
| Protein binding | Extensive (>99%) |
| Metabolism | Hepatic metabolism not observed, CYP system not involved |
| Elimination half-life | 27 hours; 40–50 hours (terminal) |
| Excretion | Feces (~30%), urine (<1%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C58H73N7O17 |
| Molar mass | 1140.254 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Anidulafungin (INN)[1]: 42 (trade names Eraxis, Ecalta) is a semisynthetic echinocandin used as an antifungal drug. It was previously known as LY303366.[3][4][5] It may also have application in treating invasive Aspergillus infection when used in combination with voriconazole.[6] It is a member of the class of antifungal drugs known as the echinocandins; its mechanism of action is by inhibition of (1→3)-β-D-glucan synthase, an enzyme important to the synthesis of the fungal cell wall.[7]
It is on the World Health Organization's List of Essential Medicines.[8]
Indications
- Candidemia and other forms of invasive Candida infections (intra-abdominal abscess and peritonitis)
- Esophageal candidiasis
Anidulafungin has not been studied in endocarditis, osteomyelitis, and meningitis due to Candida, and has not been studied in sufficient numbers of neutropenic patients to determine efficacy in this group.[2]
Pharmacology
Pharmacodynamics
Anidulafungin significantly differs from other antifungals in that it undergoes chemical degradation to inactive forms at body pH and temperature. Because it does not rely on enzymatic degradation or hepatic or renal excretion, the drug is safe to use in patients with any degree of hepatic or renal impairment.[9]
| Parameter | Value |
|---|---|
| Volume of distribution (L) | 30–50 L.[10] |
| Plasma protein binding (%) | 99%[10] |
| Elimination half-life [h] | 24 hours[10] |
Pharmacokinetics
Anidulafungin is not evidently metabolized by the liver. This specific drug undergoes slow chemical hydrolysis to an open-ring peptide which lacks antifungal activity. The half-life of the drug is 27 hours. About 30% is excreted in the feces (10% as unchanged drug). Less than 1% is excreted in the urine.[11][12][13]
Mechanism of action
Anidulafungin inhibits glucan synthase, an enzyme important in the formation of (1→3)-β-D-glucan, a major fungal cell wall component. Glucan synthase is not present in mammalian cells, so it is an attractive target for antifungal activity.[14]
Semisynthesis
Anidulafungin is manufactured via semi-synthesis. The starting material is echinocandin B (a lipopeptide fermentation product of Aspergillus nidulans or the closely related species, A. rugulosus), which undergoes deacylation (cleavage of the linoleoyl side chain) by the action of a deacylase enzyme from the bacterium Actinoplanes utahensis;[15] in three subsequent synthetic steps, including a chemical reacylation, the antifungal drug anidulafungin[14][16] is synthesized.
History
Anidulafungin was originally discovered at Lilly laboratories by Turner and Debono and licensed to Vicuron Pharmaceuticals who submitted it to the FDA.[17] Pfizer acquired the drug upon its acquisition of Vicuron in the fall of 2005.[18] Pfizer gained approval by the Food and Drug Administration (FDA) on February 21, 2006.[19]
References
- ↑ 1.0 1.1 "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary names: List 43". World Health Organization. 2000. https://www.who.int/medicines/publications/druginformation/innlists/RL43.pdf.
- ↑ 2.0 2.1 "Eraxis (anidulafungin) for Injection, for Intravenous Use. Full Prescribing Information". Roerig (Division of Pfizer, Inc.), New York, NY 10017. http://labeling.pfizer.com/ShowLabeling.aspx?id=566#section-8.1.
- ↑ "Phase 2, randomized, dose-ranging study evaluating the safety and efficacy of anidulafungin in invasive candidiasis and candidemia". Antimicrobial Agents and Chemotherapy 48 (6): 2021–2024. June 2004. doi:10.1128/AAC.48.6.2021-2024.2004. PMID 15155194.
- ↑ "In vitro activities of anidulafungin against more than 2,500 clinical isolates of Candida spp., including 315 isolates resistant to fluconazole". Journal of Clinical Microbiology 43 (11): 5425–5427. November 2005. doi:10.1128/JCM.43.11.5425-5427.2005. PMID 16272464.
- ↑ "Effectiveness of anidulafungin in eradicating Candida species in invasive candidiasis". Antimicrobial Agents and Chemotherapy 49 (11): 4795–4797. November 2005. doi:10.1128/AAC.49.11.4795-4797.2005. PMID 16251335.
- ↑ "Cost-effectiveness analysis of combination antifungal therapy with voriconazole and anidulafungin versus voriconazole monotherapy for primary treatment of invasive aspergillosis in Spain". ClinicoEconomics and Outcomes Research 9: 39–47. January 2017. doi:10.2147/CEOR.S122177. PMID 28115858.
- ↑ "Anti-Candida albicans natural products, sources of new antifungal drugs: A review". Journal de Mycologie Medicale 27 (1): 1–19. March 2017. doi:10.1016/j.mycmed.2016.10.002. PMID 27842800.
- ↑ World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. 2021. WHO/MHP/HPS/EML/2021.02.
- ↑ "Eraxis". RxList. 24 June 2009. http://www.rxlist.com/eraxis-drug.htm#cp.
- ↑ 10.0 10.1 10.2 Kofla, G.; Ruhnke, Markus (2011-04-28). "Pharmacology and metabolism of anidulafungin, caspofungin and micafungin in the treatment of invasive candidosis: review of the literature". European Journal of Medical Research 16 (4): 159–166. doi:10.1186/2047-783x-16-4-159. ISSN 0949-2321. PMID 21486730.
- ↑ "Compatibility of anidulafungin with other drugs during simulated Y-site administration". American Journal of Health-System Pharmacy 62 (8): 834–837. April 2005. doi:10.1093/ajhp/62.8.834. PMID 15821277.
- ↑ "Anidulafungin: a new echinocandin with a novel profile". Clinical Therapeutics 27 (6): 657–673. June 2005. doi:10.1016/j.clinthera.2005.06.010. PMID 16117974.
- ↑ "Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America". Clinical Infectious Diseases 46 (3): 327–360. February 2008. doi:10.1086/525258. PMID 18177225. https://pure.manchester.ac.uk/ws/files/23880159/POST-PEER-REVIEW-PUBLISHERS-DOCUMENT.PDF.
- ↑ 14.0 14.1 "Echinocandins and pneumocandins--a new antifungal class with a novel mode of action". The Journal of Antimicrobial Chemotherapy 40 (5): 611–614. November 1997. doi:10.1093/jac/dkf045. PMID 9421307.
- ↑ "Efficient bioconversion of echinocandin B to its nucleus by overexpression of deacylase genes in different host strains". Applied and Environmental Microbiology 79 (4): 1126–1133. February 2013. doi:10.1128/AEM.02792-12. PMID 23220968. Bibcode: 2013ApEnM..79.1126S.
- ↑ "Anidulafungin". EMA Europa. 2007. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/human/000788/WC500020676.pdf.
- ↑ "Vicuron Pharmaceuticals Files New Drug Application (NDA) for Anidulafungin for Treatment of Invasive Candidiasis/Candidemia". PRNewswire. 18 August 2005. http://www2.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/08-18-2005/0004090960&EDATE=.
- ↑ "Vicuron Pharmaceuticals Stockholders Approve Merger With Pfizer". PRNewswire. 15 August 2005. http://www2.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/08-15-2005/0004088704&EDATE=.
- ↑ "FDA Approves New Treatment for Fungal Infections". FDA News Release. Food and Drug Administration. 2006-02-21. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108602.htm.
 |
