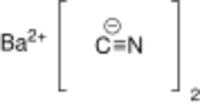
Chemistry:Barium cyanide
| |
| Names | |
|---|---|
| IUPAC name
Barium dicyanide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| Ba(CN)2 | |
| Molar mass | 189.362 g/mol |
| Appearance | white crystalline powder |
| Melting point | 600 °C (1,112 °F; 873 K) |
| 80 g/100 mL (14 °C) | |
| Solubility | Soluble in ethanol |
| Vapor pressure | 740 mmHg |
| Hazards | |
| GHS pictograms |  
|
| GHS Signal word | DANGER |
| H300, H310, H330, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+316Script error: No such module "Preview warning".Category:GHS errors, P302+352, P304+340, P316Script error: No such module "Preview warning".Category:GHS errors, P320, P321, P330, P361+364Script error: No such module "Preview warning".Category:GHS errors, P391, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Barium cyanide is a chemical compound with the formula Ba(CN)2. It is synthesized by the reaction of hydrogen cyanide and barium hydroxide in water or petroleum ether.[1] It is a white crystalline salt.
Uses
Barium cyanide is used in electroplating and other metallurgical processes.
Preparation
Barium cyanide is prepared by reacting barium hydroxide with hydrocyanic acid:
- Ba(OH)2 + 2HCN → Ba(CN)2 +2H2O
The product is crystallized from the solution.
Reactions
Barium cyanide reacts with water and carbon dioxide in air slowly, producing highly toxic hydrogen cyanide gas.[2]
When barium cyanide is heated to 300°C with steam present, the nitrogen evolves to ammonia, leaving barium formate.[3]
- Ba(CN)2 + 4 H2O = Ba(HCOO)2 + 2 NH3
Aqueous solutions of barium cyanide dissolve insoluble cyanides of some of the heavy metals forming crystalline double salts. For example, BaHg(CN)4.3H2O in needles, 2Ba(CN)2.3Hg(CN)2.23H2O in transparent octahedra, and Ba(CN)2.Hg(CN)2.HgI2.6H2O.[4]
References
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Smith, R P; Gosselin, R E (1976). "Current Concepts about the Treatment of Selected Poisonings: Nitrite, Cyanide, Sulfide, Barium, and Quinidine". Annual Review of Pharmacology and Toxicology 16: 189–99. doi:10.1146/annurev.pa.16.040176.001201. PMID 779614.
- ↑ "Barium Cyanide – a Chemical Compound" (in en-US). https://assignmentpoint.com/barium-cyanide-a-chemical-compound/.
- ↑ "Barium Cyanide, Ba(CN)2". Atomistry. http://barium.atomistry.com/barium_cyanide.html. Retrieved 2012-11-01.
 |

